Unveiling the quantum critical point of an Ising chain
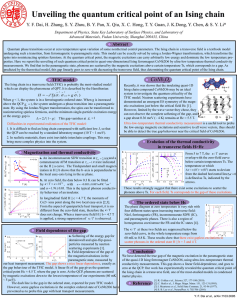
... Advanced Materials, Fudan University, Shanghai 200433, China ...
... Advanced Materials, Fudan University, Shanghai 200433, China ...
How to Study? • Reading ( ) • Ask questions (
... typically form ionic compounds that consist of cations and anions in a crystalline array. The ‘chemical bonds’ in an ionic compound are due to the Coulombic interactions (Section F.4) between all the ions in the crystal. The smallest unit of an ionic compound is called a formula unit. NaNO 3, consis ...
... typically form ionic compounds that consist of cations and anions in a crystalline array. The ‘chemical bonds’ in an ionic compound are due to the Coulombic interactions (Section F.4) between all the ions in the crystal. The smallest unit of an ionic compound is called a formula unit. NaNO 3, consis ...
The Liquid State
... interatomic potential, UCr). Contributions to this potential U(r) come from two-body, three-body) etc. potentials. From the pair correlation function one may obtain some information on the potential parameters i.e. the forces between atoms. The pair correlation function is also used to calculate the ...
... interatomic potential, UCr). Contributions to this potential U(r) come from two-body, three-body) etc. potentials. From the pair correlation function one may obtain some information on the potential parameters i.e. the forces between atoms. The pair correlation function is also used to calculate the ...
Study Guide for Composition of Matter Test - seys
... - homogenized milk Chemical Interactions 1.1 - Atoms are the smallest form of elements. Models of atoms: (Dalton) orbits & clouds: ...
... - homogenized milk Chemical Interactions 1.1 - Atoms are the smallest form of elements. Models of atoms: (Dalton) orbits & clouds: ...
Week 8 – Intermolecular Forces
... (B) H2O exhibits hydrogen bonding whereas H2S exhibits London (dispersion) forces (C) H2O exhibits hydrogen bonding whereas H2S exhibits dipole to dipole interactions (D) H2O is a liquid whereas H2S is a gas (E) H2S is more polarizable due to the greater amount of electrons 16. Of the following pure ...
... (B) H2O exhibits hydrogen bonding whereas H2S exhibits London (dispersion) forces (C) H2O exhibits hydrogen bonding whereas H2S exhibits dipole to dipole interactions (D) H2O is a liquid whereas H2S is a gas (E) H2S is more polarizable due to the greater amount of electrons 16. Of the following pure ...
Real Gases
... The dots calculated Vm from the NIST Chemistry Workbook. z - the two equations (VDW and RW) are not in quantitative agreement with accurate results the trends of dependence of z on P for different T values are correct. @ T = 200 K, z initially decreases with pressure. The compression factor only bec ...
... The dots calculated Vm from the NIST Chemistry Workbook. z - the two equations (VDW and RW) are not in quantitative agreement with accurate results the trends of dependence of z on P for different T values are correct. @ T = 200 K, z initially decreases with pressure. The compression factor only bec ...
Chemistry 534
... of HCl must react to produce 50.0 litres of hydrogen measured at 300.0K and at a pressure of 101 kPa? (4 marks) Equation : Zn(s) + 2 HCl(aq) ...
... of HCl must react to produce 50.0 litres of hydrogen measured at 300.0K and at a pressure of 101 kPa? (4 marks) Equation : Zn(s) + 2 HCl(aq) ...
Power Point Slides P..
... Atoms do not have the same coordination number Therefore are in higher energy state Surface energy, g [=] J/m2 Materials always try to reduce surface energy – tendency towards spherical shapes ...
... Atoms do not have the same coordination number Therefore are in higher energy state Surface energy, g [=] J/m2 Materials always try to reduce surface energy – tendency towards spherical shapes ...
June 2011 review
... 36. Energy is released during the fission of Pu-239 atoms as a result of the 1. formation of covalent bonds 3. conversion of matter to energy 2. formation of ionic bonds 4. conversion of energy to matter 37. A 1.00-mole sample of neon gas occupies a volume of 24.4 liters at 298 K and 101.3 kilopasca ...
... 36. Energy is released during the fission of Pu-239 atoms as a result of the 1. formation of covalent bonds 3. conversion of matter to energy 2. formation of ionic bonds 4. conversion of energy to matter 37. A 1.00-mole sample of neon gas occupies a volume of 24.4 liters at 298 K and 101.3 kilopasca ...
Chapter 8 An Introduction to Optical Atomic Spectroscopy
... sample into the atomizer with high efficiency and with no adverse interference effects. Samples are most commonly introduced in the form of solutions. Samples are introduced in the form of solids or finely dispersed powder if it is difficult to dissolve. ...
... sample into the atomizer with high efficiency and with no adverse interference effects. Samples are most commonly introduced in the form of solutions. Samples are introduced in the form of solids or finely dispersed powder if it is difficult to dissolve. ...
File - Mr Weng`s IB Chemistry
... • The mole is a fixed number of particles and refers to the amount, n, of substance. • Masses of atoms are compared on a scale relative to 12C and are expressed as relative atomic mass (Ar) and relative formula/molecular mass (Mr). • Molar mass (M) has the units g mol-1. • The empirical formula and ...
