of the atom.
... Moles of atoms in molecules • How many moles of oxygen atoms are in 5 mol of O2 ? 1 mol of O2 contains 2 mol of O atoms 5 mol of O2 contains 10 mol of O atoms • How many moles of oxygen atoms are in 5 mol of H2SO4? 1 mol of H2SO4 contains 4 mol of O atoms 5 mol of H2SO4 contains 20 mol of O atoms ...
... Moles of atoms in molecules • How many moles of oxygen atoms are in 5 mol of O2 ? 1 mol of O2 contains 2 mol of O atoms 5 mol of O2 contains 10 mol of O atoms • How many moles of oxygen atoms are in 5 mol of H2SO4? 1 mol of H2SO4 contains 4 mol of O atoms 5 mol of H2SO4 contains 20 mol of O atoms ...
Description of liquid–gas phase transition in the frame of continuum
... of interactions at a microlevel. At present time several approaches to mathematical implementation of this concept are developed. Each of them is used to solve certain problems. One of the conventional approaches is based on the assumption of a sharp interface separating different phases. The local ...
... of interactions at a microlevel. At present time several approaches to mathematical implementation of this concept are developed. Each of them is used to solve certain problems. One of the conventional approaches is based on the assumption of a sharp interface separating different phases. The local ...
Charge-density analysis of an iron–sulfur protein at an ultra
... correlation with atomic charges of S atoms. S4 with the largest atomic charge (−1.6) has no such interactions, while other bridging S atoms with smaller charges (−0.5 to −0.4) have (Fig. 2d). Furthermore, this rule is consistent for Cys-S atoms. Cys46-Sγ and Cys61-Sγ with smaller charges (−0.1 and − ...
... correlation with atomic charges of S atoms. S4 with the largest atomic charge (−1.6) has no such interactions, while other bridging S atoms with smaller charges (−0.5 to −0.4) have (Fig. 2d). Furthermore, this rule is consistent for Cys-S atoms. Cys46-Sγ and Cys61-Sγ with smaller charges (−0.1 and − ...
ANew Copper(II)ComplexwiththeN,N`-Bis(antipyryl-4
... with some oligoelements may explain their pharmaceutical activity. In this respect, complexes of some first row metal ions with ligands containing the antipyrine moiety N,N'-bis(4-antipyrylmethyl)piperazine (BAMP) have been studied [15 - 18], and some of them have been demonstrated to exhibit antitu ...
... with some oligoelements may explain their pharmaceutical activity. In this respect, complexes of some first row metal ions with ligands containing the antipyrine moiety N,N'-bis(4-antipyrylmethyl)piperazine (BAMP) have been studied [15 - 18], and some of them have been demonstrated to exhibit antitu ...
Slide 1
... • tend to be densely packed. • have several reasons for dense packing: -Typically, made of heavy element. -Metallic bonding is not directional; i.e., no restrictions as to the number and position of nearest-neighbor atoms -Nearest neighbor distances tend to be small in order to lower bond energy. ...
... • tend to be densely packed. • have several reasons for dense packing: -Typically, made of heavy element. -Metallic bonding is not directional; i.e., no restrictions as to the number and position of nearest-neighbor atoms -Nearest neighbor distances tend to be small in order to lower bond energy. ...
Crystal Structure of Mixed-metal Phosphite, Pb2Ga(HPIIIO3)3(PVO3)
... performed on a Rigaku Mercury CCD diffractometer with Mo-Kα radiation (λ=0.71073 Å) at 293(2) K. The data sets were corrected for Lorentz and polarization factors as well as absorption by the multi-scan method [15-17]. The structure was solved by the direct method and refined by full-matrix least-sq ...
... performed on a Rigaku Mercury CCD diffractometer with Mo-Kα radiation (λ=0.71073 Å) at 293(2) K. The data sets were corrected for Lorentz and polarization factors as well as absorption by the multi-scan method [15-17]. The structure was solved by the direct method and refined by full-matrix least-sq ...
powerpoint
... Image taken from http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/imgkin/vapp2.gif on 8/11/11. ...
... Image taken from http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/imgkin/vapp2.gif on 8/11/11. ...
Drawing Electron
... In carbon dioxide, CO2, octets are achieved by sharing two pairs of electrons between atoms; this is called a double bond. ...
... In carbon dioxide, CO2, octets are achieved by sharing two pairs of electrons between atoms; this is called a double bond. ...
PDF only - at www.arxiv.org.
