Cation distribution and crystal chemistry of Y3Al5
... only the dodecahedral site, and Al3+ and Ga3+ occupy both the octahedral and the tetrahedral sites. The occupancy of the dodecahedral site was therefore ®xed at unity, and the occupancies of the octahedral and the tetrahedral sites were constrained to maintain the chemical composition of each specim ...
... only the dodecahedral site, and Al3+ and Ga3+ occupy both the octahedral and the tetrahedral sites. The occupancy of the dodecahedral site was therefore ®xed at unity, and the occupancies of the octahedral and the tetrahedral sites were constrained to maintain the chemical composition of each specim ...
REACTIONS IN AQUEOUS SOLUTION
... Water is a very effective solvent for ionic compounds. Although H2O is an electrically neutral molecule, the O atom is rich in electrons and has a partial negative charge, denoted by d- . Each H atom has a partial positive charge, denoted by d+ . Cations are attracted by the negative end of H2O, and ...
... Water is a very effective solvent for ionic compounds. Although H2O is an electrically neutral molecule, the O atom is rich in electrons and has a partial negative charge, denoted by d- . Each H atom has a partial positive charge, denoted by d+ . Cations are attracted by the negative end of H2O, and ...
Experimental Chemistry I
... 1. rinse burette with calibration solution (1.000M) NaOH and fill it up to zero-mark; close stopcock before filling with titrant and clamp burette onto the stand; 2. pipet 25ml of Standardized Acid into the 0.25L Erlenmeyer flask and add approx. 0.1L of deionized water; 3. add 1 drop of PP-indicator ...
... 1. rinse burette with calibration solution (1.000M) NaOH and fill it up to zero-mark; close stopcock before filling with titrant and clamp burette onto the stand; 2. pipet 25ml of Standardized Acid into the 0.25L Erlenmeyer flask and add approx. 0.1L of deionized water; 3. add 1 drop of PP-indicator ...
Multiple-choice questions : 1. Which of the following solutions
... (1) The colour of phenolphthalein changes from colourless to pink at the end point. (2) The colour of phenolphthalein changes from pink to colourless at the end point. (3) The volume of the dilute sodium hydroxide is measured by a pipette. A. (1) only B. C. D. ...
... (1) The colour of phenolphthalein changes from colourless to pink at the end point. (2) The colour of phenolphthalein changes from pink to colourless at the end point. (3) The volume of the dilute sodium hydroxide is measured by a pipette. A. (1) only B. C. D. ...
IIT-JEE - Brilliant Public School Sitamarhi
... A closed packed structure of uniform spheres has the edge length of 534 pm. Calculate the radius of sphere, if it exist in (a) simple cubic lattice (b) BCC lattice (c) FCC lattice ...
... A closed packed structure of uniform spheres has the edge length of 534 pm. Calculate the radius of sphere, if it exist in (a) simple cubic lattice (b) BCC lattice (c) FCC lattice ...
Honors Chemistry Lab Fall
... across the entire surface, and it will continue to do so until a complete monolayer of stearic acid has been formed. If your rst few drops do not spread and evaporate quickly, either your water or watch glass is still dirty. ...
... across the entire surface, and it will continue to do so until a complete monolayer of stearic acid has been formed. If your rst few drops do not spread and evaporate quickly, either your water or watch glass is still dirty. ...
13 CHEMICAL EQUILIBRIUM W MODULE - 5
... A system consisting of only non-volatile liquid and solid phases can be kept even in an open container because such substances have no tendency to escape, e.g. ...
... A system consisting of only non-volatile liquid and solid phases can be kept even in an open container because such substances have no tendency to escape, e.g. ...
國 立 交 通 大 學
... surrounding them determines our capability to control the properties of thin films that to be produced. ...
... surrounding them determines our capability to control the properties of thin films that to be produced. ...
text
... cal reaction. A reaction that releases heat has a negative ∆H and is called exothermic. An endothermic reaction absorbs heat from its surroundings and has a positive ∆H. Entropy is a measure of energy that is unavailable for useful, chemical work. The entropy of an individual species is always posit ...
... cal reaction. A reaction that releases heat has a negative ∆H and is called exothermic. An endothermic reaction absorbs heat from its surroundings and has a positive ∆H. Entropy is a measure of energy that is unavailable for useful, chemical work. The entropy of an individual species is always posit ...
