Chapter 2: Chemical Basis of Life
... of either the components or the processes of living things without using the biochemist's terms. For example, 96% of the human body is made up of just four major elements. Chemical reactions that hold atoms together do so by forming chemical bonds—these include ionic (electrovalent) bonds, covalent ...
... of either the components or the processes of living things without using the biochemist's terms. For example, 96% of the human body is made up of just four major elements. Chemical reactions that hold atoms together do so by forming chemical bonds—these include ionic (electrovalent) bonds, covalent ...
AP Chemistry Syllabus - Tuloso
... b. Dependence of kinetic energy of molecules on temperature c. Deviations from ideal gas laws B. Liquids and solids . Liquids and solids from the kinetic-molecular viewpoint A. Phase diagrams of one-component systems B. Changes of state, including critical points and triple points C. Structure of so ...
... b. Dependence of kinetic energy of molecules on temperature c. Deviations from ideal gas laws B. Liquids and solids . Liquids and solids from the kinetic-molecular viewpoint A. Phase diagrams of one-component systems B. Changes of state, including critical points and triple points C. Structure of so ...
Syllabus of the International Chemistry Olympiad
... unravelled, how aspects of green chemistry can be treated more quantitatively, how detergents aggregate to give micelles, how a hard coating can be made, and how fuel cells can produce electricity. In the practical problems microscale equipment will be used. The synthesis of some organic compounds, ...
... unravelled, how aspects of green chemistry can be treated more quantitatively, how detergents aggregate to give micelles, how a hard coating can be made, and how fuel cells can produce electricity. In the practical problems microscale equipment will be used. The synthesis of some organic compounds, ...
electrochemistry
... In a galvanic cell electrons flow from one electrode to the other because there is an electrical potential difference between the two half-cells, simply called the cell potential. In the zinc-copper cell, electrons flow from Zn|Zn2+ half-cell to Cu|Cu2+ half-cell. The Zn|Z2+ half-cell has a higher e ...
... In a galvanic cell electrons flow from one electrode to the other because there is an electrical potential difference between the two half-cells, simply called the cell potential. In the zinc-copper cell, electrons flow from Zn|Zn2+ half-cell to Cu|Cu2+ half-cell. The Zn|Z2+ half-cell has a higher e ...
Chapter 18: The Representative Elements The Representative
... number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
... number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
Chapter 18: The Representative Elements
... number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
... number). In its elemental state, all halogens atoms combine to form diatomic molecules (ex. F2,I2,…). With the exception of F, the halogens can also lose valence electrons and their oxidation states can range from -1 to +7. Chapter 18: The Representative Elements ...
Advanced Placement Chemistry: 1984 Free Response Questions
... (a) From the data above, calculate the standard heat of formation, Hf°, for butyric acid at 25 °C. (b) Write a correctly balanced equation for the formation of butyric acid from its elements. (c) Calculate the standard entropy change, Sf°, for the formation of butyric acid at 25 °C. The entropy ch ...
... (a) From the data above, calculate the standard heat of formation, Hf°, for butyric acid at 25 °C. (b) Write a correctly balanced equation for the formation of butyric acid from its elements. (c) Calculate the standard entropy change, Sf°, for the formation of butyric acid at 25 °C. The entropy ch ...
1999 U. S. NATIONAL CHEMISTRY OLYMPIAD
... Patricia A. Metz, University of Georgia, GA Ronald O. Ragsdale, University of Utah, UT Helen M. Stone (retired), Ben L. Smith High School, NC Diane D. Wolff, Ferrum College, VA DIRECTIONS TO THE EXAMINER–PART I Part I of this test is designed to be taken with a Scantron® answer sheet on which the st ...
... Patricia A. Metz, University of Georgia, GA Ronald O. Ragsdale, University of Utah, UT Helen M. Stone (retired), Ben L. Smith High School, NC Diane D. Wolff, Ferrum College, VA DIRECTIONS TO THE EXAMINER–PART I Part I of this test is designed to be taken with a Scantron® answer sheet on which the st ...
