QUATERLY 3 REVIEW CHAPTER 12- Stoichiometry Define the law
... 11. If two containers are at the same temperature and pressure and their volumes are equal, they must contain the same ________________________________. Which scientist said this? ...
... 11. If two containers are at the same temperature and pressure and their volumes are equal, they must contain the same ________________________________. Which scientist said this? ...
CHE 1401 - Spring 2016 - Chapter 5 Homework 5 (Chapter 5
... the heat capacity of the calorimeter is measured to be 9.84 kJ/K. The heat of reaction for the combustion of a mole of Ti in this calorimeter is __________ kJ/mol. A) -0.154 B) -1.49 × 104 C) 2.67 D) -311 E) 4.98 31) The British thermal unit (Btu) is commonly used in engineering applications. A Btu ...
... the heat capacity of the calorimeter is measured to be 9.84 kJ/K. The heat of reaction for the combustion of a mole of Ti in this calorimeter is __________ kJ/mol. A) -0.154 B) -1.49 × 104 C) 2.67 D) -311 E) 4.98 31) The British thermal unit (Btu) is commonly used in engineering applications. A Btu ...
Faculty of Chemistry, Brno University of Technology Purkynova 118
... active chemical species such as hydroxyl radicals, hydrogen and oxygen radicals, hydrogen peroxide etc. are produced in water by this kind of the discharge [1]. These active species initiate many further chemical reactions. Therefore the processes taking place in such discharges can be applied in fi ...
... active chemical species such as hydroxyl radicals, hydrogen and oxygen radicals, hydrogen peroxide etc. are produced in water by this kind of the discharge [1]. These active species initiate many further chemical reactions. Therefore the processes taking place in such discharges can be applied in fi ...
CHE 1401 - Summer 2012 - Chapter 5 Homework 5 (Chapter 5
... B) A negative ΔH corresponds to an exothermic process. C) ΔE = Efinal - Einitial D) Energy lost by the system must be gained by the surroundings. E) 1 cal = 4.184 J (exactly) 9) The British thermal unit (Btu) is commonly used in engineering applications. A Btu is defined as the amount of heat requir ...
... B) A negative ΔH corresponds to an exothermic process. C) ΔE = Efinal - Einitial D) Energy lost by the system must be gained by the surroundings. E) 1 cal = 4.184 J (exactly) 9) The British thermal unit (Btu) is commonly used in engineering applications. A Btu is defined as the amount of heat requir ...
CHE 1401 - Fall 2016 - Chapter 5 Homework 5 (Chapter 5
... is __________, and therefore heat is __________ by the reaction. A) exothermic, absorbed B) exothermic, released C) endothermic, released D) endothermic, absorbed E) thermoneutral, neither released nor absorbed 27) Which of the following is a statement of Hess's law? A) The ΔH for a process in the f ...
... is __________, and therefore heat is __________ by the reaction. A) exothermic, absorbed B) exothermic, released C) endothermic, released D) endothermic, absorbed E) thermoneutral, neither released nor absorbed 27) Which of the following is a statement of Hess's law? A) The ΔH for a process in the f ...
Free Energy I
... An irreversible process is one in which the system and surroundings cannot be restored to their original state by exactly reversing the change. dropping a vase and breaking it reacting hydrogen and oxygen to form water burning a match ...
... An irreversible process is one in which the system and surroundings cannot be restored to their original state by exactly reversing the change. dropping a vase and breaking it reacting hydrogen and oxygen to form water burning a match ...
CHE 1401 - Spring 2015 - Chapter 5 Homework 5 (Chapter 5
... E) thermoneutral, neither released nor absorbed 36) When a system __________, ΔE is always negative. A) absorbs heat and has work done on it B) gives off heat and has work done on it C) gives off heat and does work D) absorbs heat and does work E) none of the above is always negative. ...
... E) thermoneutral, neither released nor absorbed 36) When a system __________, ΔE is always negative. A) absorbs heat and has work done on it B) gives off heat and has work done on it C) gives off heat and does work D) absorbs heat and does work E) none of the above is always negative. ...
