Experimental skills and abilities
... 1 The evaporation process should be done very slowly. This is because sugar can easily char as it solidifies around the sides of the evaporating basin during the evaporating process. Also the crystallisation will require a lot longer for crystals to form from the concentrated solution and may need ...
... 1 The evaporation process should be done very slowly. This is because sugar can easily char as it solidifies around the sides of the evaporating basin during the evaporating process. Also the crystallisation will require a lot longer for crystals to form from the concentrated solution and may need ...
chemical change
... which is different from the original reactant or reactant, this is often accompanied by changes in energy, which are measured as temperature changes. Thus for the reaction of the silver metal sodium with the green/yellow gas chlorine, the product is a white crystalline solid, which looks very differ ...
... which is different from the original reactant or reactant, this is often accompanied by changes in energy, which are measured as temperature changes. Thus for the reaction of the silver metal sodium with the green/yellow gas chlorine, the product is a white crystalline solid, which looks very differ ...
SOLUBILITY RULES FOR IONIC COMPOUNDS IN WATER
... occurring. Indicate the phase of each reactant and product. (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal i ...
... occurring. Indicate the phase of each reactant and product. (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal i ...
Questions
... Recalculate a value of the mean making clear which titres you choose and giving your answer to an appropriate number of significant figures. ...
... Recalculate a value of the mean making clear which titres you choose and giving your answer to an appropriate number of significant figures. ...
Mathematical Operations
... A smaller value of s indicates a higher precision, meaning that the data is more closely clustered around the average. The standard deviation has a statistical significance. Thus, if a large number of measurements is made, 68% of the measured values is expected to be within one standard deviation of ...
... A smaller value of s indicates a higher precision, meaning that the data is more closely clustered around the average. The standard deviation has a statistical significance. Thus, if a large number of measurements is made, 68% of the measured values is expected to be within one standard deviation of ...
www.XtremePapers.com UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level 5070/03
... A solid M is an alloy of iron and several other metals. Solution P has been prepared by dissolving 6.00 g of M in dilute sulphuric acid forming a mixture of metal sulphates and then adding water until the final volume is 1.00 dm3. You are to determine the percentage of iron in M by titrating solutio ...
... A solid M is an alloy of iron and several other metals. Solution P has been prepared by dissolving 6.00 g of M in dilute sulphuric acid forming a mixture of metal sulphates and then adding water until the final volume is 1.00 dm3. You are to determine the percentage of iron in M by titrating solutio ...
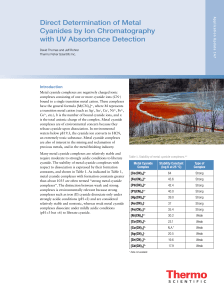
Direct Determination of Metal Cyanides by Ion
... Use high-quality water of high resistivity (18.0 MΩ-cm or better) that contains as little dissolved carbon dioxide as possible. Biological contamination should be absent. Sodium hydroxide eluent should be prepared with 50% (w/w) sodium hydroxide solution. Sodium hydroxide pellets are covered with a ...
... Use high-quality water of high resistivity (18.0 MΩ-cm or better) that contains as little dissolved carbon dioxide as possible. Biological contamination should be absent. Sodium hydroxide eluent should be prepared with 50% (w/w) sodium hydroxide solution. Sodium hydroxide pellets are covered with a ...
1. A pharmacy analyst supervises the state of a refractometer. For its
... The end point is established potentiometrically or using crystal violet as indicator. For example, phthivazid: ...
... The end point is established potentiometrically or using crystal violet as indicator. For example, phthivazid: ...
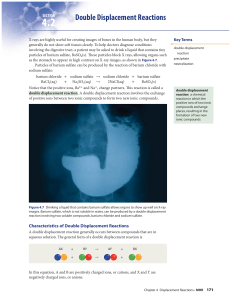
Double Displacement Reactions
... double displacement reaction produces a gas. Give the general form of each reaction. ...
... double displacement reaction produces a gas. Give the general form of each reaction. ...
handout 4
... per 1.00 gram of baking soda? If all the baking soda reacts, how many grams of water is produced? ...
