The Mole - Cloudfront.net
... unpopped popcorn kernels, and spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. • If we were able to count atoms at the rate of 10 million per second, it would take about 2 billion years to count the atoms in one mole. ...
... unpopped popcorn kernels, and spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. • If we were able to count atoms at the rate of 10 million per second, it would take about 2 billion years to count the atoms in one mole. ...
9/6/12
... - Compounds have characteristic in properties and composition - Compounds can be represented by an abbreviation or Formula o A formula has subscripts, which represent the number of different atoms in the compound. o Example: H2O has 2 hydrogen atoms and 1 oxygen atoms - Molecular formulas give infor ...
... - Compounds have characteristic in properties and composition - Compounds can be represented by an abbreviation or Formula o A formula has subscripts, which represent the number of different atoms in the compound. o Example: H2O has 2 hydrogen atoms and 1 oxygen atoms - Molecular formulas give infor ...
Name: Moles Convert 26.33 g Si to moles. Convert 3.00 mol Sn to
... Measure the mass of the sample of Silicon (Si). How many Si atoms are in your sample? How many moles are in the sample of aluminum foil? Weigh out 0.20 mol of NaCl (salt). I placed ______g of sugar in ______mL of water. What is the concentration of the solution? This is measured in Molarity (M). 5. ...
... Measure the mass of the sample of Silicon (Si). How many Si atoms are in your sample? How many moles are in the sample of aluminum foil? Weigh out 0.20 mol of NaCl (salt). I placed ______g of sugar in ______mL of water. What is the concentration of the solution? This is measured in Molarity (M). 5. ...
Elements, Compounds and Mixtures
... Questions 1. Describe the discovery that led scientists to question John Dalton’s model of the atom ad to favor J.J. Thomson’s model. 2. What experimental findings are the basis for the 1909 model of the atom? 3. What shortcomings in the atomic model of Ernest Rutherford led to the development of Ni ...
... Questions 1. Describe the discovery that led scientists to question John Dalton’s model of the atom ad to favor J.J. Thomson’s model. 2. What experimental findings are the basis for the 1909 model of the atom? 3. What shortcomings in the atomic model of Ernest Rutherford led to the development of Ni ...
Superconductivity Is Pair Work - Max-Planck
... spins of the two electrons either subtract to a total spin of zero or, more rarely, they also add up to one. Quantum particles with integer spin, however, belong to the group of particles known as bosons. Bosons are so sociable that they all like to condense together into one quantum state if the he ...
... spins of the two electrons either subtract to a total spin of zero or, more rarely, they also add up to one. Quantum particles with integer spin, however, belong to the group of particles known as bosons. Bosons are so sociable that they all like to condense together into one quantum state if the he ...
The Mole, Molar Mass and Avogadro`s Number
... The Mole, Molar Mass and Avogadro’s Number Mass Relationships in Chemical Reactions ...
... The Mole, Molar Mass and Avogadro’s Number Mass Relationships in Chemical Reactions ...
Chapter 6 Notes Packet - Mahtomedi High School
... the same number of atoms. This number would be ___________________, which is called a _______ in chemistry. This number is called __________ number for the scientist who discovered it. This information has given us two conversion factors that we will use. The first conversion factor is that the atom ...
... the same number of atoms. This number would be ___________________, which is called a _______ in chemistry. This number is called __________ number for the scientist who discovered it. This information has given us two conversion factors that we will use. The first conversion factor is that the atom ...
Liquid-gas transition of neon in quasi-one
... The PIMC method allows the calculation of quantum nondynamical properties of finite-temperature systems by mapping the quantum system onto an equivalent classical model in which each atom is described by a certain number of beads connected by springs. The beads interact with each other by a fraction ...
... The PIMC method allows the calculation of quantum nondynamical properties of finite-temperature systems by mapping the quantum system onto an equivalent classical model in which each atom is described by a certain number of beads connected by springs. The beads interact with each other by a fraction ...
06.1 - Chemical formulas and composition stoichiometry
... A 0.1647 g sample of hydrocarbon is burned in a C-H combustion train to produce 0.4931 g of CO2 and 0.2691 g of H2O. Determine the masses of C and H in the sample and the percentages of these elements in this hydrocarbon. 1. With a proportion, we use the observed masses to determine the masses of C ...
