Name
... 27. What is the solubility of sodium nitrate at 30oC? 28. A saturated solution of potassium nitrate is formed from 100 g of water. If the saturated solution is cooled from 80oC to 50oC, how many grams of precipitate are formed? ...
... 27. What is the solubility of sodium nitrate at 30oC? 28. A saturated solution of potassium nitrate is formed from 100 g of water. If the saturated solution is cooled from 80oC to 50oC, how many grams of precipitate are formed? ...
Chapter 19 CHEMICAL THERMODYNAMICS 19.1 SPONTANEOUS
... Stotal = Ssystem + Ssurr = 22.0 - 19.4 = 2.6 J/K The process is non spontaneous. If the temperature of the surroundings were not 310 K but rather some temperature infinitesimally above 273 K, the melting would be reversible instead of irreversible. In that case the entropy change of the surroundi ...
... Stotal = Ssystem + Ssurr = 22.0 - 19.4 = 2.6 J/K The process is non spontaneous. If the temperature of the surroundings were not 310 K but rather some temperature infinitesimally above 273 K, the melting would be reversible instead of irreversible. In that case the entropy change of the surroundi ...
Name - TeacherWeb
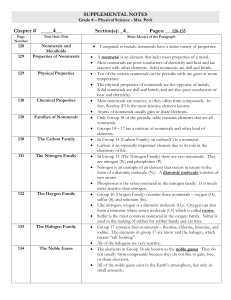
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
Redox Balancing Worksheet
... definition of redox reactions, then, involves the gain and loss of electrons rather than the gain and loss of oxygen. In the reaction below, for example, sodium metal (Na) reacts with chlorine gas (Cl 2 ) in such a way that sodium atoms lose one electron each to chlorine atoms: 2 Na + Cl 2 → 2 NaCl ...
... definition of redox reactions, then, involves the gain and loss of electrons rather than the gain and loss of oxygen. In the reaction below, for example, sodium metal (Na) reacts with chlorine gas (Cl 2 ) in such a way that sodium atoms lose one electron each to chlorine atoms: 2 Na + Cl 2 → 2 NaCl ...
Answers to examination questions
... B is trigonal planar (bond angles 120°); A, C and D are based upon a tetrahedral arrangement with four regions of high electron density. However, the water molecule has two lone pairs around the central atom, compared with one for ammonia and none for methane. Lone pair repulsion is greater than bon ...
... B is trigonal planar (bond angles 120°); A, C and D are based upon a tetrahedral arrangement with four regions of high electron density. However, the water molecule has two lone pairs around the central atom, compared with one for ammonia and none for methane. Lone pair repulsion is greater than bon ...
Ch. 11-12 Supplements
... If 30.0 grams of sulfuric acid and 25.0 grams of aluminum hydroxide react… b. How many grams of each product will be formed? c. How many grams of excess reactant remain? 4) a. Write the balanced equation for lithium metal, Li, reacting with aqueous copper (II) nitrate, Cu(NO3)2, to produce copper me ...
... If 30.0 grams of sulfuric acid and 25.0 grams of aluminum hydroxide react… b. How many grams of each product will be formed? c. How many grams of excess reactant remain? 4) a. Write the balanced equation for lithium metal, Li, reacting with aqueous copper (II) nitrate, Cu(NO3)2, to produce copper me ...
g moles molarity
... What happens when a solution of Na2CO3 is mixed with a solution of CaCl2? First assume everbody goes into solution or already in solution Identify who is charge dense and who is not charge dense ...
... What happens when a solution of Na2CO3 is mixed with a solution of CaCl2? First assume everbody goes into solution or already in solution Identify who is charge dense and who is not charge dense ...
SOLUBILITY RULES FOR IONIC COMPOUNDS IN WATER
... (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal is added to a solution of silver nitrate. (e) Solutions of le ...
... (a) An acidified solution of hydrogen peroxide is added to a solution of sodium iodide. (b) Chlorine gas is passed over powdered aluminum. (c) Solutons of mercury (I) nitrate and potassium sulfate are mixed. (d) A strip of magnesium metal is added to a solution of silver nitrate. (e) Solutions of le ...
1 - New Age International
... 7. Which of the following statements rigorously defines the ‘‘mol’’ of any substance? (a) It is the number of atoms in one gram atom of any substance (b) It refers to the number of molecules in a gram molecule of any substances (c) It refers to the number of ions in a gram ion of any ionic species. ...
... 7. Which of the following statements rigorously defines the ‘‘mol’’ of any substance? (a) It is the number of atoms in one gram atom of any substance (b) It refers to the number of molecules in a gram molecule of any substances (c) It refers to the number of ions in a gram ion of any ionic species. ...
remaster unit 8A + 7
... Why do some ionic compounds conduct better? • More ions per volume of solution (concentration) means more charge is carried (better conduction) • Ionic compounds that create more ions (higher concentration) when dissolved conduct electricity better ...
... Why do some ionic compounds conduct better? • More ions per volume of solution (concentration) means more charge is carried (better conduction) • Ionic compounds that create more ions (higher concentration) when dissolved conduct electricity better ...
practice problems of chap4_5 - Chemistry
... (a) SF4 (b) SF6 (c) S2F10 (d) S4F16 (e) S5F20 Hint: p. 211 (5.50) or p. 187. Equation (5.12). Example 5.9. In order to get the mole cular formula, we need to get the molar mass of the compound first. M=dRT/P=mRT/VP =0.100g*0.082 L• atm / (mol • K)*(20+273.15)K/[(22.0/1000)L*1.02atm]=107.1g/mole. The ...
