Communicating Research to the General Public
... periodic table and can be considered the building blocks of inorganic chemistry just as carbon is considered the building block of organic chemistry. We are familiar with many of the transition metals because of their therapeutic value and presence in our bodies, such as iron, zinc, copper, and coba ...
... periodic table and can be considered the building blocks of inorganic chemistry just as carbon is considered the building block of organic chemistry. We are familiar with many of the transition metals because of their therapeutic value and presence in our bodies, such as iron, zinc, copper, and coba ...
Chapter 4 - WordPress.com
... correct side of the “reaction arrow” • Count atoms of each element on both sides of arrow • Start with the compound which has the most complex formula • Add coefficients to chemical formulas to balance numbers of each atom • Trial and error begins... May add subscripts of (g), (s), (l), (aq) or (ppt ...
... correct side of the “reaction arrow” • Count atoms of each element on both sides of arrow • Start with the compound which has the most complex formula • Add coefficients to chemical formulas to balance numbers of each atom • Trial and error begins... May add subscripts of (g), (s), (l), (aq) or (ppt ...
Thermo Practice Test
... Which one of the following statements best describes the relationship between G and temperature? A) G is independent of T; B) G varies with T; C) G is a linear function of T; D) G usually decreases with T. Hydrogen bromide gas and chlorine gas react to produce hydrogen chloride gas and liquid o ...
... Which one of the following statements best describes the relationship between G and temperature? A) G is independent of T; B) G varies with T; C) G is a linear function of T; D) G usually decreases with T. Hydrogen bromide gas and chlorine gas react to produce hydrogen chloride gas and liquid o ...
8F Compounds and Mixtures
... reaction, i.e. any process in which atoms become joined in different ways. The steps for writing a word equation are: ...
... reaction, i.e. any process in which atoms become joined in different ways. The steps for writing a word equation are: ...
CHEMICAL EQUATIONS - Clayton State University
... Consider the following Sodium (Na) has an atomic mass of 22.99 u This implies that the mass of 1 mole of Na = 22.99 g Molar mass of Na = 22.99 g/mol Formula mass of NaCl = 58.44 u The mass of 1 mole of NaCl = 58.44 g Molar mass of NaCl = 58.88 g/mol Formula mass of CaCO3 = 100.09 u The mass of 1 mol ...
... Consider the following Sodium (Na) has an atomic mass of 22.99 u This implies that the mass of 1 mole of Na = 22.99 g Molar mass of Na = 22.99 g/mol Formula mass of NaCl = 58.44 u The mass of 1 mole of NaCl = 58.44 g Molar mass of NaCl = 58.88 g/mol Formula mass of CaCO3 = 100.09 u The mass of 1 mol ...
Theoretical Enthalpy
... generated that lists the enthalpies of formation for any pure substance that allows us to calculate the expected enthalpy change when substances undergo chemical reactions. ...
... generated that lists the enthalpies of formation for any pure substance that allows us to calculate the expected enthalpy change when substances undergo chemical reactions. ...
Descriptive Chemistry of Elements d-Block
... 1.8 Oxidation Number (O.N.) The oxidation number of a metal centre is the charge left on the metal when all the ligands are removed. For example, removal of three Br ions and three neutral ammonia molecules from [CoBr3(NH3)3] leaves three positive charges on cobalt. Therefore, the oxidation number ...
... 1.8 Oxidation Number (O.N.) The oxidation number of a metal centre is the charge left on the metal when all the ligands are removed. For example, removal of three Br ions and three neutral ammonia molecules from [CoBr3(NH3)3] leaves three positive charges on cobalt. Therefore, the oxidation number ...
13.IVA group. Carbon and Silicon and their compounds.
... temperature, with relatively high melting and boiling points of approximately 1,400 and 2,800 degrees Celsius respectively. With a relatively high thermal conductivity of 149 W·m−1·K−1, silicon conducts heat well and as a result is not often used to insulate hot objects. In its crystalline form, pur ...
... temperature, with relatively high melting and boiling points of approximately 1,400 and 2,800 degrees Celsius respectively. With a relatively high thermal conductivity of 149 W·m−1·K−1, silicon conducts heat well and as a result is not often used to insulate hot objects. In its crystalline form, pur ...