... per 1.00 gram of baking soda? If all the baking soda reacts, how many grams of water is produced? ...
ch6 - ChemistryVCE
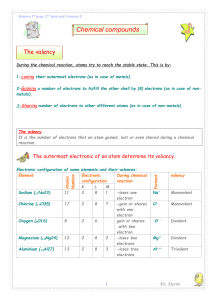
... Explain why potassium chloride has the formula KCl whereas the formula of calcium chloride is CaCl2. A7. The electronic configuration of an atom of potassium is 1s22s22p63s23p64s1 and that of an atom of chlorine is 1s22s22p63s23p5. Because an atom of K has one more electron than an atom of a noble g ...
... Explain why potassium chloride has the formula KCl whereas the formula of calcium chloride is CaCl2. A7. The electronic configuration of an atom of potassium is 1s22s22p63s23p64s1 and that of an atom of chlorine is 1s22s22p63s23p5. Because an atom of K has one more electron than an atom of a noble g ...
Document
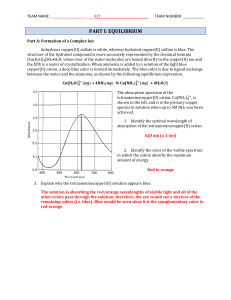
... at this temperature with [N2] = [O2] = 0.100 Mand [NO] = 0.0200 M in a 2.00 liter flask. If 0.120 mol of NO is suddenly added to the reaction mixture what will be the concentrations of all species when equilibrium is re-established? 22. CO2(g) = 2(g) A 1.00 mole sample of CO2 is placed in a 1.00 L c ...
... at this temperature with [N2] = [O2] = 0.100 Mand [NO] = 0.0200 M in a 2.00 liter flask. If 0.120 mol of NO is suddenly added to the reaction mixture what will be the concentrations of all species when equilibrium is re-established? 22. CO2(g) = 2(g) A 1.00 mole sample of CO2 is placed in a 1.00 L c ...
Answers - Scioly.org
... The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chemical formula C6H12O2, which would have the same molecular mass seen from the mass spectrum and the same empirical formula shown in the combus ...
... The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chemical formula C6H12O2, which would have the same molecular mass seen from the mass spectrum and the same empirical formula shown in the combus ...
Unit 8 Packet
... reactant or product and percent yield using particle models from a balanced chemical equation and amount of one substance in the reaction. Given quantities of multiple reactants, students will be able to determine and use the limiting reactant. ...
... reactant or product and percent yield using particle models from a balanced chemical equation and amount of one substance in the reaction. Given quantities of multiple reactants, students will be able to determine and use the limiting reactant. ...
full text pdf
... This paper presents the results of study on structural parameters (particle size, surface area, pore volume) and the sorption ability of mechanically and thermally activated sodium bicarbonate. The sorption ability of the modified sorbent was evaluated by: partial and overall SO2 removal efficiency, ...
... This paper presents the results of study on structural parameters (particle size, surface area, pore volume) and the sorption ability of mechanically and thermally activated sodium bicarbonate. The sorption ability of the modified sorbent was evaluated by: partial and overall SO2 removal efficiency, ...
Acids ,Bases and Salts
... This dissociation/ionization makes aqueous ammonia to: (i)turn litmus paper/solution blue. (ii)have pH 8/9/10/11 (iii)be a good electrical conductor (iv)react with acids to form ammonium salt and water only. NH4OH(aq) + HCl(aq) -> NH4Cl(aq) + H2O(l) (d)Ammonia gas dissolves in methylbenzene/benzene ...
... This dissociation/ionization makes aqueous ammonia to: (i)turn litmus paper/solution blue. (ii)have pH 8/9/10/11 (iii)be a good electrical conductor (iv)react with acids to form ammonium salt and water only. NH4OH(aq) + HCl(aq) -> NH4Cl(aq) + H2O(l) (d)Ammonia gas dissolves in methylbenzene/benzene ...
Chemistry Spell check on
... by the fermentation of biological material under anaerobic conditions? A Hydrogen B Methane C Methanol D Petrol ...
... by the fermentation of biological material under anaerobic conditions? A Hydrogen B Methane C Methanol D Petrol ...
Document
... by the fermentation of biological material under anaerobic conditions? A Hydrogen B Methane C Methanol D Petrol ...
