Appendices and Glossary
... but the height did not (going from six feet to 72 inches, the person did not grow or shrink). This is what we would expect upon multiplication by 1. Although this is a trivial example, we will use this same conversion factor or factor label approach for nearly all of the stoichiometric calculations ...
... but the height did not (going from six feet to 72 inches, the person did not grow or shrink). This is what we would expect upon multiplication by 1. Although this is a trivial example, we will use this same conversion factor or factor label approach for nearly all of the stoichiometric calculations ...
Chemistry Standards Clarification
... laboratory or field. Evaluate the uncertainties or validity of scientific conclusions using an understanding of sources of measurement error, the challenges of controlling variables, accuracy of data analysis, logic of argument, logic of experimental design, and/or the dependence on underlying assum ...
... laboratory or field. Evaluate the uncertainties or validity of scientific conclusions using an understanding of sources of measurement error, the challenges of controlling variables, accuracy of data analysis, logic of argument, logic of experimental design, and/or the dependence on underlying assum ...
Chapter Ten
... ► Salt solutions can be neutral, acidic, or basic, depending on the ions present, because some ions react with water to produce H+ and some ions react with water to produce OH-. ► To predict the acidity of a salt solution, it is convenient to classify salts according to the acid and base from which ...
... ► Salt solutions can be neutral, acidic, or basic, depending on the ions present, because some ions react with water to produce H+ and some ions react with water to produce OH-. ► To predict the acidity of a salt solution, it is convenient to classify salts according to the acid and base from which ...
Document
... group is MORE positive than it was in the C-O-H group By increasing the number of highly electronegative O, more electrons were drawn away from that C, making it more positive. ...
... group is MORE positive than it was in the C-O-H group By increasing the number of highly electronegative O, more electrons were drawn away from that C, making it more positive. ...
Challenge Problems
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
Chemical Equilibrium - The Gurukul Institute
... (NH4)2 CO3(s) ⇌ 2NH3(g) + CO2(g) + H2O(g) At a certain elevated temperature, the total pressure of the gases generated was 0.42 atm. At equilibrium. Calculate the equilibrium constant for the reaction. 10. if a given quantity of phosphorus pentachloride is heated at 250o C and allowed to come to equ ...
... (NH4)2 CO3(s) ⇌ 2NH3(g) + CO2(g) + H2O(g) At a certain elevated temperature, the total pressure of the gases generated was 0.42 atm. At equilibrium. Calculate the equilibrium constant for the reaction. 10. if a given quantity of phosphorus pentachloride is heated at 250o C and allowed to come to equ ...
Chemistry
... subject or as part of a balanced science course. This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need for students to develop ski ...
... subject or as part of a balanced science course. This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need for students to develop ski ...
The Oxidation States of Tin
... reason that this yield was low is mainly attributed to the iodine-zinc portion of this reaction. This part of the experiment required that the zinc and iodine be combined in a flask with water and allowed to sit in an ice bath for an extended amount of time. This reaction was performed five times w ...
... reason that this yield was low is mainly attributed to the iodine-zinc portion of this reaction. This part of the experiment required that the zinc and iodine be combined in a flask with water and allowed to sit in an ice bath for an extended amount of time. This reaction was performed five times w ...
Unit 11 acids and bases part 1
... Salts are the ionic product of an acid base neutralization reaction. Acidic Salts are formed from a strong acid and a weak base. Neutral salts are formed from a strong acid and strong base. Basic salts are formed from a strong base and a weak acid. Give the acid and base the following salts were for ...
... Salts are the ionic product of an acid base neutralization reaction. Acidic Salts are formed from a strong acid and a weak base. Neutral salts are formed from a strong acid and strong base. Basic salts are formed from a strong base and a weak acid. Give the acid and base the following salts were for ...
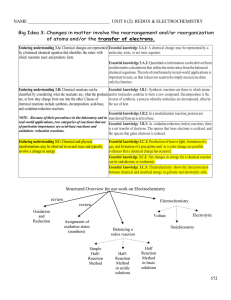
Chapter 19
... which sodium (Na) and chlorine (Cl 2) react to form the ionic compound sodium chloride (NaCl), an electron from each of two sodium atoms is transferred to the Cl 2 molecule to form two Cl - ions. Complete chemical equation: 2Na(s) + Cl 2(g) → 2NaCl(s) Net ionic equation: 2Na(s) + Cl 2(g) → 2Na + + 2 ...
... which sodium (Na) and chlorine (Cl 2) react to form the ionic compound sodium chloride (NaCl), an electron from each of two sodium atoms is transferred to the Cl 2 molecule to form two Cl - ions. Complete chemical equation: 2Na(s) + Cl 2(g) → 2NaCl(s) Net ionic equation: 2Na(s) + Cl 2(g) → 2Na + + 2 ...
Chapter 4 Chemical Reactions and Solution Stoichiometry 4.1
... When mixed, a double displacement reaction takes place, forming the soluble compound NaNO3 and the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The ...
... When mixed, a double displacement reaction takes place, forming the soluble compound NaNO3 and the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The ...
