- Kendriya Vidyalaya Jhunjhunu
... Three Marks Question 1. The oxide of a metal M was water soluble. When a blue litmus strip was dipped in this solution, it did not undergo any change in colour. Predict the nature of the oxide. 2. Why does bee-sting cause pain and irritation? What relief can be given in such a case immediately? 3. ...
... Three Marks Question 1. The oxide of a metal M was water soluble. When a blue litmus strip was dipped in this solution, it did not undergo any change in colour. Predict the nature of the oxide. 2. Why does bee-sting cause pain and irritation? What relief can be given in such a case immediately? 3. ...
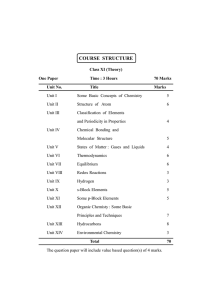
COURSE STRUCTURE
... 100 mL of concentrated aqueous sulphuric acid which contains 98% H2SO4 by mass. The density of conc. H2SO4 is 1.84 g/mL. NaOH reacts with H2SO4 according to the following equation : 2NaOH + H2SO4 Na2SO4 + 2H2O (At. mass/g mol1 H = 1, S = 32, O = 16) (a) How much Cu can be obtained from 100 g CuSO ...
... 100 mL of concentrated aqueous sulphuric acid which contains 98% H2SO4 by mass. The density of conc. H2SO4 is 1.84 g/mL. NaOH reacts with H2SO4 according to the following equation : 2NaOH + H2SO4 Na2SO4 + 2H2O (At. mass/g mol1 H = 1, S = 32, O = 16) (a) How much Cu can be obtained from 100 g CuSO ...
Chemistry STUDY OF VOLUMETRIC AND VISCOMETRIC
... estimate the correction to be applied to due to hydrolysis has shown that the magnitude of the correction lies within the range of uncertainties at the lowest concentrations studied. It was much smaller at higher concentrations due to the decrease of the degree of hydrolysis [5, 6]. The changes o ...
... estimate the correction to be applied to due to hydrolysis has shown that the magnitude of the correction lies within the range of uncertainties at the lowest concentrations studied. It was much smaller at higher concentrations due to the decrease of the degree of hydrolysis [5, 6]. The changes o ...
Chemistry Content Review Notes
... The document is a compilation of information found in the Virginia Department of Education (VDOE) Curriculum Framework, Enhanced Scope and Sequence, and Released Test items. In addition to VDOE information, Glencoe Textbook Series and resources have been used. Finally, information from various websi ...
... The document is a compilation of information found in the Virginia Department of Education (VDOE) Curriculum Framework, Enhanced Scope and Sequence, and Released Test items. In addition to VDOE information, Glencoe Textbook Series and resources have been used. Finally, information from various websi ...
Synthesis of Copper (II)-Containing Nickel (II) Hydroxide Particles as
... Ni1-xCuxO and its precursor, copper(II)-containing nickel(II) hydroxide, have potential applications in different fields of current interest. The mixed oxides provide the active materials of catalysts used in the production of synthesis gas from steam-reforming of ethanol21 and in the reduction of N ...
... Ni1-xCuxO and its precursor, copper(II)-containing nickel(II) hydroxide, have potential applications in different fields of current interest. The mixed oxides provide the active materials of catalysts used in the production of synthesis gas from steam-reforming of ethanol21 and in the reduction of N ...
IA Velikanova, AK Bolvako PHYSICAL CHEMISTRY
... determined by passing a known amount of electricity through the system before the reaction, and determining the resulting temperature change. The temperature rise on reaction could be measured using a precision thermometer. In the experiment you will measure the enthalpies of process of salt dissolu ...
... determined by passing a known amount of electricity through the system before the reaction, and determining the resulting temperature change. The temperature rise on reaction could be measured using a precision thermometer. In the experiment you will measure the enthalpies of process of salt dissolu ...
industry: applying chemical reactions
... The promise of 200 new jobs is alluring. However, all of these jobs would be in only one company. What will happen if there’s a decline in the market for ammonia or lithium-ion batteries? Wouldn’t it be more prudent to distribute those 200 jobs among several different companies? Let’s learn from our ...
