IT IS ELEMENTARY - the OLLI at UCI Blog
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
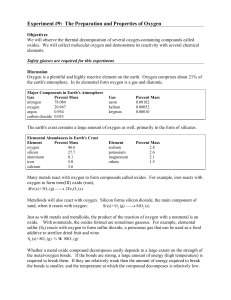
Experiment # 9 Properties of Oxygen
... oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterilize dried fruit and wine. S8 (s) +8O 2 (g) ¾ ¾ ® 8SO 2 (g) ...
... oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterilize dried fruit and wine. S8 (s) +8O 2 (g) ¾ ¾ ® 8SO 2 (g) ...
atomic number
... Sometimes, when they tried to react substances together, nothing happened! Substances that DO NOT react are Inert They found that most materials will react to form new substances. These elements are said to be chemically active (reactive) Oxygen is very reactive, so is hydrogen which we will look a ...
... Sometimes, when they tried to react substances together, nothing happened! Substances that DO NOT react are Inert They found that most materials will react to form new substances. These elements are said to be chemically active (reactive) Oxygen is very reactive, so is hydrogen which we will look a ...
Ch. 3 - My CCSD
... Sometimes, when they tried to react substances together, nothing happened! Substances that DO NOT react are Inert They found that most materials will react to form new substances. These elements are said to be chemically active (reactive) Oxygen is very reactive, so is hydrogen which we will look a ...
... Sometimes, when they tried to react substances together, nothing happened! Substances that DO NOT react are Inert They found that most materials will react to form new substances. These elements are said to be chemically active (reactive) Oxygen is very reactive, so is hydrogen which we will look a ...
Cycles of Matter
... • Rain falls and is soaked up by the ground, or it pools where it’s drank, or it is absorbed by plants, or it will sit and evaporate back up into the atmosphere. • Since it’s a cycle there is no starting point. • We’ll start with water forming in the atmosphere. ...
... • Rain falls and is soaked up by the ground, or it pools where it’s drank, or it is absorbed by plants, or it will sit and evaporate back up into the atmosphere. • Since it’s a cycle there is no starting point. • We’ll start with water forming in the atmosphere. ...
Lesson 1 Reversible reactions and equilibrium
... Plants get time to take in each application. 2. More soluble fertiliser – bad It will dissolve in rain and wash into local drains. 3. Avoid applying it before rain is due – good This means it won’t dissolve in the rain and wash into drains. 4. Grow a quick crop of legumes – good But depends on timin ...
... Plants get time to take in each application. 2. More soluble fertiliser – bad It will dissolve in rain and wash into local drains. 3. Avoid applying it before rain is due – good This means it won’t dissolve in the rain and wash into drains. 4. Grow a quick crop of legumes – good But depends on timin ...
Atomic Structure
... Complete Pre-Lab for “Mass of Candium” Lab found on page 120 of the textbook. Lab will be on Friday, November 22nd. ...
... Complete Pre-Lab for “Mass of Candium” Lab found on page 120 of the textbook. Lab will be on Friday, November 22nd. ...
File
... element with a charge due to the gain or loss of electrons. Ions are isotopes are both expressed with specific forms of notation. Ions will have a + or – sign next to the chemical symbol of the element. Magnesium has a 2+ charge, indicating that it has lost two electrons. Bromine has a – charge, ind ...
... element with a charge due to the gain or loss of electrons. Ions are isotopes are both expressed with specific forms of notation. Ions will have a + or – sign next to the chemical symbol of the element. Magnesium has a 2+ charge, indicating that it has lost two electrons. Bromine has a – charge, ind ...
Chapter 2_Application Problems
... correct according to Dalton’s model of the atom • Because the mass ratio of Fe:O in wüsite is 1.5 times larger than the Fe:O ratio in hematite, there must be 1.5 Fe atoms in a unit of wüsite and 1 Fe atom in a unit of hematite – incorrect; according to Dalton, atoms must combine in small whole-numbe ...
... correct according to Dalton’s model of the atom • Because the mass ratio of Fe:O in wüsite is 1.5 times larger than the Fe:O ratio in hematite, there must be 1.5 Fe atoms in a unit of wüsite and 1 Fe atom in a unit of hematite – incorrect; according to Dalton, atoms must combine in small whole-numbe ...
Isotopes are atoms of the same element that have different masses
... Isotopes are atoms of the same element that have different masses due to different numbers of neutrons. See the example below for two isotopes of oxygen. One has 8 neutrons while the other has 10 neutrons. Therefore, the masses are 16 amu and 18 amu respectively. ...
... Isotopes are atoms of the same element that have different masses due to different numbers of neutrons. See the example below for two isotopes of oxygen. One has 8 neutrons while the other has 10 neutrons. Therefore, the masses are 16 amu and 18 amu respectively. ...
Document
... What can geochemistry do in 2004? • The earth is made of 90 or so chemical elements, about 30 w/isotopic variations • Chemical/isotopic characteristics can be tied to geological processes - mantle isotopic chemistry is a tracer • We can tell where a particular piece of mantle has been in the past a ...
... What can geochemistry do in 2004? • The earth is made of 90 or so chemical elements, about 30 w/isotopic variations • Chemical/isotopic characteristics can be tied to geological processes - mantle isotopic chemistry is a tracer • We can tell where a particular piece of mantle has been in the past a ...
AP Chemistry Name: Ch.2 – The Nuclear Atom Date: Period:
... Writing formulas and naming compounds can be confusing because there are different types of compounds that follow different rules. Additionally, some compounds (H2O, NH3, CH4, etc.) simply have common names that must be memorized. The two types of compounds we will focus on first are ionic compounds ...