... by the fermentation of biological material under anaerobic conditions? A Hydrogen B Methane C Methanol D Petrol ...
THE s -BLOCK ELEMENTS
... These are so called because they form hydroxides on reaction with water which are strongly alkaline in nature. The elements of Group 2 include beryllium, magnesium, calcium, strontium, barium and radium. These elements with the exception of beryllium are commonly known as the alkaline earth metals. ...
... These are so called because they form hydroxides on reaction with water which are strongly alkaline in nature. The elements of Group 2 include beryllium, magnesium, calcium, strontium, barium and radium. These elements with the exception of beryllium are commonly known as the alkaline earth metals. ...
Net Ionic Equation with solubility rules
... information about quantities of each reactant! Remember which acids are strong (ionize completely) and which are weak (write as molecule). Sulfuric acid (strong acid) can be written as H+ and SO42- or as H+ and HSO4-. Ex. A solution of sulfuric acid is added to a solution of barium hydroxide until t ...
... information about quantities of each reactant! Remember which acids are strong (ionize completely) and which are weak (write as molecule). Sulfuric acid (strong acid) can be written as H+ and SO42- or as H+ and HSO4-. Ex. A solution of sulfuric acid is added to a solution of barium hydroxide until t ...
Chemistry of METALS
... thus blocking OH- ions from being discharged at the anode. (ii)Concentration of Na+(aq) ions is higher than H+ ions causing overvoltage thus blocking H+ ions from being discharged at the cathode. IV. Name the products of electrolysis in the flowing mercury-cathode cell. (i)Mercury cathode; Sodium me ...
... thus blocking OH- ions from being discharged at the anode. (ii)Concentration of Na+(aq) ions is higher than H+ ions causing overvoltage thus blocking H+ ions from being discharged at the cathode. IV. Name the products of electrolysis in the flowing mercury-cathode cell. (i)Mercury cathode; Sodium me ...
CHM 22 Test 2Take-homeKey Student Name
... B. Sodium chlorate decomposes to form sodium chloride and oxygen. C. Phosphoric acid and potassium hydroxide yields potassium phosphate and water. D. Fluorine and potassium bromide yields potassium fluoride and bromine. ...
... B. Sodium chlorate decomposes to form sodium chloride and oxygen. C. Phosphoric acid and potassium hydroxide yields potassium phosphate and water. D. Fluorine and potassium bromide yields potassium fluoride and bromine. ...
Science 1st prep. 2nd term unit 1 lesson 2 During the chemical
... 14- the chemical formula of sodium carbonate is ………………………….. and it consists of …………. Atoms of ……………………… different elements. 15- A compound has a chemical formula XO2, so the valency of X is …………………………….. ...
... 14- the chemical formula of sodium carbonate is ………………………….. and it consists of …………. Atoms of ……………………… different elements. 15- A compound has a chemical formula XO2, so the valency of X is …………………………….. ...
s-BLOCK ELEMENTS - einstein classes
... alkali metals. Because they aremore closely packed due to increasing nuclear charge. (iii) Atomic size : The atomic size of alkaline earth metals are smaller than the corresponding alkali metals. Alkaline earth metals have a higher nuclear charge and therefore, electrons are attracted more towards t ...
... alkali metals. Because they aremore closely packed due to increasing nuclear charge. (iii) Atomic size : The atomic size of alkaline earth metals are smaller than the corresponding alkali metals. Alkaline earth metals have a higher nuclear charge and therefore, electrons are attracted more towards t ...
Sodium hydroxide

Sodium hydroxide (NaOH), also known as lye and caustic soda, is an inorganic compound. It is a white solid and highly caustic metallic base and alkali salt which is available in pellets, flakes, granules, and as prepared solutions at a number of different concentrations. Sodium hydroxide forms an approximately 50% (by weight) saturated solution with water.Sodium hydroxide is soluble in water, ethanol and methanol. This alkali is deliquescent and readily absorbs moisture and carbon dioxide in air.Sodium hydroxide is used in many industries, mostly as a strong chemical base in the manufacture of pulp and paper, textiles, drinking water, soaps and detergents and as a drain cleaner. Worldwide production in 2004 was approximately 60 million tonnes, while demand was 51 million tonnes.