... The promise of 200 new jobs is alluring. However, all of these jobs would be in only one company. What will happen if there’s a decline in the market for ammonia or lithium-ion batteries? Wouldn’t it be more prudent to distribute those 200 jobs among several different companies? Let’s learn from our ...
Test 1 w/answers
... 8. The osmotic pressure of a solution of 20.00 g NaOH (MMNaOH = 40.0 g/mol) dissolved in a total volume of 500.0 mL was compared to the osmotic pressure of a solution of 14.61 g of NaCl (MMNaCl = 58.44 g/mol) dissolved in a total volume of 250.0 mL. Is the osmotic pressure of the NaOH solution highe ...
... 8. The osmotic pressure of a solution of 20.00 g NaOH (MMNaOH = 40.0 g/mol) dissolved in a total volume of 500.0 mL was compared to the osmotic pressure of a solution of 14.61 g of NaCl (MMNaCl = 58.44 g/mol) dissolved in a total volume of 250.0 mL. Is the osmotic pressure of the NaOH solution highe ...
CHAPTER 4: CHEMICAL QUANTITIES and AQUEOUS REACTIONS
... Exercise 2: On the basis of solubility rules, predict whether aluminum nitrate and iron (III) phosphate are soluble in water or not. Aluminum nitrate – Soluble (rule 1) Iron (III) phosphate – Not soluble (rule 6) ...
... Exercise 2: On the basis of solubility rules, predict whether aluminum nitrate and iron (III) phosphate are soluble in water or not. Aluminum nitrate – Soluble (rule 1) Iron (III) phosphate – Not soluble (rule 6) ...
Integrated Chemical Systems
... width of 16 G at room temperature (Figure 1A). Since the ESR spectrum of the Ti(II1) ion in Nafion is readily observable at room temperature, the configuration of ligands around Ti(II1) in Nafion clearly deviates from a species with perfect octahedral symmetry, which is ESR-inactive due to very rapi ...
... width of 16 G at room temperature (Figure 1A). Since the ESR spectrum of the Ti(II1) ion in Nafion is readily observable at room temperature, the configuration of ligands around Ti(II1) in Nafion clearly deviates from a species with perfect octahedral symmetry, which is ESR-inactive due to very rapi ...
Kitchen Chemistry Review
... the reacting substances is a. always more than the total mass of the products. b. always less than the total mass of the products. c. sometimes more and sometimes less than the total mass of the products. d. always equal to the total mass of the products. Answer: D ...
... the reacting substances is a. always more than the total mass of the products. b. always less than the total mass of the products. c. sometimes more and sometimes less than the total mass of the products. d. always equal to the total mass of the products. Answer: D ...
Research on Hydrogenation of FAME to Fatty Alcohols
... biodiesel industry. However, the choice of target products and technologic routes for producing them should follow three principles: 1) biomass feedstocks are cheaper than petroleum products when they are used as raw materials, 2) process being developed is simple and environmentally-friendly, 3) th ...
... biodiesel industry. However, the choice of target products and technologic routes for producing them should follow three principles: 1) biomass feedstocks are cheaper than petroleum products when they are used as raw materials, 2) process being developed is simple and environmentally-friendly, 3) th ...
5. Formulae, equations and amounts of substance
... conditions of temperature and pressure contain equal numbers of molecules (or atoms if the gas in monatomic) ...
... conditions of temperature and pressure contain equal numbers of molecules (or atoms if the gas in monatomic) ...
File - Fidaa`s Level 2 Portfolio
... carboxylic acids, ketones, ethers, amines, and benzenes. They all have different properties that make them unique and are very important especially for this specific lab. Esters, the main topic for this lab, are part of the artificial flavors we will be making. Ester synthesis is when a chemical rea ...
... carboxylic acids, ketones, ethers, amines, and benzenes. They all have different properties that make them unique and are very important especially for this specific lab. Esters, the main topic for this lab, are part of the artificial flavors we will be making. Ester synthesis is when a chemical rea ...