... A 0.1647 g sample of hydrocarbon is burned in a C-H combustion train to produce 0.4931 g of CO2 and 0.2691 g of H2O. Determine the masses of C and H in the sample and the percentages of these elements in this hydrocarbon. 1. With a proportion, we use the observed masses to determine the masses of C ...
Document
... 1cal is the energy needed to increase the temperature of 1g of water by 1℃, and this is about 4.184 joules. A7. The enthalpy change of a chemical reaction. The law of hess. Enthalpy, symbolized by H, is the change of the reaction heat at constant pressure. Enthalpy change is defined as the enthalpy ...
... 1cal is the energy needed to increase the temperature of 1g of water by 1℃, and this is about 4.184 joules. A7. The enthalpy change of a chemical reaction. The law of hess. Enthalpy, symbolized by H, is the change of the reaction heat at constant pressure. Enthalpy change is defined as the enthalpy ...
File
... • Chemical equations balance on an atomic level, not molecular. • You cannot change the formula of a substance, i.e. if the equation has NH3 you cannot change this you can only put a number in front of it, 2NH3, increasing the number of N’s and the number of H’s. • Never change the subscripts (small ...
... • Chemical equations balance on an atomic level, not molecular. • You cannot change the formula of a substance, i.e. if the equation has NH3 you cannot change this you can only put a number in front of it, 2NH3, increasing the number of N’s and the number of H’s. • Never change the subscripts (small ...
WHAT IS A MOLE?
... the same temperature contain equal numbers of molecules Josef Loschmidt, an Austrian high school teacher,in 1865, calculated the number of molecules in one cm3 of gaseous substance to be somewhere around 2.6 x 1019 J.Perrin (1908) estimated the value to be between 6.5 x 1023 and 6.9 x 1023 Rut ...
... the same temperature contain equal numbers of molecules Josef Loschmidt, an Austrian high school teacher,in 1865, calculated the number of molecules in one cm3 of gaseous substance to be somewhere around 2.6 x 1019 J.Perrin (1908) estimated the value to be between 6.5 x 1023 and 6.9 x 1023 Rut ...
Chapter 3: The Structure of Crystalline Solids
... • Reasons for dense packing: - Typically, only one element is present, so all atomic radii are the same. - Metallic bonding is not directional. - Nearest neighbor distances tend to be small in order to lower bond energy. - Electron cloud shields cores from each other ...
... • Reasons for dense packing: - Typically, only one element is present, so all atomic radii are the same. - Metallic bonding is not directional. - Nearest neighbor distances tend to be small in order to lower bond energy. - Electron cloud shields cores from each other ...
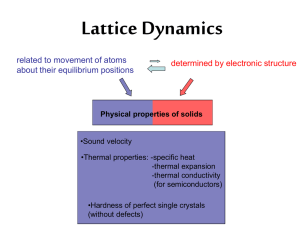
Introduction to Lattice Dynamics
... • Consider the regular lattice of atoms in a uniform solid material. • There should be energy associated with the vibrations of these atoms. • But they are tied together with bonds, so they can't vibrate independently. • The vibrations take the form of collective modes which propagate through the ma ...
... • Consider the regular lattice of atoms in a uniform solid material. • There should be energy associated with the vibrations of these atoms. • But they are tied together with bonds, so they can't vibrate independently. • The vibrations take the form of collective modes which propagate through the ma ...
Changes of State
... The freezing point of a substance is the temperature at which a liquid changes into a solid. Freezing is the opposite process of melting. Freezing and melting occur at the same temperature. Freezing is an exothermic change because energy is taken out of (exits) the substance as it changes. ...
... The freezing point of a substance is the temperature at which a liquid changes into a solid. Freezing is the opposite process of melting. Freezing and melting occur at the same temperature. Freezing is an exothermic change because energy is taken out of (exits) the substance as it changes. ...
Physical and Chemical Changes
... GIRLS: (in high pitch voice) It’s physical. BOYS: Let me see some sublimation. GIRLS: (in high pitch voice) It’s physical. Phase changes are physical, physical… Boiling is physical… Boiling is physical… BOYS: Let me see some deposition. GIRLS: (in high pitch voice) It’s physical BOYS: Let me see som ...
... GIRLS: (in high pitch voice) It’s physical. BOYS: Let me see some sublimation. GIRLS: (in high pitch voice) It’s physical. Phase changes are physical, physical… Boiling is physical… Boiling is physical… BOYS: Let me see some deposition. GIRLS: (in high pitch voice) It’s physical BOYS: Let me see som ...
