I. Celanovic, D.J. Perreault, and J.G. Kassakian, “Resonant-Cavity Enhanced Thermal Emission,” Physical Review – B , Vol. 72, No. 075127, August 2005, pp. 1-6.
... also noticed with even simple planar structures such as thinfilm emitters.13 In 3D tungsten PhC,9 strong resonant enhancement near the band edge was observed which also suggests increased spatial coherence. Yet this structure shows a large emissivity outside the photonic band gap which makes it unsu ...
... also noticed with even simple planar structures such as thinfilm emitters.13 In 3D tungsten PhC,9 strong resonant enhancement near the band edge was observed which also suggests increased spatial coherence. Yet this structure shows a large emissivity outside the photonic band gap which makes it unsu ...
Advanced Physical Chemistry Professor Angelo R. Rossi http
... The sequence of situations the system goes through in passing from the initial state to the final state is called the path taken by the system. Because the intensive variables often have no values during a process, it is usually not possible to exactly specify the path a process takes in terms of th ...
... The sequence of situations the system goes through in passing from the initial state to the final state is called the path taken by the system. Because the intensive variables often have no values during a process, it is usually not possible to exactly specify the path a process takes in terms of th ...
Magnetoconvection in HCLL Blankets
... Density changes due to temperature variations, which cause convective motions, are described by the Boussinesq approximation, which states that the fluid density is a linear function of temperature in the gravitational body force term, ρβ(T − T0 )g, and constant elsewhere. Here ρ is the density at t ...
... Density changes due to temperature variations, which cause convective motions, are described by the Boussinesq approximation, which states that the fluid density is a linear function of temperature in the gravitational body force term, ρβ(T − T0 )g, and constant elsewhere. Here ρ is the density at t ...
Physical Chemistry I – review guide
... • System: The macroscopic part of the universe under study in thermodynamics • Surroundings: The parts of the universe that can interact with the system • Open System: One where transfer of matter between system and surroundings can occur • Closed System: One where transfer of matter between system ...
... • System: The macroscopic part of the universe under study in thermodynamics • Surroundings: The parts of the universe that can interact with the system • Open System: One where transfer of matter between system and surroundings can occur • Closed System: One where transfer of matter between system ...
Title - ALevelChemistryRossett
... the reaction will be feasible. Note 2: The term T∆S is temperature dependent meaning that some reactions may be feasible at one temperature but not at another. Note 3: A reaction with ∆G negative means it can go. It does not necessarily mean that it will go. There is the kinetics of a reaction to ta ...
... the reaction will be feasible. Note 2: The term T∆S is temperature dependent meaning that some reactions may be feasible at one temperature but not at another. Note 3: A reaction with ∆G negative means it can go. It does not necessarily mean that it will go. There is the kinetics of a reaction to ta ...
An availability approach to thermal energy recovery in vehicles
... Abstract: Availability is a well-established and widely recognized way of describing the work-producing potential of energy systems. A first-law analysis is helpful in setting the energy context and ensuring that energy flows balance, but it is a second-law analysis based on availability that places a ...
... Abstract: Availability is a well-established and widely recognized way of describing the work-producing potential of energy systems. A first-law analysis is helpful in setting the energy context and ensuring that energy flows balance, but it is a second-law analysis based on availability that places a ...
Temperature and Thermal Energy
... When a liquid freezes, an amount of heat, Q = –mHf, must be removed from the liquid to turn it into a solid. The negative sign indicates that the heat is transferred from the sample to the external world. In the same way, when a vapor condenses to a liquid, an amount of heat, Q = –mHv, must be remov ...
... When a liquid freezes, an amount of heat, Q = –mHf, must be removed from the liquid to turn it into a solid. The negative sign indicates that the heat is transferred from the sample to the external world. In the same way, when a vapor condenses to a liquid, an amount of heat, Q = –mHv, must be remov ...
PHY2100 Physics Practical II
... Hence, e/me can be found from a graph of Bc2 against Va . To determine the critical magnetic field Bc from the corresponding solenoid current Ic , the standard formula is used B = µ0 In √ ...
... Hence, e/me can be found from a graph of Bc2 against Va . To determine the critical magnetic field Bc from the corresponding solenoid current Ic , the standard formula is used B = µ0 In √ ...
BTD QUESTION BANK[1].
... 5. A fluid at 0.7 bar occupying 0.09 m3 is compressed reversibly to a pressure of 3.5 bar according to a law PVn = Constant .The fluid is then heated reversibly at constant volume until the pressure is 4 bar ,The specific volume is then 0.5 m3 /Kg .A reversible expansion according to a law PV2 const ...
... 5. A fluid at 0.7 bar occupying 0.09 m3 is compressed reversibly to a pressure of 3.5 bar according to a law PVn = Constant .The fluid is then heated reversibly at constant volume until the pressure is 4 bar ,The specific volume is then 0.5 m3 /Kg .A reversible expansion according to a law PV2 const ...
Thermodynamics: the Second Law
... When a change occurs, the total energy of an isolated system remains constant but it is parceled out in different ways. Can it be that the direction of change is related to the distribution of energy? The dispersal of energy Consider a ball (the system) bouncing on the floor (the surroundings). The ...
... When a change occurs, the total energy of an isolated system remains constant but it is parceled out in different ways. Can it be that the direction of change is related to the distribution of energy? The dispersal of energy Consider a ball (the system) bouncing on the floor (the surroundings). The ...
Objective of Project
... With the invention of synthetic refrigerants based mostly on a chlorofluorocarbon (CFC) chemical, safer refrigerators were possible for home and consumer use. Freon is a trademark of the Dupont Corporation and refers to these CFC, and later hydrochlorofluorocarbon (HCFC) and hydrofluorocarbon (HFC), ...
