Atom - U of L Class Index
... identical in mass and in all other properties. 3. Different elements have different kinds of atoms; these atoms differ in mass from element to element. 4. Atoms are indestructible & retain their identity in all chemical reactions. 5. The formation of a compound from its elements occurs through the c ...
... identical in mass and in all other properties. 3. Different elements have different kinds of atoms; these atoms differ in mass from element to element. 4. Atoms are indestructible & retain their identity in all chemical reactions. 5. The formation of a compound from its elements occurs through the c ...
homework assignment 2 - the Petersen Home Page
... excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H2S. 2. A mixture of elemental carbon and sulfur has a mass of 9.00 g. The complete comb ...
... excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H2S. 2. A mixture of elemental carbon and sulfur has a mass of 9.00 g. The complete comb ...
Chemical and Physical Property Unit Test
... a2. What must often be added to increase the speed or ability of two substances to react? A. a bigger container B. adding heat C. more substances D. adding water a3. What kinds of energy are produced by bright fireworks? A. electricity, steam B. motion, gravity C. sound, magnetism D. light, heat b5. ...
... a2. What must often be added to increase the speed or ability of two substances to react? A. a bigger container B. adding heat C. more substances D. adding water a3. What kinds of energy are produced by bright fireworks? A. electricity, steam B. motion, gravity C. sound, magnetism D. light, heat b5. ...
Study Island Copyright © 2012 Study Island
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
Please do not remove this page. The periodic table, constants, and
... Which one of the following statements is not true? a. HF is a stronger acid than H2O because F is more electronegative than O. b. For 1.0 M solutions of any 2 weak bases, the solution of the base with the larger Kb will have the greater [OH–]. c. A Lewis acid is an electron pair acceptor. d. For a p ...
... Which one of the following statements is not true? a. HF is a stronger acid than H2O because F is more electronegative than O. b. For 1.0 M solutions of any 2 weak bases, the solution of the base with the larger Kb will have the greater [OH–]. c. A Lewis acid is an electron pair acceptor. d. For a p ...
Spring 2001 Key
... calcium oxide - incorrect Ca2+ O22– , the charges do not add up to neutral (CaO would be correct) potassium carbonate - incorrect K+ CO32– , the charges do not add up to neutral (K2CO3 would be correct) magnesium sulfate - correct Mg2+ SO42– This problem requires knowledge of names (inorganic nomenc ...
... calcium oxide - incorrect Ca2+ O22– , the charges do not add up to neutral (CaO would be correct) potassium carbonate - incorrect K+ CO32– , the charges do not add up to neutral (K2CO3 would be correct) magnesium sulfate - correct Mg2+ SO42– This problem requires knowledge of names (inorganic nomenc ...
Which notation represents an atom of sodium
... a) The equation represents a physical change, with the product and reactants having different chemical properties. b) The equation represents a physical change, with the product and reactants having identical chemical properties. c) The equation represents a chemical change, with the product and rea ...
... a) The equation represents a physical change, with the product and reactants having different chemical properties. b) The equation represents a physical change, with the product and reactants having identical chemical properties. c) The equation represents a chemical change, with the product and rea ...
Chapter 4 The Structure of Matter
... • a. Na+1Cl-1 or Ca+2F2-1 • b. When melted or dissolved in H2O ionic compounds will conduct electricity because the ions are free to move. • c. As solids the ions are locked so tightly that they do not conduct electricity. ...
... • a. Na+1Cl-1 or Ca+2F2-1 • b. When melted or dissolved in H2O ionic compounds will conduct electricity because the ions are free to move. • c. As solids the ions are locked so tightly that they do not conduct electricity. ...
SOLUBILITY RULES FOR IONIC COMPOUNDS IN WATER
... Wrap™ was reacted with excess hydrochloric acid, and 33.7 mL of hydrogen gas was collected at a temperature of 22ºC and a pressure of 726 torr. Determine the mass percent of aluminum in Reynolds ...
... Wrap™ was reacted with excess hydrochloric acid, and 33.7 mL of hydrogen gas was collected at a temperature of 22ºC and a pressure of 726 torr. Determine the mass percent of aluminum in Reynolds ...
