CHM 111: General Physical Chemistry 3 Units
... Historical development of the atom: definition of atoms, Daltons atomic theory, relative atomic masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole ...
... Historical development of the atom: definition of atoms, Daltons atomic theory, relative atomic masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole ...
12.1 Avogadro`s Law and Molar Volume
... molecules as a container with 1.0 L of N2 at STP, even though the mass of nitrogen is 14 times greater. The pressure is directly related to the number of particles in each container of gas. So if each container had the same amount of pressure, there would be no difference in the number of particles. ...
... molecules as a container with 1.0 L of N2 at STP, even though the mass of nitrogen is 14 times greater. The pressure is directly related to the number of particles in each container of gas. So if each container had the same amount of pressure, there would be no difference in the number of particles. ...
Properties of Gases
... 1. A gas is composed of very minute particles called molecules. 2. The molecules are in a state of constant motion in random directions. During their movement they collide with each other and also with the walls of the container. 3. The molecules are perfectly elastic and the collisions do not resul ...
... 1. A gas is composed of very minute particles called molecules. 2. The molecules are in a state of constant motion in random directions. During their movement they collide with each other and also with the walls of the container. 3. The molecules are perfectly elastic and the collisions do not resul ...
Gas Laws - myersparkphysics
... Newton’s 3rd law tells us that this force (of wall-on-particle) is equal and opposite to the force of the particle on the wall. Thus, F= + mv2/L . the total force exerted on the wall is equal to the number of particles times the average force for each particle. Since N particles move randomly in 3-D ...
... Newton’s 3rd law tells us that this force (of wall-on-particle) is equal and opposite to the force of the particle on the wall. Thus, F= + mv2/L . the total force exerted on the wall is equal to the number of particles times the average force for each particle. Since N particles move randomly in 3-D ...
Topic 3.3 Kinetic Model of Ideal Gas
... The increased temperature means the molecules are moving faster, and thus they hit the wall more often. The force from each molecule goes up on average and an increased of collisions in a given time means that the pressure increases. 4. In this final example, an isolated sample of gas is compressed. ...
... The increased temperature means the molecules are moving faster, and thus they hit the wall more often. The force from each molecule goes up on average and an increased of collisions in a given time means that the pressure increases. 4. In this final example, an isolated sample of gas is compressed. ...
Lecture 1
... Gas - a form of matter that fills any container it occupies Physical state - the state of physical properties of a sample such as volume, mole, pressure, and temperature Mole - the number of atoms or molecules equal to Avogradro’s number NA = 6.022137 × 1023, which is defined as the number of atoms ...
... Gas - a form of matter that fills any container it occupies Physical state - the state of physical properties of a sample such as volume, mole, pressure, and temperature Mole - the number of atoms or molecules equal to Avogradro’s number NA = 6.022137 × 1023, which is defined as the number of atoms ...
South Pasadena • Chemistry
... 1 mole each of He gas and CO2 gas are in a rigid container at the same temperature. The kinetic energy of He is ________________ (greater than, less than, the same as) the kinetic energy of CO2? The velocity of He is ________________ (greater than, less than, the same as) the velocity of CO2? Calcul ...
... 1 mole each of He gas and CO2 gas are in a rigid container at the same temperature. The kinetic energy of He is ________________ (greater than, less than, the same as) the kinetic energy of CO2? The velocity of He is ________________ (greater than, less than, the same as) the velocity of CO2? Calcul ...
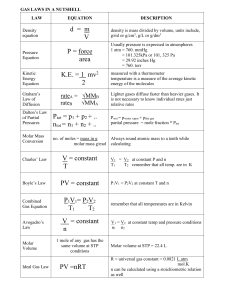
Sec. 12.3: Molecular Composition of Gases 1) Boyle`s Law: a
... a) Charles’ Law: b) Gay-Lussac’s Law: c) Avogadro’s Law: 2) Ideal gas: an _________________ gas whose particles are infinitely ____________ and do ____________ interact with each other. a) An ideal gas: i) Does _____ condense to a _____________ at ___________ temperatures. ii) Does not have forces o ...
... a) Charles’ Law: b) Gay-Lussac’s Law: c) Avogadro’s Law: 2) Ideal gas: an _________________ gas whose particles are infinitely ____________ and do ____________ interact with each other. a) An ideal gas: i) Does _____ condense to a _____________ at ___________ temperatures. ii) Does not have forces o ...