Test Chap 5 gas laws
... 12. A sealed flask contains 1 molecule of hydrogen for every 3 molecules of helium at 20 °C. If the total pressure is 400 kPa, what is the partial pressure of the hydrogen? (3 pts) 16. What is the pressure exerted by some nitrogen gas collected in a tube filled with water on a day when the room temp ...
... 12. A sealed flask contains 1 molecule of hydrogen for every 3 molecules of helium at 20 °C. If the total pressure is 400 kPa, what is the partial pressure of the hydrogen? (3 pts) 16. What is the pressure exerted by some nitrogen gas collected in a tube filled with water on a day when the room temp ...
CHE 1401 - Fall 2015 - Chapter 5 Homework 5 (Chapter 5
... MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The British thermal unit (Btu) is commonly used in engineering applications. A Btu is defined as the amount of heat required to raise the temperature of 1 lb of water by 1°F. There are _________ ...
... MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The British thermal unit (Btu) is commonly used in engineering applications. A Btu is defined as the amount of heat required to raise the temperature of 1 lb of water by 1°F. There are _________ ...
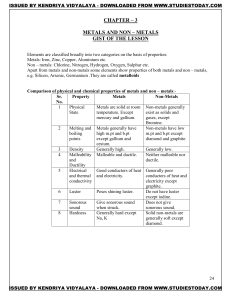
METALS AND NON – METALS Concepts
... 4. Conduction of electricity:ionic compounds in solid state-----does not conduct electricity. Reason—Ions can not move due to rigid solid structure. Ionic compounds conduct electricity in molten state. Reason-- Ions can move freely since the electrostatic forces of attraction between the oppositely ...
... 4. Conduction of electricity:ionic compounds in solid state-----does not conduct electricity. Reason—Ions can not move due to rigid solid structure. Ionic compounds conduct electricity in molten state. Reason-- Ions can move freely since the electrostatic forces of attraction between the oppositely ...
Combined
... 6. Chlorine has a relative atomic mass of 35.5 and has two isotopes with relative isotopic masses of 35 and 37. Which of the following statements about chlorine are CORRECT? (1) The isotopes have same atomic number. (2) It contains the two isotopes, chlorine-35 and chlorine-37, in a ratio of 1:3. (3 ...
... 6. Chlorine has a relative atomic mass of 35.5 and has two isotopes with relative isotopic masses of 35 and 37. Which of the following statements about chlorine are CORRECT? (1) The isotopes have same atomic number. (2) It contains the two isotopes, chlorine-35 and chlorine-37, in a ratio of 1:3. (3 ...
Chem Sheets to Memorize SOLUBILITY CHART
... *Please do all questions on a separate piece of paper. *You will need to be able to write molecular chemical reactions and do mole conversions to do these questions. 1. 30.5 g of sodium metal reacts with a solution of excess lithium bromide. How many grams of lithium metal are produced? 2. How many ...
... *Please do all questions on a separate piece of paper. *You will need to be able to write molecular chemical reactions and do mole conversions to do these questions. 1. 30.5 g of sodium metal reacts with a solution of excess lithium bromide. How many grams of lithium metal are produced? 2. How many ...
Gen Chem Final--review problems Fall 2006
... For the precipitation reaction/s above in problem 1, please add the appropriate ‘state’ (i.e. solid or aq) to each species. For the oxidation/reduction reaction/s above, please identify the species that is being oxidized and the species being reduced and assign oxidation numbers to each atom. For th ...
... For the precipitation reaction/s above in problem 1, please add the appropriate ‘state’ (i.e. solid or aq) to each species. For the oxidation/reduction reaction/s above, please identify the species that is being oxidized and the species being reduced and assign oxidation numbers to each atom. For th ...
Chem Sheets to Memorize
... *Please do all questions on a separate piece of paper. *You will need to be able to write molecular chemical reactions and do mole conversions to do these questions. 1. 30.5 g of sodium metal reacts with a solution of excess lithium bromide. How many grams of lithium metal are produced? 2. How many ...
... *Please do all questions on a separate piece of paper. *You will need to be able to write molecular chemical reactions and do mole conversions to do these questions. 1. 30.5 g of sodium metal reacts with a solution of excess lithium bromide. How many grams of lithium metal are produced? 2. How many ...