... However, after 300ºC the reflections mainly signify the hematite phase of the nano particle. In this case, the formation of the maghemite like structure is mainly restricted by the kinetic issue during the growth of hematite nano particle by the sintering process. This means that not only particle ...
... However, after 300ºC the reflections mainly signify the hematite phase of the nano particle. In this case, the formation of the maghemite like structure is mainly restricted by the kinetic issue during the growth of hematite nano particle by the sintering process. This means that not only particle ...
Phase Transformations
... In contrast to metals silicates, borates and phosphates tend to form glasses Due to high cation-cation repulsion these materials have open structures In silicates the difference in total bond energy between periodic and aperiodic array is small (bond energy is primarily determined by the firs ...
... In contrast to metals silicates, borates and phosphates tend to form glasses Due to high cation-cation repulsion these materials have open structures In silicates the difference in total bond energy between periodic and aperiodic array is small (bond energy is primarily determined by the firs ...
Atom
... That force, when related to the cross-sectional area, gives the stress to separate atoms. Deeper energy trough occurs when the interatomic forces of attractions are higher. Since deeper energy trough also leads to higher melting points as explained above, it can be observed that materials with high ...
... That force, when related to the cross-sectional area, gives the stress to separate atoms. Deeper energy trough occurs when the interatomic forces of attractions are higher. Since deeper energy trough also leads to higher melting points as explained above, it can be observed that materials with high ...
Covalent Bonding 5 Practice Problems
... side by side overlap of orbitals sigma bond electrons lie on the axis between the 2 nuclei; pi bond electrons are in a plane above and below the nuclei sigma bonds are stronger than pi bonds ...
... side by side overlap of orbitals sigma bond electrons lie on the axis between the 2 nuclei; pi bond electrons are in a plane above and below the nuclei sigma bonds are stronger than pi bonds ...
Unit 7 Slides
... Determine the molar mass when given symbol or formula Define mole and Avogadro’s number Convert moles to mass and mass to moles Convert particles to moles and moles to particles Define molar volume and convert liters to moles & moles to liters Determine % composition when given formula or name for ...
... Determine the molar mass when given symbol or formula Define mole and Avogadro’s number Convert moles to mass and mass to moles Convert particles to moles and moles to particles Define molar volume and convert liters to moles & moles to liters Determine % composition when given formula or name for ...
The Mole - Firelands Local Schools
... Example 2: find the molar mass of C6H12O6 6 C: 6 x 12.0 g = 72.0 g 12 H: 12 x 1.01 g = 12.1 g 6 O: 6 x 16.0 g = 96.0 g 180.1 g = 1 mol iii. Example 3: find the molar mass of Na2SO4 2 Na: 2 x 23.0 g = 46.0 g 1 S: 1 x 32.1 g = 32.1 g 4 O: 4 x 16.0 g = 64.0 g 142.1 g = 1 mol Na2SO4 iv. Example 4: find ...
... Example 2: find the molar mass of C6H12O6 6 C: 6 x 12.0 g = 72.0 g 12 H: 12 x 1.01 g = 12.1 g 6 O: 6 x 16.0 g = 96.0 g 180.1 g = 1 mol iii. Example 3: find the molar mass of Na2SO4 2 Na: 2 x 23.0 g = 46.0 g 1 S: 1 x 32.1 g = 32.1 g 4 O: 4 x 16.0 g = 64.0 g 142.1 g = 1 mol Na2SO4 iv. Example 4: find ...
Specific heat, magnetic susceptibility, resistivity and thermal
... the MB12 family is found in ZrB12 with Tc = 6 K.3 This relatively high value compared to other dodecaborides has been considered as puzzling in view of the particularly low density of states at the Fermi level revealed by early measurements, such as the specific heat3 and the magnetic susceptibility ...
... the MB12 family is found in ZrB12 with Tc = 6 K.3 This relatively high value compared to other dodecaborides has been considered as puzzling in view of the particularly low density of states at the Fermi level revealed by early measurements, such as the specific heat3 and the magnetic susceptibility ...
Unit 13 Stoichiometry (Theoretical)
... 9. Methanol (CH4O) is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure, according to the following chemical equation: CO (g) + H2 (g) CH4O (g) a. Balance the chemical equation showing the synthesis of methanol. ...
... 9. Methanol (CH4O) is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure, according to the following chemical equation: CO (g) + H2 (g) CH4O (g) a. Balance the chemical equation showing the synthesis of methanol. ...