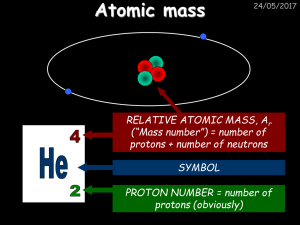
Practice problem chap3 1. The atomic mass of 35Cl (75.53%) and
... 1. The atomic mass of Cl (75.53%) and 37Cl (24.47%) are 34.968amu and 36.956amu.Calculate the average atomic mass in amu. 2. What is the mass percent (%) for O in SO2? (a) 38.09 (b) 45.41 (c) 50.00 (d) 53.86 (e) 56.43 3. How many molecules of ethane (C2H6) are present in 0.334g of C2H6?How many H at ...
... 1. The atomic mass of Cl (75.53%) and 37Cl (24.47%) are 34.968amu and 36.956amu.Calculate the average atomic mass in amu. 2. What is the mass percent (%) for O in SO2? (a) 38.09 (b) 45.41 (c) 50.00 (d) 53.86 (e) 56.43 3. How many molecules of ethane (C2H6) are present in 0.334g of C2H6?How many H at ...
PHYSICAL SETTING CHEMISTRY
... 51 What is the mass of KNO3(s) that must dissolve in 100. grams of water to form a saturated solution at 50.°C? [1] Base your answers to questions 52 through 55 on the information below. The reaction between aluminum and an aqueous solution of copper(II) sulfate is represented by the unbalanced equa ...
... 51 What is the mass of KNO3(s) that must dissolve in 100. grams of water to form a saturated solution at 50.°C? [1] Base your answers to questions 52 through 55 on the information below. The reaction between aluminum and an aqueous solution of copper(II) sulfate is represented by the unbalanced equa ...
chapter 6: chemical reactions: an introduction
... From the word description of the reaction, write formulas for all of the reactants and all of the products. Separate the reactants and products with an arrow, which means, "reacts to form". If you are having trouble writing the formulas from the word descriptions, you need to review the guidelines i ...
... From the word description of the reaction, write formulas for all of the reactants and all of the products. Separate the reactants and products with an arrow, which means, "reacts to form". If you are having trouble writing the formulas from the word descriptions, you need to review the guidelines i ...
Enthalpy and Internal Energy
... Example: Determining the Enthalpy of Reactions by Calorimetry 11) Consider the reaction of 10.08-g of aluminum hydroxide with 200.0 mL of sulfuric acid solution with a concentration high enough to react all of the base. If the reaction takes place in a coffee-cup calorimeter with a heat capacity o ...
... Example: Determining the Enthalpy of Reactions by Calorimetry 11) Consider the reaction of 10.08-g of aluminum hydroxide with 200.0 mL of sulfuric acid solution with a concentration high enough to react all of the base. If the reaction takes place in a coffee-cup calorimeter with a heat capacity o ...
1994–PTAS, Inc - mvhs
... Use appropriate ionic and molecular formulas to show the reactants and the products for the following, each of which results in a reaction occurring in aqueous solution except as indicated. Omit formulas for any ionic or molecular species that do not take part in the reaction. You need not balance. ...
... Use appropriate ionic and molecular formulas to show the reactants and the products for the following, each of which results in a reaction occurring in aqueous solution except as indicated. Omit formulas for any ionic or molecular species that do not take part in the reaction. You need not balance. ...
H3AsO4 + 3 I- + 2 H3O+ H3AsO3 + I3- + H2O
... and decreases with distance between ions. Electronegativity measures the ability of an atom to attract electrons in a covalent bond. Electronegativity generally increases from left to right in the periodic table and decreases down a column. The difference in atoms' electronegativities is used to det ...
... and decreases with distance between ions. Electronegativity measures the ability of an atom to attract electrons in a covalent bond. Electronegativity generally increases from left to right in the periodic table and decreases down a column. The difference in atoms' electronegativities is used to det ...