Chapter 30 - The Chemical Basis of Animal Life
... given point at any particular moment in time; therefore, its location is given as an electron cloud. Because the number of negatively charged electrons outside the nucleus is equal to the number of positively charged protons, the atom is electrically uncharged or neutral. The number of protons and n ...
... given point at any particular moment in time; therefore, its location is given as an electron cloud. Because the number of negatively charged electrons outside the nucleus is equal to the number of positively charged protons, the atom is electrically uncharged or neutral. The number of protons and n ...
CfE Higher Chemistry Homework 3.5
... Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of iron(II) sulfide reacts with hydrochloric acid. Your diagram should be labelled and should show the names of any chemical used. ...
... Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of iron(II) sulfide reacts with hydrochloric acid. Your diagram should be labelled and should show the names of any chemical used. ...
Chapter 8 & 9 PowerPoint
... Steps to Drawing Lewis Structures 1. Count valence electrons. 2. Connect atoms together with bonds. In molecules with a single atom of one element and several atoms of another element, the single atom is generally in the center with the other atoms attached to it. 3. Add electrons around outside of ...
... Steps to Drawing Lewis Structures 1. Count valence electrons. 2. Connect atoms together with bonds. In molecules with a single atom of one element and several atoms of another element, the single atom is generally in the center with the other atoms attached to it. 3. Add electrons around outside of ...
File
... v) Alkaline Earth Metal: the Alkaline Earth Metals is the common name for the Group II elements. The Alkaline Earth Metals include beryllium, magnesium, calcium, barium and radium. w) Noble Gas: the Noble Gases is the common name for the Group VIII elements. The Noble gas elements have a full outer ...
... v) Alkaline Earth Metal: the Alkaline Earth Metals is the common name for the Group II elements. The Alkaline Earth Metals include beryllium, magnesium, calcium, barium and radium. w) Noble Gas: the Noble Gases is the common name for the Group VIII elements. The Noble gas elements have a full outer ...
8.3 Metals - UNSW Chemistry
... Emphasise that the least reactive metals were the easiest to isolate and were the first metals to be widely used, and that it is only in modern times that industry has become sufficiently skilled to isolate and refine metals such as aluminium, and the metals that are added to iron to make steel. "Ex ...
... Emphasise that the least reactive metals were the easiest to isolate and were the first metals to be widely used, and that it is only in modern times that industry has become sufficiently skilled to isolate and refine metals such as aluminium, and the metals that are added to iron to make steel. "Ex ...
File
... Methane is a hydrocarbon, a compound that is composed only of the elements carbon and hydrogen. The combustion of hydrocarbons can be either complete or incomplete. ...
... Methane is a hydrocarbon, a compound that is composed only of the elements carbon and hydrogen. The combustion of hydrocarbons can be either complete or incomplete. ...
CMC Chapter 5
... The Atom and Unanswered Questions • Recall that in Rutherford's model, the atom’s mass is concentrated in the nucleus and electrons move around it. • The model doesn’t explain how the electrons were arranged around the nucleus. • The model doesn’t explain why negatively charged electrons aren’t pul ...
... The Atom and Unanswered Questions • Recall that in Rutherford's model, the atom’s mass is concentrated in the nucleus and electrons move around it. • The model doesn’t explain how the electrons were arranged around the nucleus. • The model doesn’t explain why negatively charged electrons aren’t pul ...
Practice Final Exam, Chemistry 2220, Organic Chem II 1. Rank the
... 6. Which one of the following compounds is NOT a product of reaction between 1,3butadiene and HBr? A. (S)-3-bromo-1-butene B. (R)-3-bromo-1-butene C. (Z)-2-bromo-2-butene D. (E)-1-bromo-2-butene 7. Choose the reagents necessary to carry out the following conversion. O ...
... 6. Which one of the following compounds is NOT a product of reaction between 1,3butadiene and HBr? A. (S)-3-bromo-1-butene B. (R)-3-bromo-1-butene C. (Z)-2-bromo-2-butene D. (E)-1-bromo-2-butene 7. Choose the reagents necessary to carry out the following conversion. O ...