Sec. 12.3: Molecular Composition of Gases 1) Boyle`s Law: a
... a) Charles’ Law: b) Gay-Lussac’s Law: c) Avogadro’s Law: 2) Ideal gas: an _________________ gas whose particles are infinitely ____________ and do ____________ interact with each other. a) An ideal gas: i) Does _____ condense to a _____________ at ___________ temperatures. ii) Does not have forces o ...
... a) Charles’ Law: b) Gay-Lussac’s Law: c) Avogadro’s Law: 2) Ideal gas: an _________________ gas whose particles are infinitely ____________ and do ____________ interact with each other. a) An ideal gas: i) Does _____ condense to a _____________ at ___________ temperatures. ii) Does not have forces o ...
Gas Sampler
... P1V1 ‗ P2V2 T1 T2 P = pressure, and calculations can be done when P is in millimeters of Hg (mm), kilopascals (kPa), or atmospheres (atm) as long as both pressures are in the same units. Standard pressure is 760 mm, 101.32 kPa, or 1 atm. T = temperature, and calculations can be done only when T is i ...
... P1V1 ‗ P2V2 T1 T2 P = pressure, and calculations can be done when P is in millimeters of Hg (mm), kilopascals (kPa), or atmospheres (atm) as long as both pressures are in the same units. Standard pressure is 760 mm, 101.32 kPa, or 1 atm. T = temperature, and calculations can be done only when T is i ...
Ideal gas
... set of randomly-moving, noninteracting point particles Properties: •randomly-moving •non-interacting •point particles •may be mono- di- atomic etc. Obeys: •the ideal gas laws, •a simplified equation of state, •is amenable to analysis under statistical mechanics ...
... set of randomly-moving, noninteracting point particles Properties: •randomly-moving •non-interacting •point particles •may be mono- di- atomic etc. Obeys: •the ideal gas laws, •a simplified equation of state, •is amenable to analysis under statistical mechanics ...
Ch 10 equations - mvhs
... Gases Equations– AP Chemistry: Remember for STP problems, use 1mol/22.4 L and Non-STP problems, use ideal gas law PV=nRT, S.T.P.= 0°C and 1 atm, R.T.P.= 25° C and 1 atm Equation ...
... Gases Equations– AP Chemistry: Remember for STP problems, use 1mol/22.4 L and Non-STP problems, use ideal gas law PV=nRT, S.T.P.= 0°C and 1 atm, R.T.P.= 25° C and 1 atm Equation ...
Chemistry Chapter 11
... Ideal Gas Law • A mathematical relationship among pressure, volume, temperature and number of moles • To derive (see p. 341) • Ideal gas law: • V= nRT/P OR PV = nRT • V is volume, P is pressure, T is temperature, n is number of moles, and R is a constant ...
... Ideal Gas Law • A mathematical relationship among pressure, volume, temperature and number of moles • To derive (see p. 341) • Ideal gas law: • V= nRT/P OR PV = nRT • V is volume, P is pressure, T is temperature, n is number of moles, and R is a constant ...
THE GASEOUS STATE
... gas molecules at room temp. and pressure. • However, they are important in liquids and solids. ...
... gas molecules at room temp. and pressure. • However, they are important in liquids and solids. ...
lecture14 - Chemistry at Winthrop University
... An ideal gas is an idealized model for real gases that have sufficiently low densities. The condition of low density means that the molecules of the gas are so far apart that they do not interact (except during collisions that are effectively elastic). The ideal gas law expresses the relationship be ...
... An ideal gas is an idealized model for real gases that have sufficiently low densities. The condition of low density means that the molecules of the gas are so far apart that they do not interact (except during collisions that are effectively elastic). The ideal gas law expresses the relationship be ...
Gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma). A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or compound molecules made from a variety of atoms (e.g. carbon dioxide). A gas mixture would contain a variety of pure gases much like the air. What distinguishes a gas from liquids and solids is the vast separation of the individual gas particles. This separation usually makes a colorless gas invisible to the human observer. The interaction of gas particles in the presence of electric and gravitational fields are considered negligible as indicated by the constant velocity vectors in the image. One type of commonly known gas is steam.The gaseous state of matter is found between the liquid and plasma states, the latter of which provides the upper temperature boundary for gases. Bounding the lower end of the temperature scale lie degenerative quantum gases which are gaining increasing attention. High-density atomic gases super cooled to incredibly low temperatures are classified by their statistical behavior as either a Bose gas or a Fermi gas. For a comprehensive listing of these exotic states of matter see list of states of matter.