CALCULUS
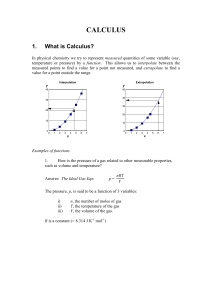
... e) From (c) above, if rate is R and temperature is T, then the shorthand for (c) is to write R(T). f) If energy is E and frequency is , then the shorthand for (d) is E(). This means that if we know the value of the symbol in brackets, we can calculate the value of the symbol outside the brackets. ...
... e) From (c) above, if rate is R and temperature is T, then the shorthand for (c) is to write R(T). f) If energy is E and frequency is , then the shorthand for (d) is E(). This means that if we know the value of the symbol in brackets, we can calculate the value of the symbol outside the brackets. ...
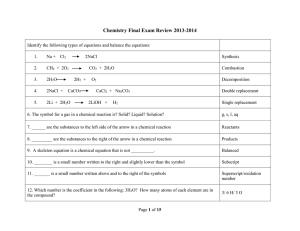
Type Of Chemical Reaction
... 32. According to the ______________ theory of ______________, the particles in a gas move rapidly, the particles in a gas are relatively far apart, the particles in a gas move independently of each other. ...
... 32. According to the ______________ theory of ______________, the particles in a gas move rapidly, the particles in a gas are relatively far apart, the particles in a gas move independently of each other. ...
Chapter 5 Gases Gases Pushing Measuring Air Pressure
... ¾But don’t be fooled into thinking all the gas particles are moving at the same speed!! ...
... ¾But don’t be fooled into thinking all the gas particles are moving at the same speed!! ...
semester two review sheet
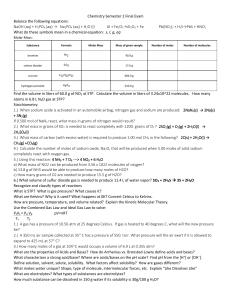
... 2. Calculate the temperature of a gas if 6.5 moles of it occupies a volume of 1.5 L at 2.4 atm 3. There is a mixture of three gases in a closed container. Gas A exerts a pressure of 10 psi, Gas B a pressure of 5.5 psi, and the total pressure is 34.7 psi. What is the partial pressure of Gas C? 4. A g ...
... 2. Calculate the temperature of a gas if 6.5 moles of it occupies a volume of 1.5 L at 2.4 atm 3. There is a mixture of three gases in a closed container. Gas A exerts a pressure of 10 psi, Gas B a pressure of 5.5 psi, and the total pressure is 34.7 psi. What is the partial pressure of Gas C? 4. A g ...
GAS PRACTICE A sample of an ideal gas is cooled from 50.0 °C to
... (D) The average kinetic energy of the hydrogen molecules is the same as the average kinetic energy of the oxygen molecules. (E) The average speed of the hydrogen molecules is the same as the average speed of the oxygen molecules. 14. At 25 °C, a sample of NH3 (molar mass 17 grams) effuses at the rat ...
... (D) The average kinetic energy of the hydrogen molecules is the same as the average kinetic energy of the oxygen molecules. (E) The average speed of the hydrogen molecules is the same as the average speed of the oxygen molecules. 14. At 25 °C, a sample of NH3 (molar mass 17 grams) effuses at the rat ...
PHYSICAL CHEMISTRY ERT 108 Semester II 2010
... Aims- find the equilibrium composition of an ideal-gas reaction mixture and - Equilibrium composition is related to the initial composition by a single variable, the equilibrium extent of reaction ...
... Aims- find the equilibrium composition of an ideal-gas reaction mixture and - Equilibrium composition is related to the initial composition by a single variable, the equilibrium extent of reaction ...
Chapter 5
... • The rates of both of both processes are inversely proportional to the molar mass of the gas • Gaseous diffusion used to separate 235U from 238U in World War II CHEM 1310 A/B Fall 2006 ...
... • The rates of both of both processes are inversely proportional to the molar mass of the gas • Gaseous diffusion used to separate 235U from 238U in World War II CHEM 1310 A/B Fall 2006 ...
111 Exam II Outline
... A. Gases are composed of such extremely tiny atoms or molecules that are widely separated by empty space. B. Gas particles move in a random,rapid, and continuous motion, thus has kinetic energy. C. Gas particles moves so rapidly and are so far apart the there is essentially no force of attraction be ...
... A. Gases are composed of such extremely tiny atoms or molecules that are widely separated by empty space. B. Gas particles move in a random,rapid, and continuous motion, thus has kinetic energy. C. Gas particles moves so rapidly and are so far apart the there is essentially no force of attraction be ...
******************Q***********Q*******Q****** Q***Q***Q***Q***Q***Q
... be considered as the starting point of statistical mechanics, the first time a macroscopic, thermodynamic concept such as temperature was quantitatively related to the microscopic dynamics. Boltzmann's later work really laid down the foundations for this discipline, with the first microscopic analys ...
... be considered as the starting point of statistical mechanics, the first time a macroscopic, thermodynamic concept such as temperature was quantitatively related to the microscopic dynamics. Boltzmann's later work really laid down the foundations for this discipline, with the first microscopic analys ...
Lab Stuff:
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
Lab # 28: Calculating the Value of the Ideal Gas Constant “R”
... this lab, you will use the ideal gas law to calculate the value of the ideal gas constant “R”. We will collect a sample of gas from a lighter by water displacement. See Figure 1 for the set up. Here are a few “nuts and bolts” of the lab to keep in mind: - The volume of gas, V, is simply the volume o ...