Chapter 4
... nitrate [Ca(NO3)2], are strong electrolytes. It is interesting to note that human body fluids contain many strong and weak electrolytes. Water is a very effective solvent for ionic compounds. Although water is an electrically neutral molecule, it has a positive region (the H atoms) and a negative re ...
... nitrate [Ca(NO3)2], are strong electrolytes. It is interesting to note that human body fluids contain many strong and weak electrolytes. Water is a very effective solvent for ionic compounds. Although water is an electrically neutral molecule, it has a positive region (the H atoms) and a negative re ...
Octahedral and Tetrahedral Cobalt(II) Sites in Cobalt Chloride
... box and room temperature magnetic susceptibility measurements were performed by a Sherwood Scientific MBS-1 balance using Hg[Co(NCS)4] as a standard. Diamagnetic corrections were applied using Pascal's constants and the magnetic moments were calculated.22 Synthesis of [CoCl2(dme)]2 (1) Solvent (dme, ...
... box and room temperature magnetic susceptibility measurements were performed by a Sherwood Scientific MBS-1 balance using Hg[Co(NCS)4] as a standard. Diamagnetic corrections were applied using Pascal's constants and the magnetic moments were calculated.22 Synthesis of [CoCl2(dme)]2 (1) Solvent (dme, ...
Document
... [2] Rapidly solidified liquids are amorphous substances, e.g. Glass, rubber etc. [3] These solids are generally Isotropic. Note : Inotropic substances/Solids are the substances have the same values of physical properties (such as electrical conductivity, refractive index, thermal expansion etc.) in ...
... [2] Rapidly solidified liquids are amorphous substances, e.g. Glass, rubber etc. [3] These solids are generally Isotropic. Note : Inotropic substances/Solids are the substances have the same values of physical properties (such as electrical conductivity, refractive index, thermal expansion etc.) in ...
4 Types of Chemical Reactions and Solution Stoichiometry
... To understand the chemistry that occurs in such diverse places as the human body, the groundwater, the oceans, the local water treatment plant, your hair as you shampoo it, and so on, we must understand how substances dissolved in water react with each other. However, before we can understand soluti ...
... To understand the chemistry that occurs in such diverse places as the human body, the groundwater, the oceans, the local water treatment plant, your hair as you shampoo it, and so on, we must understand how substances dissolved in water react with each other. However, before we can understand soluti ...
CHAPTER TWO SOLID STATE REACTIONS 2.0 Introduction The
... reactions, with product selectivity different from that expected in the liquid phase [8, 9, 16, 17, 18]. It is expected that the crystalline solid state may affect the product selectivity of chemical reactions, not only by rate accelerations but also by slowing down rates of competing processes. Man ...
... reactions, with product selectivity different from that expected in the liquid phase [8, 9, 16, 17, 18]. It is expected that the crystalline solid state may affect the product selectivity of chemical reactions, not only by rate accelerations but also by slowing down rates of competing processes. Man ...
The Mole - Bakersfield College
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
... Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ...
preview as pdf
... Types of Solutes and Solvents Solutes and solvents may be solids, liquids, or gases. The solution that forms has the same physical state as the solvent. When sugar crystals are dissolved in water, the resulting sugar solution is liquid. Sugar is the solute, and water is the solvent. Soda water and s ...
... Types of Solutes and Solvents Solutes and solvents may be solids, liquids, or gases. The solution that forms has the same physical state as the solvent. When sugar crystals are dissolved in water, the resulting sugar solution is liquid. Sugar is the solute, and water is the solvent. Soda water and s ...
Equilibrium - Tenafly High School
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
Equilibrium - AP Chemistry
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
The Equilibrium Constant
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
... 1. Express the equilibrium concentrations of all species in terms of the initial concentrations and a single unknown x, which represents the change in concentration. 2. Write the equilibrium constant expression in terms of the equilibrium concentrations. Knowing the value of the equilibrium constant ...
1. A pharmacy analyst supervises the state of a refractometer. For its
... A Beta-lactam cycle B Thiazolidine cycle C Isoxazole cycle D Furan cycle E Thiadiazole cycle Oxacillin sodium belongs to β-lactam antibiotics. Due to the presence of β-lactam cycle it reacts with hydroxylamine hydrochloride solution in presence of sodium hydroxide solution and copper or iron salts. ...
