H 2 - PPC10
... Ps and H2 yields given by quantitative model of intratrack processes based on the above reaction scheme ...
... Ps and H2 yields given by quantitative model of intratrack processes based on the above reaction scheme ...
Chapter 4 (Hill/Petrucci/McCreary/Perry Chemical Reactions in
... <<100% ionization in water = weak base Most common weak bases: NH3 and amines Reaction of Acids with Bases: Neutralization neutralization reaction: the reaction of ionizable H+ ions on acid molecules with OH1- or other anions (such as HCO3 1- or CO3 2-) on base “molecules” Example. We represent an a ...
... <<100% ionization in water = weak base Most common weak bases: NH3 and amines Reaction of Acids with Bases: Neutralization neutralization reaction: the reaction of ionizable H+ ions on acid molecules with OH1- or other anions (such as HCO3 1- or CO3 2-) on base “molecules” Example. We represent an a ...
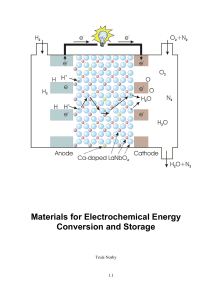
Materials for Electrochemical Energy Conversion and Storage
... For electrolytic cells (electrolysers and charging batteries) the same sum of resistances applies, now only with the current having the opposite sign, whereby the overpotentials serve to increase the applied voltage. Obviously, the goal of materials research on electrochemical energy conversion and ...
... For electrolytic cells (electrolysers and charging batteries) the same sum of resistances applies, now only with the current having the opposite sign, whereby the overpotentials serve to increase the applied voltage. Obviously, the goal of materials research on electrochemical energy conversion and ...
Equilibrium Constant
... This specific reaction simplifies matters to the point that we can consider the activities of the products when determining Ksp. We would call this the activity product [AP] or ion activity product if only charged species are involved [IAP]. If the [AP] = Ksp, the system is in equilibrium,. If the [ ...
... This specific reaction simplifies matters to the point that we can consider the activities of the products when determining Ksp. We would call this the activity product [AP] or ion activity product if only charged species are involved [IAP]. If the [AP] = Ksp, the system is in equilibrium,. If the [ ...
AS 2, Organic, Physical and Inorganic Chemistry
... Suggest and explain how each of the following changes affects the position of the equilibrium and the rate of the reaction. ...
... Suggest and explain how each of the following changes affects the position of the equilibrium and the rate of the reaction. ...
Chemistry MSL Practical Style Review 1. What is the nuclear
... 17. In a titration experiment, if 30.0 mL of an HCl solution reacts with 24.6 mL of a 0.50-M NaOH solution, what is the concentration of the HCl solution? A B C D ...
... 17. In a titration experiment, if 30.0 mL of an HCl solution reacts with 24.6 mL of a 0.50-M NaOH solution, what is the concentration of the HCl solution? A B C D ...
Final Exam Practice Questions for General Chemistry NOTICE TO
... 16. What is the coefficient of H2 when the following equation is correctly balanced? Na(s) + H2O(l) → NaOH(aq) + H2 (g) a) 1 b) 2 c) 3 d) 4 e) 5 ...
... 16. What is the coefficient of H2 when the following equation is correctly balanced? Na(s) + H2O(l) → NaOH(aq) + H2 (g) a) 1 b) 2 c) 3 d) 4 e) 5 ...
112- Unit I -Electrochem -pdf
... The amount of electricity that must be supplied to a cell in order to deliver 1 mole of electrons is called a Faraday. 1 F = 96500 coulomb Ag + (aq) + 1 e- _____> 1mole=107.87 g 1 mole ...
... The amount of electricity that must be supplied to a cell in order to deliver 1 mole of electrons is called a Faraday. 1 F = 96500 coulomb Ag + (aq) + 1 e- _____> 1mole=107.87 g 1 mole ...
Electrochemical Fundamentals
... exception that it offers very high capacitance in a small package. Energy storage is by means of static charge rather than of an electrochemical process that is inherent to the battery. The concept is similar to an electrical charge that builds up when walking on a carpet. The supercapacitor concept ...
... exception that it offers very high capacitance in a small package. Energy storage is by means of static charge rather than of an electrochemical process that is inherent to the battery. The concept is similar to an electrical charge that builds up when walking on a carpet. The supercapacitor concept ...
phase diagrams and IMF
... 1.) Explain why the formation of solid and/or liquid phase(s) would be favored under conditions of: Explain using the ideas of kinetic energy (molecular motion), how close together molecules are/could be, ability to be attracted to its neighbor: ...
... 1.) Explain why the formation of solid and/or liquid phase(s) would be favored under conditions of: Explain using the ideas of kinetic energy (molecular motion), how close together molecules are/could be, ability to be attracted to its neighbor: ...
June 2010 Regents Exam Part C Questions
... Q24 Which statement describes one characteristic of an operating electrolytic cell? (1) It produces electrical energy. (2) It requires an external energy source. (3) It uses radioactive nuclides. (4) It undergoes a spontaneous redox reaction. Q25 Which compound when dissolved in water is an Arrh ...
... Q24 Which statement describes one characteristic of an operating electrolytic cell? (1) It produces electrical energy. (2) It requires an external energy source. (3) It uses radioactive nuclides. (4) It undergoes a spontaneous redox reaction. Q25 Which compound when dissolved in water is an Arrh ...