No Slide Title
... Zn is oxidized, lost electrons Zn is the reducing agent Cu2+ is reduced, gained electrons ...
... Zn is oxidized, lost electrons Zn is the reducing agent Cu2+ is reduced, gained electrons ...
The structure of Matter
... O The electron may be more strongly attracted to one of the atoms and spend more time revolving around that nucleus instead of the other. O This results in a positive and negative end for the compound (the end with the electron most of the time is negative…). O This is called a polar bond. ...
... O The electron may be more strongly attracted to one of the atoms and spend more time revolving around that nucleus instead of the other. O This results in a positive and negative end for the compound (the end with the electron most of the time is negative…). O This is called a polar bond. ...
Limiting Reactant WS with Answers
... some bromine is left over, along with a single compound that contains the two elements. The excess bromine is removed and allowed to react with excess sodium sulfite and excess sodium hydroxide, producing a mixture that contains 1.534 g of sodium sulfate. The balanced equation for this second reacti ...
... some bromine is left over, along with a single compound that contains the two elements. The excess bromine is removed and allowed to react with excess sodium sulfite and excess sodium hydroxide, producing a mixture that contains 1.534 g of sodium sulfate. The balanced equation for this second reacti ...
File first semester final study guide key
... 11. How many protons, neutrons and electrons does a neutral atom of an element with atomic number 50 and mass number 120 contain? Identify the element. Protons – 50; neutrons – 70; electrons - 50 Tin is the element 12. What is the family/group name for the elements in column 6A (16)? Oxygen family/C ...
... 11. How many protons, neutrons and electrons does a neutral atom of an element with atomic number 50 and mass number 120 contain? Identify the element. Protons – 50; neutrons – 70; electrons - 50 Tin is the element 12. What is the family/group name for the elements in column 6A (16)? Oxygen family/C ...
AQA Additional Sci C2 Revision Guide
... outer shell of its atoms. Elements in groups 1 and 2 of the periodic table only have 1 or 2 electrons in their outer shells so these form positive ions by losing their outer electrons. Elements in groups 6 and 7 of the periodic table only need 1 or 2 electrons to fill up their outer shells so these ...
... outer shell of its atoms. Elements in groups 1 and 2 of the periodic table only have 1 or 2 electrons in their outer shells so these form positive ions by losing their outer electrons. Elements in groups 6 and 7 of the periodic table only need 1 or 2 electrons to fill up their outer shells so these ...
Thermochemistry
... energy and heat associated with chemical reactions and/or physical transformations. Calorimetry is the science of measuring the heat of chemical reactions or physical changes. ...
... energy and heat associated with chemical reactions and/or physical transformations. Calorimetry is the science of measuring the heat of chemical reactions or physical changes. ...
Chemistry SOL Review Test
... (IONIC, VERY POLAR COVALENT, MODERATELY POLAR COVALENT, OR NONPOLAR COVALENT) will form between the atoms of the following elements. IONIC 97) Mg and F MODERATELY POLAR COVALENT 98) C & H POLAR COVALENT 99) C and O IONIC 100) Na & F MODERATELY POLAR COVALENT 101) H and Br ...
... (IONIC, VERY POLAR COVALENT, MODERATELY POLAR COVALENT, OR NONPOLAR COVALENT) will form between the atoms of the following elements. IONIC 97) Mg and F MODERATELY POLAR COVALENT 98) C & H POLAR COVALENT 99) C and O IONIC 100) Na & F MODERATELY POLAR COVALENT 101) H and Br ...
3UE-Exam Review-June2010 - Savita Pall and Chemistry
... 39. The increase in boiling points observed for F2 , Cl2 , Br2 , I2 is best attributed to... a) an increase in Van der Waals’ forces with increasing atomic number. b) a decrease in the electronegativity with increasing atomic number. c) an increase in the X - X bond energy with increasing atomic nu ...
