Write this into your supplemental packet opposite page
... 5. Predict the transition metal cation charge for iron, Fe, in the ionic salt Fe 2 (SO4 )3 , and place it in the cation box below. 6. Give a name for Fe 2 (SO4 )3 . Since transition metals can variable charge, you must some how indicate metal cation charge in its name. ...
... 5. Predict the transition metal cation charge for iron, Fe, in the ionic salt Fe 2 (SO4 )3 , and place it in the cation box below. 6. Give a name for Fe 2 (SO4 )3 . Since transition metals can variable charge, you must some how indicate metal cation charge in its name. ...
PHYSICAL SETTING CHEMISTRY
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
... A separate answer sheet for Part A and Part B–1 has been provided to you. Follow the instructions from the proctor for completing the student information on your answer sheet. Record your answers to the Part A and Part B–1 multiple-choice questions on this separate answer sheet. Record your answers ...
IChO 35 Theoretical Exam
... evaporates, but the trapped molecules push against the lid, hence lowering the measurement of the balance by m. Therefore, the force exerted on the lid is f = m g. The force is also equal to the rate of change of the momentum of the evaporating molecules, i.e., f = ½ u dm/dt. Using the data provid ...
... evaporates, but the trapped molecules push against the lid, hence lowering the measurement of the balance by m. Therefore, the force exerted on the lid is f = m g. The force is also equal to the rate of change of the momentum of the evaporating molecules, i.e., f = ½ u dm/dt. Using the data provid ...
Chemistry Summer Work (30 questions):
... Question #6. In every Periodic Table, even the most basic, one can find two numbers associated with each type of element. For example, for zinc (Zn) those numbers are 30 and 65.409. Explain what these numbers represent. Give units if the numbers have units. Question #7. 55Fe is a radioactive isotop ...
... Question #6. In every Periodic Table, even the most basic, one can find two numbers associated with each type of element. For example, for zinc (Zn) those numbers are 30 and 65.409. Explain what these numbers represent. Give units if the numbers have units. Question #7. 55Fe is a radioactive isotop ...
File - Meissnerscience.com
... How many molecules of oxygen are required to generate 100 water molecules? ...
... How many molecules of oxygen are required to generate 100 water molecules? ...
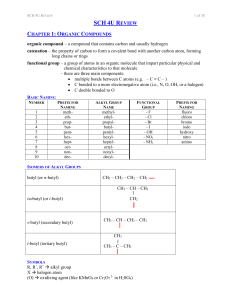
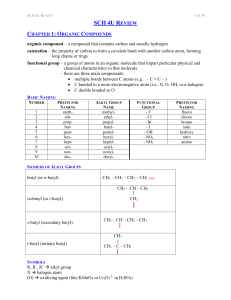
SCH 4U REVIEW Notes
... less polar than carboxylic acids (loss of OH- group) dispersion forces medium (lower than carboxylic acids, higher than aldehydes / ketones of similar length due to extra O) less soluble than acids condensation (formation) carboxylic acid + alcohol ester + water ethanoic acid + methanol ...
... less polar than carboxylic acids (loss of OH- group) dispersion forces medium (lower than carboxylic acids, higher than aldehydes / ketones of similar length due to extra O) less soluble than acids condensation (formation) carboxylic acid + alcohol ester + water ethanoic acid + methanol ...
Biol 1406 notes Ch 2 8thed
... Ionic compounds can have ratios of elements different from 1:1. o For example, the ionic compound magnesium chloride (MgCl2) has two chloride atoms per magnesium atom. o Magnesium needs to lose 2 electrons to drop to a full outer shell; each chlorine atom needs to gain 1 electron. Entire molecul ...
... Ionic compounds can have ratios of elements different from 1:1. o For example, the ionic compound magnesium chloride (MgCl2) has two chloride atoms per magnesium atom. o Magnesium needs to lose 2 electrons to drop to a full outer shell; each chlorine atom needs to gain 1 electron. Entire molecul ...
File
... Nuclear Fission - People are scared of nuclear energy, so as we know it, it will be impossible to get people to accept it! Nuclear Fusion - A form of nuclear energy that has tremendous potential, but at the current time cannot be controlled safely. The internal confinement and laser fusion methods b ...