Topic 1222 Equation of State: Real Gases: van der Waals and Other
... Equation of State: Real Gases: van der Waals and Other Equations The properties of gases pose a formidable challenge for chemists who seek to understand their p-V-T properties. Chemists adopt an approach which starts by defining the properties of a (hypothetical) ideal gas (Topics 1220 and 2588). Th ...
... Equation of State: Real Gases: van der Waals and Other Equations The properties of gases pose a formidable challenge for chemists who seek to understand their p-V-T properties. Chemists adopt an approach which starts by defining the properties of a (hypothetical) ideal gas (Topics 1220 and 2588). Th ...
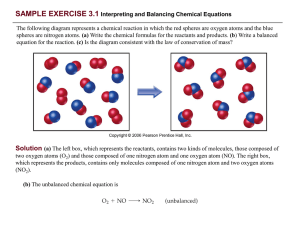
PRACTICE EXERCISE - Needham.K12.ma.us
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
Chapter 5: Chemical Formula Relationships – The Mole Counting by
... atoms. The mass difference is due to the nuclear binding energy required to overcome the repellent forces of the positive charges (proton) which are forced to come into close proximity in forming the nucleus. Where does this energy come from? Consider Einstein’s famous equation relating mass and ene ...
... atoms. The mass difference is due to the nuclear binding energy required to overcome the repellent forces of the positive charges (proton) which are forced to come into close proximity in forming the nucleus. Where does this energy come from? Consider Einstein’s famous equation relating mass and ene ...
Free energy and surface tension of arbitrarily large
... ~Ar,Kr,Xe! and molecules ~CO,CH4! have shown special stability at magic numbers 13, 55, 147, and 309 corresponding to Mackay icosahedral structures.1 Extensive progress has been made in determining the minimum potential energy configuration of such clusters2 and it has been shown3 that Mackay cluste ...
... ~Ar,Kr,Xe! and molecules ~CO,CH4! have shown special stability at magic numbers 13, 55, 147, and 309 corresponding to Mackay icosahedral structures.1 Extensive progress has been made in determining the minimum potential energy configuration of such clusters2 and it has been shown3 that Mackay cluste ...
Basics of material sciece - E
... the structure into repetitive small repeat entities called unit cells, i.e. in every crystal some fundamental grouping of particles is repeated. Obviously, a unit cell is the smallest component of the space lattice. The unit cell is the basic structural unit or building block of the crystal structur ...
... the structure into repetitive small repeat entities called unit cells, i.e. in every crystal some fundamental grouping of particles is repeated. Obviously, a unit cell is the smallest component of the space lattice. The unit cell is the basic structural unit or building block of the crystal structur ...
Exam2
... Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (molar mass =405.8 g/mole). a. 8.01 x 1023 atoms b. 2.67 x 1023 atoms c. 12.0 atoms d. 6.68 x 1022 atoms e. 1.07 x 1024 atoms Which of these is the correct name for Li2SO4? a. lithium sulfide b. dilithium sulfide c. dilithium sulfate d. lit ...
... Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (molar mass =405.8 g/mole). a. 8.01 x 1023 atoms b. 2.67 x 1023 atoms c. 12.0 atoms d. 6.68 x 1022 atoms e. 1.07 x 1024 atoms Which of these is the correct name for Li2SO4? a. lithium sulfide b. dilithium sulfide c. dilithium sulfate d. lit ...
On the relation between diffusion, sedimentation, and friction
... this result is indeed the same as Eq. (21). Note that the frictional coefficient, which can be determined by the ultracentrifuge measurements, is also with regard to the volume fixed frame of reference. Very recently, a paper by Schurr 19 appeared discussing some aspects of the same problem treated ...
... this result is indeed the same as Eq. (21). Note that the frictional coefficient, which can be determined by the ultracentrifuge measurements, is also with regard to the volume fixed frame of reference. Very recently, a paper by Schurr 19 appeared discussing some aspects of the same problem treated ...
Manufacturing Processes - Philadelphia University Jordan
... (2) Plastic deformation: that type of deformation where the atoms in the lattice have permanently moved from their previous locations, and a new equilibrium lattice has been formed (Fig. 2-16 c). If the force is removed, the lattice structure (and therefore the crystal) does not return to its origin ...
... (2) Plastic deformation: that type of deformation where the atoms in the lattice have permanently moved from their previous locations, and a new equilibrium lattice has been formed (Fig. 2-16 c). If the force is removed, the lattice structure (and therefore the crystal) does not return to its origin ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.