... 39. The increase in boiling points observed for F2 , Cl2 , Br2 , I2 is best attributed to... a) an increase in Van der Waals’ forces with increasing atomic number. b) a decrease in the electronegativity with increasing atomic number. c) an increase in the X - X bond energy with increasing atomic nu ...
CHEMSTRY FREE-RESPONSE QUESTIONS (Form B)
... (d) Let the variables g, M, and V be defined as follows: g = the mass, in grams, of the sample of the iron(II) compound M= the molarity of the MnO4-(aq)) used as the titrant V = the volume, in liters, of MnO4-(aq)) added to reach the end point In terms of these variables, the number of moles of MnO4 ...
... (d) Let the variables g, M, and V be defined as follows: g = the mass, in grams, of the sample of the iron(II) compound M= the molarity of the MnO4-(aq)) used as the titrant V = the volume, in liters, of MnO4-(aq)) added to reach the end point In terms of these variables, the number of moles of MnO4 ...
Document
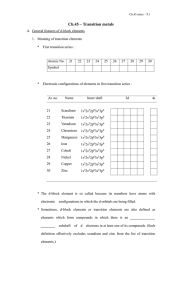
... first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons. * The higher oxidation states arise through ____________ bonding with more electronegative elements such as fluorine, chlorine and oxygen, as in TiCl4 , ...
... first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons. * The higher oxidation states arise through ____________ bonding with more electronegative elements such as fluorine, chlorine and oxygen, as in TiCl4 , ...
CHEMISTRY FALL FINAL PRACTICE 2016
... Write the nuclear notation for the most common isotope of chromium. ...
... Write the nuclear notation for the most common isotope of chromium. ...
Answers - Benjamin
... be measured, its concentration read off the graph and the percentage of manganese in the paper clip determined. Most steels contain less than 2% manganese and a typical value for the steel used in a paper clip is about 0.3%. Since the mass of manganese involved is very small, a more concentrated sol ...
... be measured, its concentration read off the graph and the percentage of manganese in the paper clip determined. Most steels contain less than 2% manganese and a typical value for the steel used in a paper clip is about 0.3%. Since the mass of manganese involved is very small, a more concentrated sol ...
MASS-INDEPENDENT ISOTOPE FRACTIONATION OF CHROMIUM
... Cr anomalies observed in primitive meteorites and planetary bodies [9,10] are now accepted as widespread nebula wide phenomena. There are some unresolved issues in Cr isotope cosmochemistry, however. For example 54Cr anomalies were observed by one laboratory [9] but not by the other [7] in the same ...
... Cr anomalies observed in primitive meteorites and planetary bodies [9,10] are now accepted as widespread nebula wide phenomena. There are some unresolved issues in Cr isotope cosmochemistry, however. For example 54Cr anomalies were observed by one laboratory [9] but not by the other [7] in the same ...
In organic chemistry, we studied a lot about the essential elements
... from inorganic substances (carbon dioxide and water) to organic substances like sugar carbohydrate and oxygen. How about the process of photosynthesize? Well, we all just know that they convert carbon dioxide into oxygen. But when I have been studying Biology about the “Pathway of Photosynthesis”, I ...
... from inorganic substances (carbon dioxide and water) to organic substances like sugar carbohydrate and oxygen. How about the process of photosynthesize? Well, we all just know that they convert carbon dioxide into oxygen. But when I have been studying Biology about the “Pathway of Photosynthesis”, I ...
PVS103 - unit 6 notes
... • Boron is unique in the group in that it is clearly a non-metal, we will concentrate on its properties, as it is very interesting. • The molecules boron forms are unique in that they do not conform fully to Lewis theory, for instance BH3 is a stable molecule, but there is no octet of electrons on b ...
... • Boron is unique in the group in that it is clearly a non-metal, we will concentrate on its properties, as it is very interesting. • The molecules boron forms are unique in that they do not conform fully to Lewis theory, for instance BH3 is a stable molecule, but there is no octet of electrons on b ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.