... • The mole is a fixed number of particles and refers to the amount, n, of substance. • Masses of atoms are compared on a scale relative to 12C and are expressed as relative atomic mass (Ar) and relative formula/molecular mass (Mr). • Molar mass (M) has the units g mol-1. • The empirical formula and ...
Chapter 3: Atoms: The Building Blocks of Matter
... Isotopes are atoms of the same element that have different masses. Isotopes will have the same number of protons and electrons, but will differ in the number of neutrons. Tin has the most isotopes (10) For example, Hydrogen has 3 isotopes: ...
... Isotopes are atoms of the same element that have different masses. Isotopes will have the same number of protons and electrons, but will differ in the number of neutrons. Tin has the most isotopes (10) For example, Hydrogen has 3 isotopes: ...
Chapter 3: Atoms: The Building Blocks of Matter
... Isotopes are atoms of the same element that have different masses. Isotopes will have the same number of protons and electrons, but will differ in the number of neutrons. Tin has the most isotopes (10) For example, Hydrogen has 3 isotopes: ...
... Isotopes are atoms of the same element that have different masses. Isotopes will have the same number of protons and electrons, but will differ in the number of neutrons. Tin has the most isotopes (10) For example, Hydrogen has 3 isotopes: ...
THE MOLE - Laureate International College
... How many molecules of CO2 are in 4.56 moles of CO2? 2.75 x 10 molecules How many moles of water is 5.87 x 1022 molecules? 0.0975 mol (or 9.75 x 10 ) How many moles is 7.7812.9xmoles ...
... How many molecules of CO2 are in 4.56 moles of CO2? 2.75 x 10 molecules How many moles of water is 5.87 x 1022 molecules? 0.0975 mol (or 9.75 x 10 ) How many moles is 7.7812.9xmoles ...
Document
... ii. Many of these symbols consist of the first letter or the first two letters of the element name. c. 3.3 Dalton’s Atomic Theory i. In the early 1800’s English scientist John Dalton came up with an explanation of how atoms combine to form compounds. ii. Dalton’s atomic theory has five main points: ...
... ii. Many of these symbols consist of the first letter or the first two letters of the element name. c. 3.3 Dalton’s Atomic Theory i. In the early 1800’s English scientist John Dalton came up with an explanation of how atoms combine to form compounds. ii. Dalton’s atomic theory has five main points: ...
Plasma and Flames - Coalition for Plasma Science
... Plasma and Flames – The Burning Question Do flames contain plasma? Phrased one way or another, this is a frequently asked question. The Short Answer: Some do; some don’t. The Medium Answer: Whether a plasma exists in a flame depends on the material being burned and the temperature. The temperature i ...
... Plasma and Flames – The Burning Question Do flames contain plasma? Phrased one way or another, this is a frequently asked question. The Short Answer: Some do; some don’t. The Medium Answer: Whether a plasma exists in a flame depends on the material being burned and the temperature. The temperature i ...
Ch. 6 packet - Mahtomedi High School
... all of the atoms in the compound. For example, the compound CO2 would have a molar mass of 44 g because carbon has a mass of 12 g and oxygen has a mass of 16 g. We need to add the mass of two oxygen atoms, since there are two oxygen atoms in the formula. Molar masses give us a conversion factor that ...
... all of the atoms in the compound. For example, the compound CO2 would have a molar mass of 44 g because carbon has a mass of 12 g and oxygen has a mass of 16 g. We need to add the mass of two oxygen atoms, since there are two oxygen atoms in the formula. Molar masses give us a conversion factor that ...
First-Sample_Exam-1-Key
... There are more things in Heaven and Earth, Horatio, than are dreamt of in your Philosophy. - Hamlet on quantum mechanics Instructions: ...
... There are more things in Heaven and Earth, Horatio, than are dreamt of in your Philosophy. - Hamlet on quantum mechanics Instructions: ...
Quantities, Units, Symbols and Nomenclature used in
... n, amount of substance, expressed in moles. It is incorrect to use the term ‘number of moles’. (See details under ‘Amount of Substance’ below.) c, amount concentration, is expressed as moles per litre, also denoted by the format [ ]. Concentrations may also be written as mass concentration, expresse ...
... n, amount of substance, expressed in moles. It is incorrect to use the term ‘number of moles’. (See details under ‘Amount of Substance’ below.) c, amount concentration, is expressed as moles per litre, also denoted by the format [ ]. Concentrations may also be written as mass concentration, expresse ...
Formula Mass (weight)
... – Balance elements that appear in only one compound first • Remember that compound stoichiometry (subscripts) can not change ...
... – Balance elements that appear in only one compound first • Remember that compound stoichiometry (subscripts) can not change ...
Elaine Teto
... relative amounts of each substance present. However, in using these coefficients we come to our next topic, balancing chemical reactions. One of the most important laws to remember, when considering balancing equations is the Law of Conservation of Mass, which states that matter can neither be creat ...
... relative amounts of each substance present. However, in using these coefficients we come to our next topic, balancing chemical reactions. One of the most important laws to remember, when considering balancing equations is the Law of Conservation of Mass, which states that matter can neither be creat ...
Bose–Einstein condensate

A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of bosons cooled to temperatures very close to absolute zero (that is, very near 5000000000000000000♠0 K or 5000000000000000000♠−273.15 °C). Under such conditions, a large fraction of bosons occupy the lowest quantum state, at which point macroscopic quantum phenomena become apparent.This state was first predicted, generally, in 1924–25 by Satyendra Nath Bose and Albert Einstein.