... this lab, you will use the ideal gas law to calculate the value of the ideal gas constant “R”. We will collect a sample of gas from a lighter by water displacement. See Figure 1 for the set up. Here are a few “nuts and bolts” of the lab to keep in mind: - The volume of gas, V, is simply the volume o ...
Name__________________________ Honors Chemistry Final
... 6. At constant pressure, a balloon containing 0.5 L of an unknown gas at 28 oC is placed in a refrigerator and allowed to cool to 4 oC. What is its new volume? ...
... 6. At constant pressure, a balloon containing 0.5 L of an unknown gas at 28 oC is placed in a refrigerator and allowed to cool to 4 oC. What is its new volume? ...
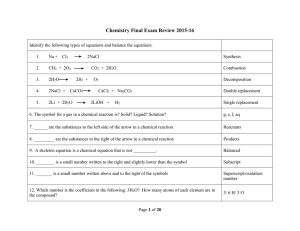
Type Of Chemical Reaction
... 32. According to the ______________ theory of ______________, the particles in a gas move rapidly, the particles in a gas are relatively far apart, the particles in a gas move independently of each other. ...
... 32. According to the ______________ theory of ______________, the particles in a gas move rapidly, the particles in a gas are relatively far apart, the particles in a gas move independently of each other. ...
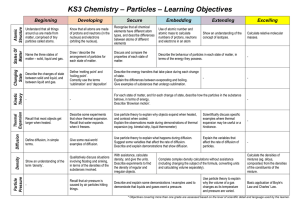
Particles - Townley Grammar School
... Explain the differences between evaporating and boiling. Give examples of substances that undergo sublimation. ...
... Explain the differences between evaporating and boiling. Give examples of substances that undergo sublimation. ...
CST Review Part 2
... and molecules and explains the properties of gases. As a basis for understanding this concept: a. Students know the random motion of molecules and their collisions with a surface create the observable pressure on that surface. b. Students know the random motion of molecules explains the diffusion of ...
... and molecules and explains the properties of gases. As a basis for understanding this concept: a. Students know the random motion of molecules and their collisions with a surface create the observable pressure on that surface. b. Students know the random motion of molecules explains the diffusion of ...
1) abcde 2) abcde 3) abcde 4) abcde 5) abcde 6) abcde 7) abcde 8
... 36) Write a balanced equation for the following. When calcium oxide is added to water, calcium hydroxide is formed. The temperature of the water increases during the reaction. [2 marks] 37) Classify each of the following as combination, decomposition, single displacement or double displacement react ...
... 36) Write a balanced equation for the following. When calcium oxide is added to water, calcium hydroxide is formed. The temperature of the water increases during the reaction. [2 marks] 37) Classify each of the following as combination, decomposition, single displacement or double displacement react ...
CHEMISTRY 102A/102C Spring 2014 Hour Exam II Page _____ For
... numbers of molecules. II. On average, a N2 molecule will possess the same kinetic energy as a CO2 molecule at the same temperature. III. On average, an H2 molecule has a faster average velocity than a N2 molecule at the ...
... numbers of molecules. II. On average, a N2 molecule will possess the same kinetic energy as a CO2 molecule at the same temperature. III. On average, an H2 molecule has a faster average velocity than a N2 molecule at the ...
Chemistry Midterm Review 2006
... 1. Define the 6 changes of state. Which ones are exothermic and which ones are endothermic? 2. Create a chart comparing& contrasting characteristics of solids, liquids, and gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. E ...
... 1. Define the 6 changes of state. Which ones are exothermic and which ones are endothermic? 2. Create a chart comparing& contrasting characteristics of solids, liquids, and gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. E ...
Chapter 10 Notes
... mixture with an oxygen partial pressure of 28 atm and a nitrogen partial pressure of 110 atm is used. What is the total pressure in the tank? ...
... mixture with an oxygen partial pressure of 28 atm and a nitrogen partial pressure of 110 atm is used. What is the total pressure in the tank? ...
practice quiz5
... E) None of the above Question 9 A mixture of neon, argon, and xenon had a total pressure of 1560 mm Hg at 298 K. The mixture was found to contain 1.50 mol Ne, 2.65 mol Ar, and 1.75 mol Xe. What is the partial pressure of Xe? A) 701 mm Hg B) 658 mm Hg C) 396 mm Hg D) 463 mm Hg E) None of the above Qu ...
... E) None of the above Question 9 A mixture of neon, argon, and xenon had a total pressure of 1560 mm Hg at 298 K. The mixture was found to contain 1.50 mol Ne, 2.65 mol Ar, and 1.75 mol Xe. What is the partial pressure of Xe? A) 701 mm Hg B) 658 mm Hg C) 396 mm Hg D) 463 mm Hg E) None of the above Qu ...
Gas
Gas is one of the four fundamental states of matter (the others being solid, liquid, and plasma). A pure gas may be made up of individual atoms (e.g. a noble gas like neon), elemental molecules made from one type of atom (e.g. oxygen), or compound molecules made from a variety of atoms (e.g. carbon dioxide). A gas mixture would contain a variety of pure gases much like the air. What distinguishes a gas from liquids and solids is the vast separation of the individual gas particles. This separation usually makes a colorless gas invisible to the human observer. The interaction of gas particles in the presence of electric and gravitational fields are considered negligible as indicated by the constant velocity vectors in the image. One type of commonly known gas is steam.The gaseous state of matter is found between the liquid and plasma states, the latter of which provides the upper temperature boundary for gases. Bounding the lower end of the temperature scale lie degenerative quantum gases which are gaining increasing attention. High-density atomic gases super cooled to incredibly low temperatures are classified by their statistical behavior as either a Bose gas or a Fermi gas. For a comprehensive listing of these exotic states of matter see list of states of matter.