... (a) SF4 (b) SF6 (c) S2F10 (d) S4F16 (e) S5F20 Hint: p. 211 (5.50) or p. 187. Equation (5.12). Example 5.9. In order to get the mole cular formula, we need to get the molar mass of the compound first. M=dRT/P=mRT/VP =0.100g*0.082 L• atm / (mol • K)*(20+273.15)K/[(22.0/1000)L*1.02atm]=107.1g/mole. The ...
Moles and Equations
... Up to this point state symbols have not been used in any of the equations: (s) ...
... Up to this point state symbols have not been used in any of the equations: (s) ...
on line measurement of oxygen
... TDLs are currently mostly used in the near infrared (IR) (< 2.5 microns) region of the electromagnetic spectrum. The availability of these tunable lasers operating at around room temperatures derives from their usefulness in the telecommunications industry. Near IR absorptions are usually overtones ...
... TDLs are currently mostly used in the near infrared (IR) (< 2.5 microns) region of the electromagnetic spectrum. The availability of these tunable lasers operating at around room temperatures derives from their usefulness in the telecommunications industry. Near IR absorptions are usually overtones ...
chapters 16-17 test re
... 3. _______ A low Ea means that relatively few collisions will have the required energy to produce the activated complex, and the reaction rate is fast. 4. _______ Catalysts are enzymes that aren’t consumed in a chemical reaction, but they raise the reaction rate by lowering the Ea. 5. _______ To cal ...
... 3. _______ A low Ea means that relatively few collisions will have the required energy to produce the activated complex, and the reaction rate is fast. 4. _______ Catalysts are enzymes that aren’t consumed in a chemical reaction, but they raise the reaction rate by lowering the Ea. 5. _______ To cal ...
3.1 - Weathering Define mechanical and chemical weathering
... of mechanical weathering, by digging or blasting intorock to build homes, roads, subways, or to quarry stone. ...
... of mechanical weathering, by digging or blasting intorock to build homes, roads, subways, or to quarry stone. ...
Renal Physiology 9 (Acid Base 1)
... solution will have more free H3O+ ions and therefore a lower pH. ...
... solution will have more free H3O+ ions and therefore a lower pH. ...
elements of chemistry unit
... hydrogen atoms. This means oxygen has 2 “extra” electrons that give it a – 2 oxidation number. Each hydrogen in the water molecule has a single electron pulled away from it. Therefore, both hydrogen atoms have a +1 oxidation number. PURE ELEMENTS All pure elements, for example H2 and Cl2, are assign ...
... hydrogen atoms. This means oxygen has 2 “extra” electrons that give it a – 2 oxidation number. Each hydrogen in the water molecule has a single electron pulled away from it. Therefore, both hydrogen atoms have a +1 oxidation number. PURE ELEMENTS All pure elements, for example H2 and Cl2, are assign ...
Chemistry 12 - hrsbstaff.ednet.ns.ca
... 40. What effect does a catalyst have? A. increases the reaction rate by decreasing the heat of reaction B. increases the reaction rate by increasing the activation energy of the reverse reaction C. increases the reaction rate by lowering the activation energy of the forward reaction only D. increase ...
... 40. What effect does a catalyst have? A. increases the reaction rate by decreasing the heat of reaction B. increases the reaction rate by increasing the activation energy of the reverse reaction C. increases the reaction rate by lowering the activation energy of the forward reaction only D. increase ...
Describing Chemical Reactions
... produce water. The reverse of this reaction can also occur. In other words, water can be broken down to make hydrogen gas and oxygen gas. The breakdown of water is a decomposition reaction. The unbalanced equation for this reaction is shown below. H 2O H2 ...
... produce water. The reverse of this reaction can also occur. In other words, water can be broken down to make hydrogen gas and oxygen gas. The breakdown of water is a decomposition reaction. The unbalanced equation for this reaction is shown below. H 2O H2 ...
Chapter 3 Chemical Reactions
... achieved, the amount of each reactant and product remains constant. ...
... achieved, the amount of each reactant and product remains constant. ...
Name chemistry Unit 8 worksheet 1. Why do
... 12. What is meant by the term reaction rate? The change in the amount of products or reactants in a given amount of time 13. For each of the following pairs, choose the substance or process you would expect to react more rapidly. a. Granulated sugar or powdered sugar b. Zinc in HCl at 298 K or zinc ...
... 12. What is meant by the term reaction rate? The change in the amount of products or reactants in a given amount of time 13. For each of the following pairs, choose the substance or process you would expect to react more rapidly. a. Granulated sugar or powdered sugar b. Zinc in HCl at 298 K or zinc ...
sec chemistry may 2011 marking scheme
... • different sized molecules / different RMM / more or less (carbon) atoms (in molecules) / different structures. Ignore different densities. • fuels have different boiling points • fuels condense at different temperatures. Cracking is the process where • large hydrocarbon molecules are broken into s ...
... • different sized molecules / different RMM / more or less (carbon) atoms (in molecules) / different structures. Ignore different densities. • fuels have different boiling points • fuels condense at different temperatures. Cracking is the process where • large hydrocarbon molecules are broken into s ...
1. Natures Chemistry Unit Questions
... (ii) A barrel containing 195 litres of cask strength whisky costs £1300. The cask strength whisky is diluted with water to reduce the concentration of alcohol from 65% abv to 46% abv before it is bottled. Calculate the cost of the cask strength whisky needed to produce a 0·70 litre bottle. Show you ...
... (ii) A barrel containing 195 litres of cask strength whisky costs £1300. The cask strength whisky is diluted with water to reduce the concentration of alcohol from 65% abv to 46% abv before it is bottled. Calculate the cost of the cask strength whisky needed to produce a 0·70 litre bottle. Show you ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.