CHEM 2: Exam 3
... 16. A cylinder contains equal amounts of the 4 gases below. If the temperature of the cylinder was gradually decreased, which gas would be the first to condense to form a liquid? A. CH4 B. CH3CH3 C. NH3 D. B2H6 E. None of the above: they will all stay as gases even as the temperature decreases. 17. ...
... 16. A cylinder contains equal amounts of the 4 gases below. If the temperature of the cylinder was gradually decreased, which gas would be the first to condense to form a liquid? A. CH4 B. CH3CH3 C. NH3 D. B2H6 E. None of the above: they will all stay as gases even as the temperature decreases. 17. ...
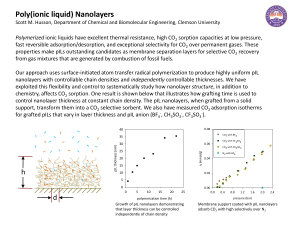
Molecular Level Models for CO2 Sorption in
... Adsorption of carbon dioxide in slit-shaped carbon micropores at 273 K has been studied by means of the grand canonical Monte Carlo (GCMC) simulations and the nonlocal density functional theory (NLDFT). Three molecular models of CO2 have been used. Long-run GCMC simulations were performed with the t ...
... Adsorption of carbon dioxide in slit-shaped carbon micropores at 273 K has been studied by means of the grand canonical Monte Carlo (GCMC) simulations and the nonlocal density functional theory (NLDFT). Three molecular models of CO2 have been used. Long-run GCMC simulations were performed with the t ...
PPT
... suspended somewhere in the atmosphere. The reason this column of air does not "fall" down under the pull of gravity is because the pressure acting on its bottom surface is higher than the pressure acting on its top surface. The pressure difference Δp exactly balances the weight (per unit area) of th ...
... suspended somewhere in the atmosphere. The reason this column of air does not "fall" down under the pull of gravity is because the pressure acting on its bottom surface is higher than the pressure acting on its top surface. The pressure difference Δp exactly balances the weight (per unit area) of th ...
Heterogeneous catalysis (I)
... gases). It is characterized by the presence of “active sites” on the catalyst surface. In a homogeneous reaction, the catalyst is in the same phase as the reactants (as the hydrolysis of esters by acid catalysts, all reactants and catalyst are dissolved in water: H+ CH3CO2CH3(aq) + H2O(l) CH3CO2H(aq ...
... gases). It is characterized by the presence of “active sites” on the catalyst surface. In a homogeneous reaction, the catalyst is in the same phase as the reactants (as the hydrolysis of esters by acid catalysts, all reactants and catalyst are dissolved in water: H+ CH3CO2CH3(aq) + H2O(l) CH3CO2H(aq ...
Annexure `CD-01` L T P/S SW/FW TOTAL CREDIT UNITS 3 1 2 0 5
... Zeta potential, Electrophoresis, origin of charge on colloidal particles, protective colloids, gold number, applications. Module III Adsorption Heat of adsorption, Freundlich adsorption isotherm, physisorption and chemisorption, Langmuir’s theory, B.E.T. Theory, applications Module IV Photoche ...
... Zeta potential, Electrophoresis, origin of charge on colloidal particles, protective colloids, gold number, applications. Module III Adsorption Heat of adsorption, Freundlich adsorption isotherm, physisorption and chemisorption, Langmuir’s theory, B.E.T. Theory, applications Module IV Photoche ...
IB 1 CHEMISTRY
... a compound in oxygen so that all its elements forms oxides. The amount of oxides can be determined and that gives the original amount of each element. The empirical formula shows the simplest whole number ratio of atoms of each element in a particle of that substance, ...
... a compound in oxygen so that all its elements forms oxides. The amount of oxides can be determined and that gives the original amount of each element. The empirical formula shows the simplest whole number ratio of atoms of each element in a particle of that substance, ...
Modelling Mass Transfer in Nitrification Processes Using
... strongly related to the lattice structure. • Most selective oxidation reactions kinetics can be described in terms of the “REDOX” mechanism. • Until recently, few studies focussed on the catalyst surface dynamics in relation to the “REDOX” properties. The secret is ...
... strongly related to the lattice structure. • Most selective oxidation reactions kinetics can be described in terms of the “REDOX” mechanism. • Until recently, few studies focussed on the catalyst surface dynamics in relation to the “REDOX” properties. The secret is ...
KLH2002 - the Research Group of Angelika Kühnle
... recognition and interaction: the chemical and biological properties of molecules depend not only on the nature of their constituent atoms but also on how these atoms are positioned in space. Chiral speci®city is consequently fundamental in chemical biology and pharmacology1,2 and has accordingly bee ...
... recognition and interaction: the chemical and biological properties of molecules depend not only on the nature of their constituent atoms but also on how these atoms are positioned in space. Chiral speci®city is consequently fundamental in chemical biology and pharmacology1,2 and has accordingly bee ...
Modification of the surface electronic and chemical properties of Pt
... slabs, where the second layer of metal atoms has been replaced by one of the 3d transition metals. Some explanation of why this is a reasonable and relevant model is needed. Pt segregates to the surface in many Pt-bimetallic mixtures, and a Pt-rich surface is expected in all of the cases in the pres ...