... Writing formulas and naming compounds can be confusing because there are different types of compounds that follow different rules. Additionally, some compounds (H2O, NH3, CH4, etc.) simply have common names that must be memorized. The two types of compounds we will focus on first are ionic compounds ...
Unit 1: Stoichiometry
... There are two naturally occurring isotopes of chlorine: chlorine‐35 and chlorine‐37. The atomic mass of this element is a combination of the two isotopes. The relative abundance of chlorine atoms in nature is 75% chlorine‐35 and 25% chlorine‐37. Average atomic mass is the weighted average of the ato ...
... There are two naturally occurring isotopes of chlorine: chlorine‐35 and chlorine‐37. The atomic mass of this element is a combination of the two isotopes. The relative abundance of chlorine atoms in nature is 75% chlorine‐35 and 25% chlorine‐37. Average atomic mass is the weighted average of the ato ...
(or radioactive isotopes).
... • In a cyclotron, target nuclei are bombarded with positive particles such as protons or atoms of helium or carbon, at great speed, until they fuse together on collision. • Very high speeds are necessary because the positive particles have to fuse with a positive nucleus and the two experience repul ...
... • In a cyclotron, target nuclei are bombarded with positive particles such as protons or atoms of helium or carbon, at great speed, until they fuse together on collision. • Very high speeds are necessary because the positive particles have to fuse with a positive nucleus and the two experience repul ...
EPSc 446 STABLE ISOTOPE GEOCHEMISTRY Instructor: Bob Criss
... 5) !Abundance peaks at C, O, Fe. ! High for nuclides with Mass # A of multiples of 4 = α particle mass! Bulk Earth > 92 wt % Fe, O, Si, Mg {56Fe, 16O, 28Si, 24Mg} ! ...
... 5) !Abundance peaks at C, O, Fe. ! High for nuclides with Mass # A of multiples of 4 = α particle mass! Bulk Earth > 92 wt % Fe, O, Si, Mg {56Fe, 16O, 28Si, 24Mg} ! ...
OC 583- ISOTOPE BIGEOCHEMISTRY
... a. Learn how to use isotopic measurements to improve our understanding of biogeochemical cycles. b. Large Range of Applications of Isotopes -common isotopes: carbon, nitrogen, hydrogen, oxygen and sulfur isotopes -fundamental cycles: water, CO2, nutrients, food webs, climate, trace gases, etc. -many ...
... a. Learn how to use isotopic measurements to improve our understanding of biogeochemical cycles. b. Large Range of Applications of Isotopes -common isotopes: carbon, nitrogen, hydrogen, oxygen and sulfur isotopes -fundamental cycles: water, CO2, nutrients, food webs, climate, trace gases, etc. -many ...
Atomic structure Atomic masses
... Relative atomic mass, Ar: the weighted mean mass of an atom of an element compared with 1/12 of the the mass of an atom of carbon-12 The term ‘weighted mean mass’ is used to account for the contribution made by each isotope to the overall mass of an element. The contribution made by an isotope to th ...
... Relative atomic mass, Ar: the weighted mean mass of an atom of an element compared with 1/12 of the the mass of an atom of carbon-12 The term ‘weighted mean mass’ is used to account for the contribution made by each isotope to the overall mass of an element. The contribution made by an isotope to th ...
Environmental Chemistry
... Recall, in our previous discussion about non-metal oxides, oxides of sulfur and nitrogen form strong acids (completely dissociate) that lead to acid rain. SO2 + H2O H2SO3 (sulfurous acid) SO3 + H2O H2SO4 (sulfuric acid) 2NO2 + H2O HNO3 + HNO2 (nitric and nitrous acid) These non-metal oxides ar ...
... Recall, in our previous discussion about non-metal oxides, oxides of sulfur and nitrogen form strong acids (completely dissociate) that lead to acid rain. SO2 + H2O H2SO3 (sulfurous acid) SO3 + H2O H2SO4 (sulfuric acid) 2NO2 + H2O HNO3 + HNO2 (nitric and nitrous acid) These non-metal oxides ar ...
GHW - Louisiana Tech University
... stoichiometric coefficient found in chemical equation and why do they react in simple ratios of moles? Could you measure moles directly measured using a chemical balances which give readings in grams? How is this problem get boiled down to: converting amount of a substance from grams to mole, and la ...
... stoichiometric coefficient found in chemical equation and why do they react in simple ratios of moles? Could you measure moles directly measured using a chemical balances which give readings in grams? How is this problem get boiled down to: converting amount of a substance from grams to mole, and la ...
Jeopardy - SchoolRack
... there the cathode rays were negative but he knew that not all matter was negative so then he determined that there must be a positive charge. ...
... there the cathode rays were negative but he knew that not all matter was negative so then he determined that there must be a positive charge. ...
Isotope analysis

Isotope analysis is the identification of isotopic signature, the distribution of certain stable isotopes and chemical elements within chemical compounds. This can be applied to a food web to make it possible to draw direct inferences regarding diet, trophic level, and subsistence. Variations in isotope ratios from isotopic fractionation are measured using mass spectrometry, which separates the different isotopes of an element on the basis of their mass-to-charge ratio.The ratios of isotopic oxygen are also differentially affected by global weather patterns and regional topography as moisture is transported. Areas of lower humidity cause the preferential loss of 18O water in the form of vapor and precipitation. Furthermore, evaporated 16O water returns preferentially to the atmospheric system as it evaporates and 18O remains in liquid form or is incorporated into the body water of plants and animals.