The Ideal Gas Laws
... After this chapter, students will: Know what a “mole” is Understand and apply atomic mass, the atomic mass unit, and Avogadro’s number Understand how an ideal gas differs from real ...
... After this chapter, students will: Know what a “mole” is Understand and apply atomic mass, the atomic mass unit, and Avogadro’s number Understand how an ideal gas differs from real ...
Atomic Bonding - New Academic Science
... accurately to determine the position of the electron, but it is possible to calculate the probability of finding the electron at any point around the nucleus. Within a hydrogen atom the probability of distribution of electrons is spherical around the nucleus and it is possible to draw a spherical bo ...
... accurately to determine the position of the electron, but it is possible to calculate the probability of finding the electron at any point around the nucleus. Within a hydrogen atom the probability of distribution of electrons is spherical around the nucleus and it is possible to draw a spherical bo ...
Section3a - Lyle School of Engineering
... – An energy is associated with grain boundaries, as explained in Section 4.5. As grains increase in size, the total boundary area decreases, yielding an attendant reduction in the total energy; this is the driving force for grain growth. – Grain growth occurs by the migration of grain boundaries. Ob ...
... – An energy is associated with grain boundaries, as explained in Section 4.5. As grains increase in size, the total boundary area decreases, yielding an attendant reduction in the total energy; this is the driving force for grain growth. – Grain growth occurs by the migration of grain boundaries. Ob ...
Phonons: Lattice Dynamics
... • Consider the regular lattice of atoms in a uniform solid material. • There should be energy associated with the vibrations of these atoms. • But they are tied together with bonds, so they can't vibrate independently. • The vibrations take the form of collective modes which propagate through the ma ...
... • Consider the regular lattice of atoms in a uniform solid material. • There should be energy associated with the vibrations of these atoms. • But they are tied together with bonds, so they can't vibrate independently. • The vibrations take the form of collective modes which propagate through the ma ...
1 mole
... What do you want in the end? Moles of C Use the conversion factor given: 1 mole Atomic mass 24 g of C x 1 mol of C = 2.0 mol of Carbon 12.011 g of C ...
... What do you want in the end? Moles of C Use the conversion factor given: 1 mole Atomic mass 24 g of C x 1 mol of C = 2.0 mol of Carbon 12.011 g of C ...
On-surface photo-dissociation of C
... XPS measurements were performed at the I311 beamline at the MAX-IV laboratory (Lund, Sweden). The spectra were obtained in normal incidence geometry, the photoelectron detection angle being 55° off the surface normal, with a kinetic energy resolution of 50 meV. The Au 4f7/2 core level was used as re ...
... XPS measurements were performed at the I311 beamline at the MAX-IV laboratory (Lund, Sweden). The spectra were obtained in normal incidence geometry, the photoelectron detection angle being 55° off the surface normal, with a kinetic energy resolution of 50 meV. The Au 4f7/2 core level was used as re ...
1.1.4 Amount of Substance / The Mole
... and found that the ratios of volumes of the reacting gases were small integer numbers e.g two volumes of hydrogen react with one of oxygen to produce two volumes of water ...
... and found that the ratios of volumes of the reacting gases were small integer numbers e.g two volumes of hydrogen react with one of oxygen to produce two volumes of water ...
INTRODUCTION TO CHEMISTRY
... __PHYSICAL__ properties can be observed without chemically changing matter. ___CHEMICAL_____ properties describe how a substance interacts with other substances. __SOLIDS___ have definite shapes and definite volumes. __LIQUIDS_____ have indefinite shapes and definite volumes. ___GASES/ PLASMA______ ...
... __PHYSICAL__ properties can be observed without chemically changing matter. ___CHEMICAL_____ properties describe how a substance interacts with other substances. __SOLIDS___ have definite shapes and definite volumes. __LIQUIDS_____ have indefinite shapes and definite volumes. ___GASES/ PLASMA______ ...
Chapter 2 What Is Matter
... If a mixture contains large particles dispersed in a liquid or a gas that will settle out, it is classified as a suspension. If the particles of a suspension are not stirred constantly, they will settle out. Particles of a suspension can also be filtered out. The particles are large enough to be ca ...
... If a mixture contains large particles dispersed in a liquid or a gas that will settle out, it is classified as a suspension. If the particles of a suspension are not stirred constantly, they will settle out. Particles of a suspension can also be filtered out. The particles are large enough to be ca ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.