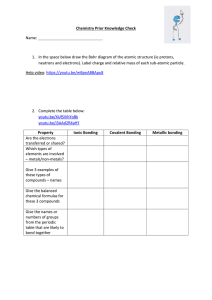
Ionic and Covalent Compounds: Naming, Formulas, Properties 1
... b) What is the mole ratio of oxygen gas to carbon dioxide gas? ___mol O 2 :___mol CO 2 [10 points] c) If one mole of oxygen gas reacts, how many moles of carbon dioxide gas are produced? [8 points] d) What are the molar masses of isopentane, C 5 H 12 , AND carbon dioxide, CO 2 ? [8 points] e) The vo ...
... b) What is the mole ratio of oxygen gas to carbon dioxide gas? ___mol O 2 :___mol CO 2 [10 points] c) If one mole of oxygen gas reacts, how many moles of carbon dioxide gas are produced? [8 points] d) What are the molar masses of isopentane, C 5 H 12 , AND carbon dioxide, CO 2 ? [8 points] e) The vo ...
Making Connections - SCH4U1-CCVI
... Hess’ Law and Heats of Formation A State Function: _____________________ Definition: When a reaction that can be expressed as the ____________ ____, , of two or more __________ reactions, the enthalpy of reaction, Hrxn, is the algebraic sum of the ___________________ rxn enthalpies, Hx. Standa ...
... Hess’ Law and Heats of Formation A State Function: _____________________ Definition: When a reaction that can be expressed as the ____________ ____, , of two or more __________ reactions, the enthalpy of reaction, Hrxn, is the algebraic sum of the ___________________ rxn enthalpies, Hx. Standa ...
Practice Test Packet
... 19. A weak acid, HF, is in solution with dissolved sodium fluoride, NaF. If HCl is added, which ion will react with the extra hydrogen ions from the HCl to keep the pH from changing? [A] F [B] Na+[C] Na [D] OH 20. Which of the following is true for a buffered solution? [A] The solution resists chang ...
... 19. A weak acid, HF, is in solution with dissolved sodium fluoride, NaF. If HCl is added, which ion will react with the extra hydrogen ions from the HCl to keep the pH from changing? [A] F [B] Na+[C] Na [D] OH 20. Which of the following is true for a buffered solution? [A] The solution resists chang ...
Final "I Can Statements" Answer Key
... heat – energy the moves between two substances due to differences in exothermic. temperature between the substances ...
... heat – energy the moves between two substances due to differences in exothermic. temperature between the substances ...
File - ARC: Chemistry
... ____ 17. An ionic bond is a bond between ____. a. valence electrons and cations c. the ions of two different nonmetals b. the ions of two different metals d. a cation and an anion ____ 18. Which of the compound is formed between the ions Potassium and Oxgen? a. Potassium Oxygen c. Potassium II Oxide ...
... ____ 17. An ionic bond is a bond between ____. a. valence electrons and cations c. the ions of two different nonmetals b. the ions of two different metals d. a cation and an anion ____ 18. Which of the compound is formed between the ions Potassium and Oxgen? a. Potassium Oxygen c. Potassium II Oxide ...
Atomic combinations: Electronegativity and ionic
... atom to the oxygen atom. Magnesium loses two electrons to form M g 2+ , and oxygen gains two electrons to form O2− . The attractive force between the oppositely charged ions is what holds the molecule together. The balanced equation for the reaction is: 2M g + O2 → 2M gO ...
... atom to the oxygen atom. Magnesium loses two electrons to form M g 2+ , and oxygen gains two electrons to form O2− . The attractive force between the oppositely charged ions is what holds the molecule together. The balanced equation for the reaction is: 2M g + O2 → 2M gO ...
summer learning G10
... top of the test-tube, it relit, proving that oxygen gas was also produced. A fine black solid, copper(II) oxide, was left in the test-tube. a. Assess which substances are reactants and which are products. ...
