metIstudyguide_S16
... 2. What layer of atmosphere contains weather? 3. Which layer of atmosphere is most thin? 4. What is the most abundant gas in the atmosphere? 5. Put the following gases in order from most abundant to least: O2, N2, CO2, and Ar 6. Increased altitudes have _____________ pressure because _______________ ...
... 2. What layer of atmosphere contains weather? 3. Which layer of atmosphere is most thin? 4. What is the most abundant gas in the atmosphere? 5. Put the following gases in order from most abundant to least: O2, N2, CO2, and Ar 6. Increased altitudes have _____________ pressure because _______________ ...
Teacher Vocabulary Guide
... IR Satellite: an image taken in the infrared band of light and showing relative warmth of objects. Colder objects are brighter and warmer objects are darker. Lower layers of clouds, generally warmer and lower in altitude, are colored gray. Colder and generally higher clouds tops are highlighted in c ...
... IR Satellite: an image taken in the infrared band of light and showing relative warmth of objects. Colder objects are brighter and warmer objects are darker. Lower layers of clouds, generally warmer and lower in altitude, are colored gray. Colder and generally higher clouds tops are highlighted in c ...
metIstudyguide F14
... 2. What layer of atmosphere contains weather? 3. Which layer of atmosphere is most thin? 4. What is the most abundant gas in the atmosphere? 5. Put the following gases in order from most abundant to least: O2, N2, CO2, and Ar 6. Increased altitudes have _____________ pressure because _______________ ...
... 2. What layer of atmosphere contains weather? 3. Which layer of atmosphere is most thin? 4. What is the most abundant gas in the atmosphere? 5. Put the following gases in order from most abundant to least: O2, N2, CO2, and Ar 6. Increased altitudes have _____________ pressure because _______________ ...
Matter – Chemical and Physical Properties
... atomic bonding – linking together of atoms to form larger structures, including solids Types of Bonds Covalent bond – one or more electrons are shared between two atoms Ionic bond – one or more electrons are transferred from one atom to another Metallic bond – described as a “sea of electrons”. Elec ...
... atomic bonding – linking together of atoms to form larger structures, including solids Types of Bonds Covalent bond – one or more electrons are shared between two atoms Ionic bond – one or more electrons are transferred from one atom to another Metallic bond – described as a “sea of electrons”. Elec ...
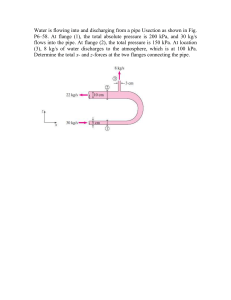
Chapter 2 Homework 1. Define Pressure Pressure is the force per
... increase in weight on your body from the water. The pressure is the weight of the water on the surface area of your body. 11. Why do we take the water off the bottom of the dam in a hydroelectric power plant? The deeper the water the greater the pressure, the greater pressure means an increase in fo ...
... increase in weight on your body from the water. The pressure is the weight of the water on the surface area of your body. 11. Why do we take the water off the bottom of the dam in a hydroelectric power plant? The deeper the water the greater the pressure, the greater pressure means an increase in fo ...