Collected Essays chapter 13 answers
... (b) Using the equilibrium concentrations given above, calculate the value of Kc, the equilibrium constant for the reaction. 5.0 (c) Determine Kp, in terms of Kc for this system. Kp = 5.0 (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to ...
... (b) Using the equilibrium concentrations given above, calculate the value of Kc, the equilibrium constant for the reaction. 5.0 (c) Determine Kp, in terms of Kc for this system. Kp = 5.0 (d) When the system is cooled from 2,000 K to a lower temperature, 30.0 percent of the CO(g) is converted back to ...
General chemistry laboratory activities, Lorentz
... Figure 2. Reaction flask, distillation arm, reagent flask, and glass tubing Reaction flasks (see Figure 2) are usually spherical (i.e. round-bottom flask) and are accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (suc ...
... Figure 2. Reaction flask, distillation arm, reagent flask, and glass tubing Reaction flasks (see Figure 2) are usually spherical (i.e. round-bottom flask) and are accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (suc ...
Chem 11 Stoichiometry (mol-mol) Using the formulas we have
... Proof of the Conservation of Mass: From the balanced equation, we can say that 1 molecule of N2 and 3 molecules of H2 combine to form 2 molecules of NH3 OR we can say that 1 mole of N2 and 3 moles of H2 combine to form 2 moles of NH3. Using the coefficients as moles, we can determine the mass of the ...
... Proof of the Conservation of Mass: From the balanced equation, we can say that 1 molecule of N2 and 3 molecules of H2 combine to form 2 molecules of NH3 OR we can say that 1 mole of N2 and 3 moles of H2 combine to form 2 moles of NH3. Using the coefficients as moles, we can determine the mass of the ...
File
... Take out your stapled packet and complete the left side of the second page (stoichiometry and limiting reagents) Quiz tomorrow will be based on the left side ...
... Take out your stapled packet and complete the left side of the second page (stoichiometry and limiting reagents) Quiz tomorrow will be based on the left side ...
1. What energy changes occur when chemical bonds are formed
... The reaction is spontaneous at low temperatures but becomes non-spontaneous at high temperatures. ...
... The reaction is spontaneous at low temperatures but becomes non-spontaneous at high temperatures. ...
Thermochemistry Diploma Questions
... all current vehicles operate on solar cells. solar energy warms a vehicle’s interior on a sunny day. the energy stored in fossil fuels is a result of photosynthesis. solar energy is the ideal renewable, non-polluting energy source. ...
... all current vehicles operate on solar cells. solar energy warms a vehicle’s interior on a sunny day. the energy stored in fossil fuels is a result of photosynthesis. solar energy is the ideal renewable, non-polluting energy source. ...
Acids and Bases
... all bitter compounds are basic). Caffeine and milk of magnesia (chemical formula Mg(OH)2 ) are two bases that you may have had the opportunity to taste, although the bitterness is generally masked by other flavors when these compounds are consumed. Other common bases are found in a number of cleaning ...
... all bitter compounds are basic). Caffeine and milk of magnesia (chemical formula Mg(OH)2 ) are two bases that you may have had the opportunity to taste, although the bitterness is generally masked by other flavors when these compounds are consumed. Other common bases are found in a number of cleaning ...
1.2 Calculations
... conditions of temperature and pressure contain equal numbers of molecules (or atoms if the gas in monatomic) ...
... conditions of temperature and pressure contain equal numbers of molecules (or atoms if the gas in monatomic) ...
AP Chemistry
... Mg(OH)2(s) + 2 H+ Mg2+ + 2 H2O CuS(s) + 2 H+ Cu2+ + H2S(g) c. salts with Cl-, Br-, I-, SO42- are unaffected by H+ 3. formation of complex ions a. polar molecules or anions are strongly attracted to transition metal cation forming a complex ion 1. polar molecule or anion is called ligand a. molec ...
... Mg(OH)2(s) + 2 H+ Mg2+ + 2 H2O CuS(s) + 2 H+ Cu2+ + H2S(g) c. salts with Cl-, Br-, I-, SO42- are unaffected by H+ 3. formation of complex ions a. polar molecules or anions are strongly attracted to transition metal cation forming a complex ion 1. polar molecule or anion is called ligand a. molec ...
Chapter 6: Thermochemistry
... 36. A 26.2 g piece of copper metal is heated from 21.5°C to 201.6°C. Calculate the amount of heat absorbed by the metal. The specific heat of Cu is 0.385 J/g·°C. 37. The value of H°rxn for the following reaction is –6535 kJ/mol. 2C6H6(l) + 15O2(g) 12CO2(g) + 6H2O(g) How many kilojoules of heat wi ...
... 36. A 26.2 g piece of copper metal is heated from 21.5°C to 201.6°C. Calculate the amount of heat absorbed by the metal. The specific heat of Cu is 0.385 J/g·°C. 37. The value of H°rxn for the following reaction is –6535 kJ/mol. 2C6H6(l) + 15O2(g) 12CO2(g) + 6H2O(g) How many kilojoules of heat wi ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.