Chapter8 - Louisiana Tech University
... solid -> gas : Sublimation gas -> solid : Deposition Example: H2O(l) H2O(g) Solubility Supersaturated solution Chemical Equilibrium When a chemical reaction takes place in a container which prevents the entry or escape of any of the substances involved in the reaction, the quantities of these comp ...
... solid -> gas : Sublimation gas -> solid : Deposition Example: H2O(l) H2O(g) Solubility Supersaturated solution Chemical Equilibrium When a chemical reaction takes place in a container which prevents the entry or escape of any of the substances involved in the reaction, the quantities of these comp ...
1. Bromine exists naturally as a mixture of bromine
... Potassium forms an oxide containing 1 oxygen atom for every 2 atoms of potassium. What is the coefficient of oxygen in the balanced equation for the reaction of potassium with oxygen to form this ...
... Potassium forms an oxide containing 1 oxygen atom for every 2 atoms of potassium. What is the coefficient of oxygen in the balanced equation for the reaction of potassium with oxygen to form this ...
pdfCfE Higher - Unit 3 - Pupil Booklet 2 MB
... In a reversible reaction when the curve levels off this indicates that the reaction has reached equilibrium. (it does not indicate that the reaction has stopped). Shifting the Position of Equilibrium If, once an equilibrium has been established the reaction conditions are changed then the position o ...
... In a reversible reaction when the curve levels off this indicates that the reaction has reached equilibrium. (it does not indicate that the reaction has stopped). Shifting the Position of Equilibrium If, once an equilibrium has been established the reaction conditions are changed then the position o ...
Notes
... Reactants Products The arrow means “yields” or “produces” (s) = solid (g)= gas (l)= liquid (aq)= aqueous (dissolved in water) or = reversible reaction ...
... Reactants Products The arrow means “yields” or “produces” (s) = solid (g)= gas (l)= liquid (aq)= aqueous (dissolved in water) or = reversible reaction ...
AP Chemistry Review Assignment Brown and LeMay: Chemistry the
... and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether solids A and B and the gas C are elements or compounds? Explain your conclusions for each substance. 16. In the process of ...
... and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether solids A and B and the gas C are elements or compounds? Explain your conclusions for each substance. 16. In the process of ...
physical and chemical change
... A physical property is a property of a substance that can be observed without changing the substance into another substance. For example, the melting point of a solid is a physical property. Color, hardness, shape and texture are other physical properties. A chemical property is a property of a subs ...
... A physical property is a property of a substance that can be observed without changing the substance into another substance. For example, the melting point of a solid is a physical property. Color, hardness, shape and texture are other physical properties. A chemical property is a property of a subs ...
physical and chemical change
... A physical property is a property of a substance that can be observed without changing the substance into another substance. For example, the melting point of a solid is a physical property. Color, hardness, shape and texture are other physical properties. A chemical property is a property of a subs ...
... A physical property is a property of a substance that can be observed without changing the substance into another substance. For example, the melting point of a solid is a physical property. Color, hardness, shape and texture are other physical properties. A chemical property is a property of a subs ...
ASFG High School Summer Assignment Summer 2016
... lab reports will also be assigned and evaluated. Do not expect any grade curves or ‘fluff’ assignments this year! Assessments are administered and graded as if they are AP exams. Be aggressive in pursuit of knowledge not just the grades. Prioritize your learn ...
... lab reports will also be assigned and evaluated. Do not expect any grade curves or ‘fluff’ assignments this year! Assessments are administered and graded as if they are AP exams. Be aggressive in pursuit of knowledge not just the grades. Prioritize your learn ...
Equilibrium Constant
... product if only charged species are involved [IAP]. If the [AP] = Ksp, the system is in equilibrium,. If the [AP] < Ksp, the solution is undersaturated with respect to gypsum. If the [AP] > than the Ksp, the solution is supersaturated with respect to gypsum. ...
... product if only charged species are involved [IAP]. If the [AP] = Ksp, the system is in equilibrium,. If the [AP] < Ksp, the solution is undersaturated with respect to gypsum. If the [AP] > than the Ksp, the solution is supersaturated with respect to gypsum. ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.