Chemical Reactions
... describe what they see. (light begins to dim as solid forms) 10. Add the last 5 mL drop-by-drop until the endpoint is reached (phenolphthalein will turn pink and remain pink for 30 seconds and the light will go out) 11. Ask your students to explain their observations. 12. Set the resultant material ...
... describe what they see. (light begins to dim as solid forms) 10. Add the last 5 mL drop-by-drop until the endpoint is reached (phenolphthalein will turn pink and remain pink for 30 seconds and the light will go out) 11. Ask your students to explain their observations. 12. Set the resultant material ...
Electrochemistry
... When a metal rod is dipped in its salt solution, two charges occur. (a) Metal ions pass from the electrode into solution leaving an excess of electrons and thus a negative charge on the electrode.. (b) Metal ions in solution gain electrons from the electrode leaving a positive charge on the electrod ...
... When a metal rod is dipped in its salt solution, two charges occur. (a) Metal ions pass from the electrode into solution leaving an excess of electrons and thus a negative charge on the electrode.. (b) Metal ions in solution gain electrons from the electrode leaving a positive charge on the electrod ...
Qualitative Analysis of Anions
... Add 1 mL of 0.1M AgNO3. (Note: If you are testing the unknown and no precipitate forms at this point, the lack of precipitate proves the absence of CI , Br and I ) If a precipitate forms, note the color of the precipitate. AgCl is white, AgBr is cream colored, and AgI is yellow. The color may be a c ...
... Add 1 mL of 0.1M AgNO3. (Note: If you are testing the unknown and no precipitate forms at this point, the lack of precipitate proves the absence of CI , Br and I ) If a precipitate forms, note the color of the precipitate. AgCl is white, AgBr is cream colored, and AgI is yellow. The color may be a c ...
IChO 2012
... One of the simplest boron-nitrogen compounds is H3N–BH3, the ammonia-borane adduct. Pyrolysis of this compound leads to the generation of H2 gas and polyborazylene. H3N–BH3(s) 2.5 H2(g) + (polyborazylene, BNH) (If an efficient and low-cost method can be found to regenerate H 3N–BH3 from BNH, the s ...
... One of the simplest boron-nitrogen compounds is H3N–BH3, the ammonia-borane adduct. Pyrolysis of this compound leads to the generation of H2 gas and polyborazylene. H3N–BH3(s) 2.5 H2(g) + (polyborazylene, BNH) (If an efficient and low-cost method can be found to regenerate H 3N–BH3 from BNH, the s ...
Part One: Ions in Aqueous Solution A. Electrolytes and Non
... Combustion reaction = a reaction in which a substance reacts with oxygen, sometimes with rapid release of heat to product a flame. ...
... Combustion reaction = a reaction in which a substance reacts with oxygen, sometimes with rapid release of heat to product a flame. ...
updated chem cp final review key
... g. Breaking a reactant into smaller pieces Increases rate of reaction 43. Know what conditions are true of a chemical reaction at equilibrium. (1) rates of forward and reverse reactions are equal. (2) The concentrations of all substances involved stop changing (3) reactants and products are both pre ...
... g. Breaking a reactant into smaller pieces Increases rate of reaction 43. Know what conditions are true of a chemical reaction at equilibrium. (1) rates of forward and reverse reactions are equal. (2) The concentrations of all substances involved stop changing (3) reactants and products are both pre ...
A Few Things You Might Want To Know
... Depends on attractive forces and structure (long hydrocarbons!) ...
... Depends on attractive forces and structure (long hydrocarbons!) ...
rate of chemical reaction and chemical equilibrium
... OF DYNAMIC EQUILIBRIUM (i) At equilibrium, macroscopic (observable) properties such as concentration, density, colour, etc., are constant under the given condition of temperature, pressure and initial amount of the substances. (ii) At equilibrium, microscopic (at molecular level) processes continue, ...
... OF DYNAMIC EQUILIBRIUM (i) At equilibrium, macroscopic (observable) properties such as concentration, density, colour, etc., are constant under the given condition of temperature, pressure and initial amount of the substances. (ii) At equilibrium, microscopic (at molecular level) processes continue, ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.