... Nuclear Fission - People are scared of nuclear energy, so as we know it, it will be impossible to get people to accept it! Nuclear Fusion - A form of nuclear energy that has tremendous potential, but at the current time cannot be controlled safely. The internal confinement and laser fusion methods b ...
Charging of Oil-Water Interfaces Due to Spontaneous Adsorption of
... coalescence of the emulsion droplets. In order to increase the reliability of the data, two alternative procedures for emulsion preparation were used. The first procedure was close to the standard method for preparation of surfactant containing emulsions. The purified and presaturated liquid phases ...
... coalescence of the emulsion droplets. In order to increase the reliability of the data, two alternative procedures for emulsion preparation were used. The first procedure was close to the standard method for preparation of surfactant containing emulsions. The purified and presaturated liquid phases ...
FREE Sample Here
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
... Ans: Ethanol can form hydrogen bonds with water molecules, but ethane cannot. When ethanol dissolves, the decrease in the system's entropy that results from formation of ordered arrays of water around the CH3CH2– group is partly compensated by the favorable interactions (hydrogen bonds) of the hydro ...
Compounds of Chlorine
... Hydrogen chloride (HCl) is prepared by the reaction of concentrated sulfuric acid (H2 SO4 ) with either NaCl or concentrated HCl solution. Hydrogen chloride is a polar molecule with a dipole of 1.08 D. However, the lower polarity as compared to that of hydrogen uoride (1.91 D) is consistent with th ...
... Hydrogen chloride (HCl) is prepared by the reaction of concentrated sulfuric acid (H2 SO4 ) with either NaCl or concentrated HCl solution. Hydrogen chloride is a polar molecule with a dipole of 1.08 D. However, the lower polarity as compared to that of hydrogen uoride (1.91 D) is consistent with th ...
Test3_sp2012with answers
... D) it increases or decreases depending on how large a temperature change occurs _D__13. Which of the following influence the vapor pressure of liquids? A) polarity of liquid molecules B) mass of liquid molecules C) temperature of liquid D) more than one response is correct __B_14. Which of the follo ...
... D) it increases or decreases depending on how large a temperature change occurs _D__13. Which of the following influence the vapor pressure of liquids? A) polarity of liquid molecules B) mass of liquid molecules C) temperature of liquid D) more than one response is correct __B_14. Which of the follo ...
Briefing Session on 2012 HKDSE Examination (December 2012)
... Deduce the order of reaction with respect to NO(g) and that to H2(g). (2 marks, well answered) ✔ Many candidates were able to use ‘deduction method’ or an ‘arithmetic approach’ to find the order required ...
... Deduce the order of reaction with respect to NO(g) and that to H2(g). (2 marks, well answered) ✔ Many candidates were able to use ‘deduction method’ or an ‘arithmetic approach’ to find the order required ...
Aqueous Solutions
... Figure 4.3: (a) The ethanol molecule contains a polar O—H bond similar to those in the water molecule. (b) The polar water molecule interacts strongly with the polar O—H bond in ethanol. This is a case of "like dissolving like." ...
... Figure 4.3: (a) The ethanol molecule contains a polar O—H bond similar to those in the water molecule. (b) The polar water molecule interacts strongly with the polar O—H bond in ethanol. This is a case of "like dissolving like." ...
Open questions (66 points total
... (NOTE There are 2 NMR spectra with this problem. Below the 1H spectrum, the integrals (= areas) of the signals are given as numbers ratios). From the IR spectrum of an unknown substance X with M = 102, we know X to be an ester. 6p 1 Calculate the molecular formula of substance X. Give all possible ...
... (NOTE There are 2 NMR spectra with this problem. Below the 1H spectrum, the integrals (= areas) of the signals are given as numbers ratios). From the IR spectrum of an unknown substance X with M = 102, we know X to be an ester. 6p 1 Calculate the molecular formula of substance X. Give all possible ...
Stoichiometry
... • A 2.00 g sample of ammonia is mixed with 4.00 g of oxygen. Which is the limiting reactant and how much excess reactant remains after the reaction has stopped? • First, we need to create a balanced equation for the reaction: 4 NH3(g) + 5 O2(g)4 NO(g) + 6 H2O(g) • Next we can use stoichiometry to ca ...
... • A 2.00 g sample of ammonia is mixed with 4.00 g of oxygen. Which is the limiting reactant and how much excess reactant remains after the reaction has stopped? • First, we need to create a balanced equation for the reaction: 4 NH3(g) + 5 O2(g)4 NO(g) + 6 H2O(g) • Next we can use stoichiometry to ca ...