... top of the test-tube, it relit, proving that oxygen gas was also produced. A fine black solid, copper(II) oxide, was left in the test-tube. a. Assess which substances are reactants and which are products. ...
Equilibrium
... Next, add 8 drops of ferric nitrate to the second test tube. Record your observations. Based on the color observed, what appeared to happen to the amount of Fe(SCN)2+(aq)? Now add a few crystals of Na2HPO4 to the first test tube. Nake sure you shake for at least 20 seconds. Record your observations. ...
... Next, add 8 drops of ferric nitrate to the second test tube. Record your observations. Based on the color observed, what appeared to happen to the amount of Fe(SCN)2+(aq)? Now add a few crystals of Na2HPO4 to the first test tube. Nake sure you shake for at least 20 seconds. Record your observations. ...
Notebook - Science
... element: distinguished by its atomic number, the number of protons in the nuclei of its atoms Chemistry: Matter (4) anion: negatively charged ion cation: positively charged ion complex ion: an ion containing a central metal cation bonded to one or more molecules or ions physical property: can be mea ...
... element: distinguished by its atomic number, the number of protons in the nuclei of its atoms Chemistry: Matter (4) anion: negatively charged ion cation: positively charged ion complex ion: an ion containing a central metal cation bonded to one or more molecules or ions physical property: can be mea ...
Solution - gearju.com
... about 0.25 g of chloride, which is a little less than half of the mass of the original sample. Therefore, the calculated percent chloride of 47.51 percent is reasonable. ...
... about 0.25 g of chloride, which is a little less than half of the mass of the original sample. Therefore, the calculated percent chloride of 47.51 percent is reasonable. ...
Specific Reactions Quiz.wpd
... c) since energy is still tied up in carbon product bonds, energy is not released all at once d) the presence of random carbon products prevents balancing e) energy is lost because it remains trapped within any remnant fuel bonds ...
... c) since energy is still tied up in carbon product bonds, energy is not released all at once d) the presence of random carbon products prevents balancing e) energy is lost because it remains trapped within any remnant fuel bonds ...
Chapter 12 Review “Stoichiometry”
... of the following is true for: C5H12(l) + 8O2(g) → 5CO2(g) + 6H2O(l) a) 9 moles of reactants chemically change into 11 moles of product, or b) 9 atoms of reactants chemically change into 11 atoms of product? How many moles of aluminum react with 1.2 mol of FeO: 2Al(s) + 3FeO(s) → 3Fe(s) + Al2O3(s)? ...
... of the following is true for: C5H12(l) + 8O2(g) → 5CO2(g) + 6H2O(l) a) 9 moles of reactants chemically change into 11 moles of product, or b) 9 atoms of reactants chemically change into 11 atoms of product? How many moles of aluminum react with 1.2 mol of FeO: 2Al(s) + 3FeO(s) → 3Fe(s) + Al2O3(s)? ...
Page | 1 MATS1101 Chemistry notes semester 2 2012 TOPIC 1
... Using this theory we can explain three fundamental laws of chemical behaviour: 1. Law of Conservation of Mass and Energy: Matter is neither created or destroyed in a chemical reaction. Energy is neither created or destroyed in a chemical reaction, but it may be transformed from one form to another. ...
... Using this theory we can explain three fundamental laws of chemical behaviour: 1. Law of Conservation of Mass and Energy: Matter is neither created or destroyed in a chemical reaction. Energy is neither created or destroyed in a chemical reaction, but it may be transformed from one form to another. ...
Chemistry 2008 Multiple Choice
... volumetric flask, filled the flask to its calibration mark with the liquid, and then measured the mass of the flask and liquid. The recorded measurements are shown in the table. Mass (g) ...
... volumetric flask, filled the flask to its calibration mark with the liquid, and then measured the mass of the flask and liquid. The recorded measurements are shown in the table. Mass (g) ...
Chapter1 - WilsonChemWiki
... Ionic compounds: are formed between Metals and Non-metals when the valence electrons are transferred. (Metal) cations and (nonmetal) anions are held together by an electrostatic attraction between opposite charges in what is called an ionic bond. Example: Li+ is attracted to F- to form LiF compound. ...