... A Beta-lactam cycle B Thiazolidine cycle C Isoxazole cycle D Furan cycle E Thiadiazole cycle Oxacillin sodium belongs to β-lactam antibiotics. Due to the presence of β-lactam cycle it reacts with hydroxylamine hydrochloride solution in presence of sodium hydroxide solution and copper or iron salts. ...
Chapter 3 2013
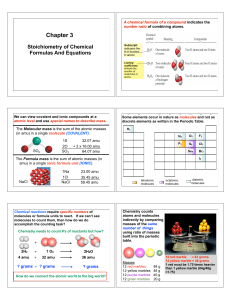
... What’s In A Chemical Formula? Urea, (NH2)2CO, is a nitrogen containing compound used as a fertilizer around the globe? Calculate the following for 25.6 g of urea: a) the molar mass of urea? b) the number of moles of urea in 25.6 g urea? b) # of molecules of urea in 25.6 g of urea? c) # hydrogen ato ...
... What’s In A Chemical Formula? Urea, (NH2)2CO, is a nitrogen containing compound used as a fertilizer around the globe? Calculate the following for 25.6 g of urea: a) the molar mass of urea? b) the number of moles of urea in 25.6 g urea? b) # of molecules of urea in 25.6 g of urea? c) # hydrogen ato ...
Grade XII Unit 1 - Ethiopian Ministry of Education
... Ethanol mixes with water but oil does not. Why? Solubility is a measure of how much solute will dissolve in a solvent at a specific temperature. Do you know the principle “like dissolves like”? The “like dissolves like” principle is helpful in predicting the solubility of a substance in a given solv ...
... Ethanol mixes with water but oil does not. Why? Solubility is a measure of how much solute will dissolve in a solvent at a specific temperature. Do you know the principle “like dissolves like”? The “like dissolves like” principle is helpful in predicting the solubility of a substance in a given solv ...
Unit 2: Matter as Solutions and Gases
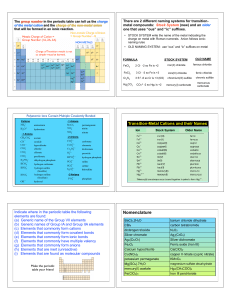
... 2. Most F− are soluble (except with Li+, Mg2+, Ca2+, Sr2+, Ba2+ and Fe2+ Hg22+ and Pb2+). 3. Most Cl−, Br−, and I− salts are soluble (except with Cu+, Ag+, Hg22+, Hg2+, and Pb2+). 4. Most SO42− are soluble (except with Ca2+, Sr2+, Ba2+, Hg22+, Pb2+ and Ag+). 5. Only H+, NH4+, Na+, K+ cations with PO ...
... 2. Most F− are soluble (except with Li+, Mg2+, Ca2+, Sr2+, Ba2+ and Fe2+ Hg22+ and Pb2+). 3. Most Cl−, Br−, and I− salts are soluble (except with Cu+, Ag+, Hg22+, Hg2+, and Pb2+). 4. Most SO42− are soluble (except with Ca2+, Sr2+, Ba2+, Hg22+, Pb2+ and Ag+). 5. Only H+, NH4+, Na+, K+ cations with PO ...
Review Packet Answers - Bremerton School District
... The equilibrium pressure of NH3 gas would decrease. In order for the pressure equilibrium constant, Kp, to remain constant, the equilibrium pressure of NH3 must decrease when the pressure of H2S is increased. Kp = (PNH3) (PH2S) (A complete explanation based on Le Chatelier's principle is also accept ...
... The equilibrium pressure of NH3 gas would decrease. In order for the pressure equilibrium constant, Kp, to remain constant, the equilibrium pressure of NH3 must decrease when the pressure of H2S is increased. Kp = (PNH3) (PH2S) (A complete explanation based on Le Chatelier's principle is also accept ...
Crystallization

Crystallization is the (natural or artificial) process of formation of solid crystals precipitating from a solution, melt or more rarely deposited directly from a gas. Crystallization is also a chemical solid–liquid separation technique, in which mass transfer of a solute from the liquid solution to a pure solid crystalline phase occurs. In chemical engineering crystallization occurs in a crystallizer. Crystallization is therefore an aspect of precipitation, obtained through a variation of the solubility conditions of the solute in the solvent, as compared to precipitation due to chemical reaction.