... With the invention of synthetic refrigerants based mostly on a chlorofluorocarbon (CFC) chemical, safer refrigerators were possible for home and consumer use. Freon is a trademark of the Dupont Corporation and refers to these CFC, and later hydrochlorofluorocarbon (HCFC) and hydrofluorocarbon (HFC), ...
VALIDITY OF HENRY`S LAW IN DILUTE SOLUTIONS (l)
... 1. It is only for gas absorption that the distribution equilibrium can be interpreted. The B(c) constant of the gas is positive only above the Boyle point and its maximal value for P k ?'8 50 atm (0 = 5) is 0.25. When, therefore, the gas is in the "more dilute" phase B(c) cannot be greater than' 0.2 ...
... 1. It is only for gas absorption that the distribution equilibrium can be interpreted. The B(c) constant of the gas is positive only above the Boyle point and its maximal value for P k ?'8 50 atm (0 = 5) is 0.25. When, therefore, the gas is in the "more dilute" phase B(c) cannot be greater than' 0.2 ...
this PDF file - Publications of the Serbian Chemical Society
... the metal-nonmetal transition is surely neither first nor second order, and it can occur separately or simultaneously with the liquid-vapor transition (LVT). In other words, the metal-nonmetal transition proceeds gradually, which is indicated by some changes in liquid properties such as lower conduc ...
... the metal-nonmetal transition is surely neither first nor second order, and it can occur separately or simultaneously with the liquid-vapor transition (LVT). In other words, the metal-nonmetal transition proceeds gradually, which is indicated by some changes in liquid properties such as lower conduc ...
- White Rose Research Online
... phase transition from tetragonal to orthorhombic phase. Note that the diffraction profiles can switch back to the initial positions upon the removal of the poling field, implying a reversible nature of the electric-field-induced phase transition (refer to the 3D contour figures on the right panel in ...
... phase transition from tetragonal to orthorhombic phase. Note that the diffraction profiles can switch back to the initial positions upon the removal of the poling field, implying a reversible nature of the electric-field-induced phase transition (refer to the 3D contour figures on the right panel in ...
A new, intrinsic, thermal parameter for enzymes reveals true
... fundamental third thermal parameter of enzymes, Teq, arising from a sub-second timescale, reversible, temperature-dependent equilibrium between the active enzyme and an inactive (or less active) form. Thus, at temperatures above its optimum, the decrease in enzyme activity arising from the temperatu ...
... fundamental third thermal parameter of enzymes, Teq, arising from a sub-second timescale, reversible, temperature-dependent equilibrium between the active enzyme and an inactive (or less active) form. Thus, at temperatures above its optimum, the decrease in enzyme activity arising from the temperatu ...
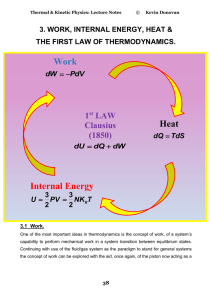
Internal Energy Work Heat
... To be reversible there cannot be any loss or dissipation in the system and therefore the piston must be frictionless. In actuality a reversible process can only be approximately achieved but the concept is extremely important in enabling the theoretical description of the thermodynamic process. In t ...
... To be reversible there cannot be any loss or dissipation in the system and therefore the piston must be frictionless. In actuality a reversible process can only be approximately achieved but the concept is extremely important in enabling the theoretical description of the thermodynamic process. In t ...
heat and temperature
... thermometric, coincides with the corresponding temperature. In this way, in the familiar mercury thermometer the height of the column of mercury is made to correspond to the temperature, or in the case of digital thermometers an electric circuit is used in which the electrical resistance is very sen ...
... thermometric, coincides with the corresponding temperature. In this way, in the familiar mercury thermometer the height of the column of mercury is made to correspond to the temperature, or in the case of digital thermometers an electric circuit is used in which the electrical resistance is very sen ...
Acetic acid-water system thermodynamical correlation of vapor
... tetramer is not considered and isobaric data are directly handled : this is equivalent to neglecting the temperature effect on the activity coefficients. This is justified by the facts that the influence of both the tetramer formation and the temperature effect do not introduce serious error, on one ...
... tetramer is not considered and isobaric data are directly handled : this is equivalent to neglecting the temperature effect on the activity coefficients. This is justified by the facts that the influence of both the tetramer formation and the temperature effect do not introduce serious error, on one ...
Magnetostriction of GdAg2, PrFe4As12, and GdVO3 measured with
... nuts, and (r) needle bearing positions (Ag). ................................................................ 17 Figure 2.3: Capacitance dilatometer mounted on the holder........................................ 18 Figure 2.4: Schematic drawing of the capacitance dilatometer. The dashed lines represe ...
... nuts, and (r) needle bearing positions (Ag). ................................................................ 17 Figure 2.3: Capacitance dilatometer mounted on the holder........................................ 18 Figure 2.4: Schematic drawing of the capacitance dilatometer. The dashed lines represe ...
ee11042602mpt3.mov 110426ph423main3.mov Example of the
... conceptually about the partial derivatives they encounter in thermodynamic contexts. The instructor, David Roundy, invites students to think about how they would measure the quantities represented by a partial derivative. During this class session, Day 2 of the thermodynamics paradigm, he introduces ...
... conceptually about the partial derivatives they encounter in thermodynamic contexts. The instructor, David Roundy, invites students to think about how they would measure the quantities represented by a partial derivative. During this class session, Day 2 of the thermodynamics paradigm, he introduces ...









![BTD QUESTION BANK[1].](http://s1.studyres.com/store/data/009330461_1-f5de3108f7a7a17ebe3a8cbd391865db-300x300.png)