Science 10 Chem - Holy Trinity Academy
... Lesson 4: Pure Substance: made of only one kind of matter and has a unique set of properties (chemical and physical). e.g., mercury (element) and sugar (compound). Element: a substance that cannot be broken down any further by a chemical reaction into any simpler substance. pure substances that ...
... Lesson 4: Pure Substance: made of only one kind of matter and has a unique set of properties (chemical and physical). e.g., mercury (element) and sugar (compound). Element: a substance that cannot be broken down any further by a chemical reaction into any simpler substance. pure substances that ...
Chapter 11 Chemical Reactions
... Example (needs to be a double replacement reaction) AgNO3 + NaCl AgCl + NaNO3 1. this is the full balanced equation 2. next, write it as an ionic equation by splitting the compounds into their ions: Ag1+ + NO31- + Na1+ + Cl1- AgCl + Na1+ + NO31Note that the AgCl did not ionize, because it is a “ ...
... Example (needs to be a double replacement reaction) AgNO3 + NaCl AgCl + NaNO3 1. this is the full balanced equation 2. next, write it as an ionic equation by splitting the compounds into their ions: Ag1+ + NO31- + Na1+ + Cl1- AgCl + Na1+ + NO31Note that the AgCl did not ionize, because it is a “ ...
Chemistry Cram Sheet
... 4. The numbers you get are the subscripts in your formula. To calculate M.F.: Divide the molar mass (given) by the empirical mass. Multiply that number by the E.F. A compound is 80% C and 20% H. If the molar mass of the molecular formula is 45 g/mol, find the empirical and molecular formulas of this ...
... 4. The numbers you get are the subscripts in your formula. To calculate M.F.: Divide the molar mass (given) by the empirical mass. Multiply that number by the E.F. A compound is 80% C and 20% H. If the molar mass of the molecular formula is 45 g/mol, find the empirical and molecular formulas of this ...
Calculation Booklet - Clydebank High School
... 14.9g of potassium chloride, KCl, is dissolved in 0.2kg of water. The temperature falls from 22.5oC to 18.5oC. Calculate the enthalpy of solution of potassium chloride. 0.05 mol of a compound is dissolved in 500cm3 of water causing the temperature to rise from 19oC to 21oC. Find the enthalpy of solu ...
... 14.9g of potassium chloride, KCl, is dissolved in 0.2kg of water. The temperature falls from 22.5oC to 18.5oC. Calculate the enthalpy of solution of potassium chloride. 0.05 mol of a compound is dissolved in 500cm3 of water causing the temperature to rise from 19oC to 21oC. Find the enthalpy of solu ...
9077590 Chem. Rege. Jan. 01
... increasing atomic number, the van der Waals forces between the atoms in a given sample of each of these gases (1) decrease (2) increase (3) remains the same ...
... increasing atomic number, the van der Waals forces between the atoms in a given sample of each of these gases (1) decrease (2) increase (3) remains the same ...
dutch national chemistry olympiad
... Aspro-Clear tablets contain also citric acid (C6H8O7, molecular mass 192.1 u), which reacts with hydrogen carbonate producing carbon dioxide. Acetylsalicylic acid is a monovalent acid and citric acid is a trivalent acid. However, when the gas production, occurring after an Aspro-Clear tablet is put ...
... Aspro-Clear tablets contain also citric acid (C6H8O7, molecular mass 192.1 u), which reacts with hydrogen carbonate producing carbon dioxide. Acetylsalicylic acid is a monovalent acid and citric acid is a trivalent acid. However, when the gas production, occurring after an Aspro-Clear tablet is put ...
AQA GCSE Chemistry My Revision Notes
... (a) Nitrogen is taken from the air. (b) There is more chance of them colliding/coming into contact with each other. (c) The rate of reaction is increased by using an iron catalyst , at high temperatures and high pressures . (d) Percentage yield = 49% (e) Temperature: lower temperature in ...
... (a) Nitrogen is taken from the air. (b) There is more chance of them colliding/coming into contact with each other. (c) The rate of reaction is increased by using an iron catalyst , at high temperatures and high pressures . (d) Percentage yield = 49% (e) Temperature: lower temperature in ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.