Technical Data Sheet (E
... The zinc phosphate coating remains fine-grained regardless of the cleaning method used prior to application. It can be applied by either immersion or spray. E-Phos 660 is an excellent paint base. The fine-grained, low porosity coating reduces paint consumption by reducing the amount of paint absorbe ...
... The zinc phosphate coating remains fine-grained regardless of the cleaning method used prior to application. It can be applied by either immersion or spray. E-Phos 660 is an excellent paint base. The fine-grained, low porosity coating reduces paint consumption by reducing the amount of paint absorbe ...
1 [Turn Over Section A For each question there are four possible
... When iron filings are added to nitric acid, a yellow solution and nitrogen dioxide gas are formed. On the addition of ammonium thiocyanate to the resultant solution, a bloodred colouration due to an iron (III) complex is formed. Which statements are correct? ...
... When iron filings are added to nitric acid, a yellow solution and nitrogen dioxide gas are formed. On the addition of ammonium thiocyanate to the resultant solution, a bloodred colouration due to an iron (III) complex is formed. Which statements are correct? ...
Part II - American Chemical Society
... order and illustrate how the reaction conditions above would be changed so that the [I–] would be pseudo first order. e. The activation energy for this reaction was found to be 84 kJ·mol –1 at 25 °C. How much faster would this reaction proceed if the activation energy were lowered by 10 kJ·mol–1 (fo ...
... order and illustrate how the reaction conditions above would be changed so that the [I–] would be pseudo first order. e. The activation energy for this reaction was found to be 84 kJ·mol –1 at 25 °C. How much faster would this reaction proceed if the activation energy were lowered by 10 kJ·mol–1 (fo ...
Experiment 15: Reduction and Oxidation of Organic Compounds
... solutions. The oxidizing agent used in this procedure is hypochlorous acid, the active component of household bleach. Although chromic acid, H2CrO4, is a more commonly used reagent for the oxidation of alcohols, it is a suspected carcinogen and generates hazardous waste. Replacing a harmful and poll ...
... solutions. The oxidizing agent used in this procedure is hypochlorous acid, the active component of household bleach. Although chromic acid, H2CrO4, is a more commonly used reagent for the oxidation of alcohols, it is a suspected carcinogen and generates hazardous waste. Replacing a harmful and poll ...
Energy and Energy Changes Heat Transfer and The Measurement
... Therefore the metal cannot be pure gold. ...
... Therefore the metal cannot be pure gold. ...
Questions - Scheikundeolympiade
... evaporates, but the trapped molecules push against the lid, hence lowering the measurement of the balance by m. Therefore, the force exerted on the lid is f = m g. The force is also equal to the rate of change of the momentum of the evaporating molecules, i.e., f = ½ u dm/dt. Using the data provid ...
... evaporates, but the trapped molecules push against the lid, hence lowering the measurement of the balance by m. Therefore, the force exerted on the lid is f = m g. The force is also equal to the rate of change of the momentum of the evaporating molecules, i.e., f = ½ u dm/dt. Using the data provid ...
Chapter 8 Chemical Equations and Reactions
... Indications of a Chemical Reaction Certain easily observed changes usually indicate that a chemical reaction has occurred. i) Evolution of energy as heat and light ii) Production of a gas iii) Formation of a precipitate A solid that is produced as a result of a chemical reaction in solution and tha ...
... Indications of a Chemical Reaction Certain easily observed changes usually indicate that a chemical reaction has occurred. i) Evolution of energy as heat and light ii) Production of a gas iii) Formation of a precipitate A solid that is produced as a result of a chemical reaction in solution and tha ...
sch4ureview
... less polar than carboxylic acids (loss of OH- group) dispersion forces medium (lower than carboxylic acids, higher than aldehydes / ketones of similar length due to extra O) less soluble than acids condensation (formation) carboxylic acid + alcohol ester + water ethanoic acid + methanol ...
... less polar than carboxylic acids (loss of OH- group) dispersion forces medium (lower than carboxylic acids, higher than aldehydes / ketones of similar length due to extra O) less soluble than acids condensation (formation) carboxylic acid + alcohol ester + water ethanoic acid + methanol ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.