Unit 12 Worksheet Answers
... 38. What is the nuclear charge of an iron atom? a. +26 b. +36 c. +56 d. +82 ...
... 38. What is the nuclear charge of an iron atom? a. +26 b. +36 c. +56 d. +82 ...
1442 Final Review
... 35. What is the best definition of a Brønsted-Lowry base? a) electron-pair acceptor b) electron-pair donor *c) proton acceptor d) proton donor e) produces hydroxide ions in aqueous solutions 36. If the concentration of hydroxide ion in a certain solution is 5.8 x 10-3 M, what is the pH of the soluti ...
... 35. What is the best definition of a Brønsted-Lowry base? a) electron-pair acceptor b) electron-pair donor *c) proton acceptor d) proton donor e) produces hydroxide ions in aqueous solutions 36. If the concentration of hydroxide ion in a certain solution is 5.8 x 10-3 M, what is the pH of the soluti ...
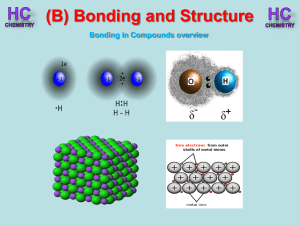
Chapter3 Solutions
... together by pure covalent bonds (O=O, N≡N, Cl−Cl). There is little attraction between the molecules because there are no dipoles, thus the molecules remain separate and the elements are gases. Another factor that contributes is the fact that the elements are relatively light, compared to iodine, for ...
... together by pure covalent bonds (O=O, N≡N, Cl−Cl). There is little attraction between the molecules because there are no dipoles, thus the molecules remain separate and the elements are gases. Another factor that contributes is the fact that the elements are relatively light, compared to iodine, for ...
Net ionic equation
... • Some metals are easily oxidized (lose e–), others are not. • Activity series: list of metals in decreasing ease of ...
... • Some metals are easily oxidized (lose e–), others are not. • Activity series: list of metals in decreasing ease of ...
LESSON ASSIGNMENT LESSON 2 Elements of Chemical Change
... (1) All elements in their free and uncombined state are considered to have a valence of zero. This holds even for those elements that are diatomic molecules in their free state. (2) All atoms can exist in a number of valence states. The common valences which you learned previously are the preferred ...
... (1) All elements in their free and uncombined state are considered to have a valence of zero. This holds even for those elements that are diatomic molecules in their free state. (2) All atoms can exist in a number of valence states. The common valences which you learned previously are the preferred ...
File - Mc Guckin Science
... o) Electron Configuration: a way of showing where the electrons are found in an atom. Includes the number of electrons found in each quantum level of the atom, arranged in order from lowest to highest energy. p) Orbital: a region in three-dimensional space around the nucleus of an atom where there i ...
... o) Electron Configuration: a way of showing where the electrons are found in an atom. Includes the number of electrons found in each quantum level of the atom, arranged in order from lowest to highest energy. p) Orbital: a region in three-dimensional space around the nucleus of an atom where there i ...
Balancing Chemical Equations – A Primer
... Using the Periodic Table, identify the ionic charges of each element Na is element #11. It is found in Column 1. To realize a stable electron configuration, Na will give away one electron. This gives Na a full outer valence shell. The Na ion will have 11+ protons and 10- electrons. It has one more + ...
... Using the Periodic Table, identify the ionic charges of each element Na is element #11. It is found in Column 1. To realize a stable electron configuration, Na will give away one electron. This gives Na a full outer valence shell. The Na ion will have 11+ protons and 10- electrons. It has one more + ...
CHAPTER 1 Differentiate b/w Mendeleev`s periodic law and modern
... is to be given. The reason is that the second incoming electron is repelled byO - 1 . Why does electron affinity decrease from top to bottom in a group? Ans.Electron affinity decreases form top to bottom because atomic size increases due to gradual in number of shells and shielding effect also incre ...
... is to be given. The reason is that the second incoming electron is repelled byO - 1 . Why does electron affinity decrease from top to bottom in a group? Ans.Electron affinity decreases form top to bottom because atomic size increases due to gradual in number of shells and shielding effect also incre ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.