... slabs, where the second layer of metal atoms has been replaced by one of the 3d transition metals. Some explanation of why this is a reasonable and relevant model is needed. Pt segregates to the surface in many Pt-bimetallic mixtures, and a Pt-rich surface is expected in all of the cases in the pres ...
The nature of matter
... Gay-Lussac and law of combining volumes When gases react at constant temperature and pressure, they combine in volumes that are related to each other as ratios of small whole numbers His experiments with hydrogen and oxygen had implications for the understanding of the atom and the structures o ...
... Gay-Lussac and law of combining volumes When gases react at constant temperature and pressure, they combine in volumes that are related to each other as ratios of small whole numbers His experiments with hydrogen and oxygen had implications for the understanding of the atom and the structures o ...
Downloaded on 2017-02
... activation energy for proton transfer from the incoming H2 O molecule to the carbon of the remaining ligands is calculated (see SI Fig. 2). For the first incoming H2 O molecule in the H2 O pulse, Ea = 0.52 eV is required for its dissociation as it adsorbs (Table 1 reaction 5). Transfer of the proton ...
... activation energy for proton transfer from the incoming H2 O molecule to the carbon of the remaining ligands is calculated (see SI Fig. 2). For the first incoming H2 O molecule in the H2 O pulse, Ea = 0.52 eV is required for its dissociation as it adsorbs (Table 1 reaction 5). Transfer of the proton ...
Surface electrochemistry
... kurių metu išsiskiria energija. Short course annotation in English (up to 500 characters) The course presents the electrochemical processes on the surfaces making emphasis on adsorption, corrosion and oxidation. The other key topics include the study of the relationship between chemical change and e ...
... kurių metu išsiskiria energija. Short course annotation in English (up to 500 characters) The course presents the electrochemical processes on the surfaces making emphasis on adsorption, corrosion and oxidation. The other key topics include the study of the relationship between chemical change and e ...
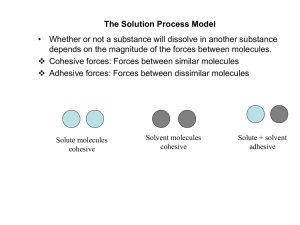
Adsorption
Adsorption is the adhesion of atoms, ions, or molecules from a gas, liquid, or dissolved solid to a surface. This process creates a film of the adsorbate on the surface of the adsorbent. This process differs from absorption, in which a fluid (the absorbate) permeates or is dissolved by a liquid or solid (the absorbent). Adsorption is a surface-based process while absorption involves the whole volume of the material. The term sorption encompasses both processes, while desorption is the reverse of it. Adsorption is a surface phenomenon.Similar to surface tension, adsorption is a consequence of surface energy. In a bulk material, all the bonding requirements (be they ionic, covalent, or metallic) of the constituent atoms of the material are filled by other atoms in the material. However, atoms on the surface of the adsorbent are not wholly surrounded by other adsorbent atoms and therefore can attract adsorbates. The exact nature of the bonding depends on the details of the species involved, but the adsorption process is generally classified as physisorption (characteristic of weak van der Waals forces) or chemisorption (characteristic of covalent bonding). It may also occur due to electrostatic attraction.Adsorption is present in many natural, physical, biological, and chemical systems, and is widely used in industrial applications such as activated charcoal, capturing and using waste heat to provide cold water for air conditioning and other process requirements (adsorption chillers), synthetic resins, increase storage capacity of carbide-derived carbons, and water purification. Adsorption, ion exchange, and chromatography are sorption processes in which certain adsorbates are selectively transferred from the fluid phase to the surface of insoluble, rigid particles suspended in a vessel or packed in a column. Pharmaceutical industry applications, which use adsorption as a means to prolong neurological exposure to specific drugs or parts thereof, are lesser known.However, it should be remarked that the distinction between adsorption and absorption vanishes as we go from perfectly crystalline macroscopic materials to porous/structured materials, aggregates and composites made out of increasingly smaller grains, viz., micron-sized particles to nanoparticles, sub-nano particles and finally molecules (or atoms). In such nano-composites, the internal surface area of particulate matter is very large. Then the adsorption on internal surfaces simply becomes absorption when viewed from the bulk. Then the distinction between adsorption and absorption vanishes. On the other hand, the distinction is clearest between bulk solids without internal structure, but having only surfaces where only adsorption can occur on the outer surfaces, and nanocomposites or aggregates with internal structure where absorption by the host material is simply adsorption on internal surfaces of the host material. As an example, we may consider a crystalline piece of silicon dioxide (quartz) which can adsorb water molecules on its surface. However, if the quartz is ground into very fine sand, the pile of sand (an aggregate) has a very large internal surface area. A very large amount of water can be adsorbed by the ""internal"" surfaces of the grains in the pile of sand, and this absorption is simply ""internal adsorption. If water is made to flow thorugh such a pile of sand, ions and toxins in the water may be preferentially adsorbed by the surfaces of the grains of sand, providing a simple, well-known water purification application.The word ""adsorption"" was coined in 1881 by German physicist Heinrich Kayser (1853-1940).