CFE Higher Chemistry in Society Homework EB
... starch and potassium iodide solution. The paper changes colour when ozone is present. Ozone reacts with potassium iodide and water to form iodine, oxygen and potassium hydroxide. Write the balanced chemical equation for this reaction. ...
... starch and potassium iodide solution. The paper changes colour when ozone is present. Ozone reacts with potassium iodide and water to form iodine, oxygen and potassium hydroxide. Write the balanced chemical equation for this reaction. ...
Aqueous chemistry is a very important component to laboratory
... The partially negative oxygen atom in water will surround the positive cations, while the partially positive hydrogen atoms will surround the negative anions. If two electrodes (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solut ...
... The partially negative oxygen atom in water will surround the positive cations, while the partially positive hydrogen atoms will surround the negative anions. If two electrodes (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solut ...
Balancing RedOx reactions handout
... 1. The oxidation number of an element in it’s elemental state is always zero. (Na, H 2, Li, O2 , etc.) 2. The oxidation number of a monatomic ion is equal to it’s charge. 3. If a group of atoms is ionic, the individual oxidation numbers must add up to its charge. (CO32-, NO3- , etc.) 4. The oxidatio ...
... 1. The oxidation number of an element in it’s elemental state is always zero. (Na, H 2, Li, O2 , etc.) 2. The oxidation number of a monatomic ion is equal to it’s charge. 3. If a group of atoms is ionic, the individual oxidation numbers must add up to its charge. (CO32-, NO3- , etc.) 4. The oxidatio ...
Chemistry - Halifax County Public Schools
... Dr. S. Stuart Flanagan, Professor Emeritus College of William and Mary Copyright 2005, S. Stuart Flanagan and David E. W. Mott Do not reproduce without permission. ...
... Dr. S. Stuart Flanagan, Professor Emeritus College of William and Mary Copyright 2005, S. Stuart Flanagan and David E. W. Mott Do not reproduce without permission. ...
Chemistry 40S – Exam Review
... d) CaO + SO3 CaSO4 10. What is the oxidation number for the Mn in MnO4- ? 11. What is the coefficient for ClO4- when the following oxidation-reduction equation is balanced? Au(s) + ClO4- + H+ ClO2+ + Au3+ + H2O 12. Describe how redox reactions generate an electric current. 13. Diagram and label ...
... d) CaO + SO3 CaSO4 10. What is the oxidation number for the Mn in MnO4- ? 11. What is the coefficient for ClO4- when the following oxidation-reduction equation is balanced? Au(s) + ClO4- + H+ ClO2+ + Au3+ + H2O 12. Describe how redox reactions generate an electric current. 13. Diagram and label ...
Second Year Chemistry
... current of 0.682 A from a 12.0 V supply is passed for 500 s through a heating coil immersed in the boiling liquid, it is found that the temperature remains constant but 4.33 g of ethanol is vapourised. What is the enthalpy of vapourisation of ethanol at its boiling point at 1 atm? Calculate the st ...
... current of 0.682 A from a 12.0 V supply is passed for 500 s through a heating coil immersed in the boiling liquid, it is found that the temperature remains constant but 4.33 g of ethanol is vapourised. What is the enthalpy of vapourisation of ethanol at its boiling point at 1 atm? Calculate the st ...
Chem 171 Review Exam 2
... CH4 (g) + NH3 (g) HCN (g) + 3 H2 (g) What is ∆H of this reaction at constant pressure? Use the following thermochemical equations: N2 (g) + 3 H2 (g) 2 NH3 (g) ΔH = —91.8 kJ C (graphite) + 2 H2 (g) CH4 (g) ΔH = —74.9 kJ H2 (g) + 2 C (graphite) + N2 (g) 2 HCN (g) ΔH = 270.3 kJ ...
... CH4 (g) + NH3 (g) HCN (g) + 3 H2 (g) What is ∆H of this reaction at constant pressure? Use the following thermochemical equations: N2 (g) + 3 H2 (g) 2 NH3 (g) ΔH = —91.8 kJ C (graphite) + 2 H2 (g) CH4 (g) ΔH = —74.9 kJ H2 (g) + 2 C (graphite) + N2 (g) 2 HCN (g) ΔH = 270.3 kJ ...
2009 Chemistry Midterm Review Packet
... ionic crystal. After entering the solution, the dissolved ion is surrounded completely by water molecules, which tends to prevent the ion from reentering the crystal. 5. saturated 6. unsaturated 7. variable 11. An increase in temperature means an increase in the average kinetic energy. In a warmer s ...
... ionic crystal. After entering the solution, the dissolved ion is surrounded completely by water molecules, which tends to prevent the ion from reentering the crystal. 5. saturated 6. unsaturated 7. variable 11. An increase in temperature means an increase in the average kinetic energy. In a warmer s ...
Balancing Chemical Reactions
... 1.) The formulas of the reactants and products cannot be changed, do not alter subscripts or charges. 2.) The only numbers that can be changed are the numbers indicating how many molecules or atoms, which are called coefficients. 3.) A coefficient is assumed to be one if there is not a number in fro ...
... 1.) The formulas of the reactants and products cannot be changed, do not alter subscripts or charges. 2.) The only numbers that can be changed are the numbers indicating how many molecules or atoms, which are called coefficients. 3.) A coefficient is assumed to be one if there is not a number in fro ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.