... Ionic compounds: are formed between Metals and Non-metals when the valence electrons are transferred. (Metal) cations and (nonmetal) anions are held together by an electrostatic attraction between opposite charges in what is called an ionic bond. Example: Li+ is attracted to F- to form LiF compound. ...
organic chemistry - Peoria Public Schools
... The name of that branch (or side group) should indicate the number of carbon atoms in it and should end with –yl to indicate it is a branch e.g. methyl, ethyl, propyl. You need to identify the longest chain in the branched compound and any carbon atoms not in it must be part of a branch. The name of ...
... The name of that branch (or side group) should indicate the number of carbon atoms in it and should end with –yl to indicate it is a branch e.g. methyl, ethyl, propyl. You need to identify the longest chain in the branched compound and any carbon atoms not in it must be part of a branch. The name of ...
Electrons - biospaces
... • In a nonpolar covalent bond, the atoms share the electron equally • In a polar covalent bond, one atom is more electronegative, and the atoms do not share the electron equally • Unequal sharing of electrons causes a partial positive or negative charge for each atom or molecule ...
... • In a nonpolar covalent bond, the atoms share the electron equally • In a polar covalent bond, one atom is more electronegative, and the atoms do not share the electron equally • Unequal sharing of electrons causes a partial positive or negative charge for each atom or molecule ...
Chemistry Spring Final Review
... 21. In which compound is the percent composition by mass of chlorine equal to 47%? A. HClO B. HClO 2 C. HClO 3 D. HClO 4 22. What is the percent composition by mass of sulfur in the compound MgSO 4 ? A. 20% B. 27% C. 46% D. 53% 23. Which of the following best describes exothermic chemical reactions? ...
... 21. In which compound is the percent composition by mass of chlorine equal to 47%? A. HClO B. HClO 2 C. HClO 3 D. HClO 4 22. What is the percent composition by mass of sulfur in the compound MgSO 4 ? A. 20% B. 27% C. 46% D. 53% 23. Which of the following best describes exothermic chemical reactions? ...
Chapter 10 - Chemical Reactions
... However, you should be familiar with the rules which describe balanced chemical reactions. 1. Number of Atoms of each element conserved in reactants and products 2. Cannot change formula of reactants or products 3. Can only change coefficients to balance equation Hints to help in balancing equations ...
... However, you should be familiar with the rules which describe balanced chemical reactions. 1. Number of Atoms of each element conserved in reactants and products 2. Cannot change formula of reactants or products 3. Can only change coefficients to balance equation Hints to help in balancing equations ...
Chemistry Mid-Term Review Sheet
... 2. List and define the 5 areas of Chemistry. 3. What are the 5 steps of the scientific method (IN ORDER)? Chapter 2 4. Define the following: mass, volume, and matter. 5. Define pure substance. What are the two categories of pure substances? 6. What is a compound? List an example. 7. What is an eleme ...
... 2. List and define the 5 areas of Chemistry. 3. What are the 5 steps of the scientific method (IN ORDER)? Chapter 2 4. Define the following: mass, volume, and matter. 5. Define pure substance. What are the two categories of pure substances? 6. What is a compound? List an example. 7. What is an eleme ...
MYP 10 PeriodicityWS
... 5(a) Draw a diagram to show the structure of sodium chloride. Explain, in terms of bonding, why sodium chloride has a high melting point. (b) Lithium reacts with water. Write an equation for the reaction and state two observations that could be made during the reaction. [SL paper 2, Nov 05] 6 (a) Fo ...
... 5(a) Draw a diagram to show the structure of sodium chloride. Explain, in terms of bonding, why sodium chloride has a high melting point. (b) Lithium reacts with water. Write an equation for the reaction and state two observations that could be made during the reaction. [SL paper 2, Nov 05] 6 (a) Fo ...
File
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.