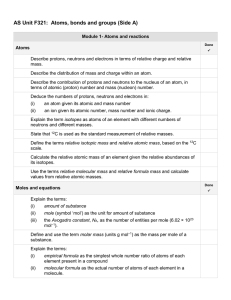
AS Unit F321 Unit 1 Side A check list
... Explain that an acid releases H+ ions in aqueous solution. State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. Stat ...
... Explain that an acid releases H+ ions in aqueous solution. State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. Stat ...
lect7_conductivity_meter
... In this last figure we get a better idea of how this instrument can function as either a conductivity meter or an ion counter and how it transitions from one to the other. For a given rate of air flow through the cylindrical capacitor we monitor the signal current as the potential difference betwee ...
... In this last figure we get a better idea of how this instrument can function as either a conductivity meter or an ion counter and how it transitions from one to the other. For a given rate of air flow through the cylindrical capacitor we monitor the signal current as the potential difference betwee ...
Chapter 4 Outline
... Ch4.1: I can apply the solubility rule to solutions Ch4.2: I can illustrate dissociation correctly with equations and particle diagrams. Ch4.3: I can differentiate between a strong, weak and non electrolyte, and predict which type a substance would be. Ch4.4: I can do calculations with molarity of s ...
... Ch4.1: I can apply the solubility rule to solutions Ch4.2: I can illustrate dissociation correctly with equations and particle diagrams. Ch4.3: I can differentiate between a strong, weak and non electrolyte, and predict which type a substance would be. Ch4.4: I can do calculations with molarity of s ...
Types of Reactions and Solution Chemistry
... state? Notice, we have formed a solid. That means that ions that were once dissolved in solution came together and made a solid, or a precipitate. The ionic equation will help us determine what is going on in solution. Since our two reactants are aqueous that means that they are dissolved in water. ...
... state? Notice, we have formed a solid. That means that ions that were once dissolved in solution came together and made a solid, or a precipitate. The ionic equation will help us determine what is going on in solution. Since our two reactants are aqueous that means that they are dissolved in water. ...
Copy of Acids, bases, salts answer key
... o If the pH paper turns blue, it means the approximate pH value of the solution is 10 and it is basic. B. Through universal indicator : o Universal indicator is a mixture of many different indicators or say dyes which when added to a solution, change in the colour of the solution and thus indicate i ...
... o If the pH paper turns blue, it means the approximate pH value of the solution is 10 and it is basic. B. Through universal indicator : o Universal indicator is a mixture of many different indicators or say dyes which when added to a solution, change in the colour of the solution and thus indicate i ...
CHEMISTRY NOTES – CHAPTERS 20 AND 21
... The terms concentrated and dilute should not be confused with the terms strong and weak in relation to acids and bases. Concentrated and dilute refer to how much acid or bases is present, not the extent to which that acid or base will ionize. The following diagrams should illustrate the differences. ...
... The terms concentrated and dilute should not be confused with the terms strong and weak in relation to acids and bases. Concentrated and dilute refer to how much acid or bases is present, not the extent to which that acid or base will ionize. The following diagrams should illustrate the differences. ...
pH scale. Buffer solutions. Colligative properties of solutions
... mucosa. By a process known as active transport, H+ ions move across the membrane into the stomach interior. (Active transport processes are aided by enzymes.) To maintain electrical balance, an equal number of Cl– ions also move from the blood plasma into the stomach. Once in the stomach, most of th ...
... mucosa. By a process known as active transport, H+ ions move across the membrane into the stomach interior. (Active transport processes are aided by enzymes.) To maintain electrical balance, an equal number of Cl– ions also move from the blood plasma into the stomach. Once in the stomach, most of th ...
Unit 9 – Behavior of Gases
... 30. Write how the following ionic compounds will dissociate in water. For each compound, what is the total number of ions represented in the formula? a. CaCl2 b. CaCO3 c. Mg3(PO4)2 31. Write the net ionic equation for the reaction between calcium chloride and potassium carbonate. 32. What is the mol ...
... 30. Write how the following ionic compounds will dissociate in water. For each compound, what is the total number of ions represented in the formula? a. CaCl2 b. CaCO3 c. Mg3(PO4)2 31. Write the net ionic equation for the reaction between calcium chloride and potassium carbonate. 32. What is the mol ...
Mr. Dehne AP Chem Name: ___________ Date: Per#: ___ AP
... 34. Saccharin (C7H5NO3S) is sometimes dispensed in tablet form. Ten tablets with a total mass of 0.5894g were dissolved in water. They were oxidized to convert all of the sulfur to sulfate ion, which was precipitated by adding an excess of barium chloride solution. The mass of BaSO4 obtained was 0.5 ...
... 34. Saccharin (C7H5NO3S) is sometimes dispensed in tablet form. Ten tablets with a total mass of 0.5894g were dissolved in water. They were oxidized to convert all of the sulfur to sulfate ion, which was precipitated by adding an excess of barium chloride solution. The mass of BaSO4 obtained was 0.5 ...
Chapter 13
... called non-ideal solutions because they deviate from the expected behavior. • Very few solutions actually approach ideality, but Raoult's law for the ideal solution is a good enough approximation for the non- ideal solutions ...
... called non-ideal solutions because they deviate from the expected behavior. • Very few solutions actually approach ideality, but Raoult's law for the ideal solution is a good enough approximation for the non- ideal solutions ...
water, h2o
... unique mode of transport in water and, by extension, in other highly connected hydrogen bonding systems. The Grotthuss mechanism involves a simple shift of hydrogen bonds to effectively relocate a net protonic charge from one position to another without significantly moving the mass of the proton. I ...
... unique mode of transport in water and, by extension, in other highly connected hydrogen bonding systems. The Grotthuss mechanism involves a simple shift of hydrogen bonds to effectively relocate a net protonic charge from one position to another without significantly moving the mass of the proton. I ...
Solution chemistry and reaction mechanism taking place during the
... In(OH)xSy contain InCl3, acetic acid and thioacetamide. As it was stated in a previous work [4], the chemical species present in the solution and the rate of thioacetamide hydrolysis, depend on the solution pH. Therefore, the pH of solutions containing thioacetamide, acetic acid and InCl3 was measur ...
... In(OH)xSy contain InCl3, acetic acid and thioacetamide. As it was stated in a previous work [4], the chemical species present in the solution and the rate of thioacetamide hydrolysis, depend on the solution pH. Therefore, the pH of solutions containing thioacetamide, acetic acid and InCl3 was measur ...
review sheet
... 14. Where will water boil at a lower temperature 250 feet or 10,000 feet above sea level? _____________________________________________ Why?_____________________ 15. Explain on the molecular level how solids, liquids and gases compare. Which has the most kinetic energy? ...
... 14. Where will water boil at a lower temperature 250 feet or 10,000 feet above sea level? _____________________________________________ Why?_____________________ 15. Explain on the molecular level how solids, liquids and gases compare. Which has the most kinetic energy? ...
Review Package
... 26) a) If apple juice has a pH of 5 and vinegar has a pH of 3, which is more acidic? __________________ b) How many more hydrogen ions are there in the more acidic substance? ____________________ 27) How much more acidic is a solution with a pH of 4.5 than a solution with a pH of a) 5.5? b) 6.5? 28) ...
... 26) a) If apple juice has a pH of 5 and vinegar has a pH of 3, which is more acidic? __________________ b) How many more hydrogen ions are there in the more acidic substance? ____________________ 27) How much more acidic is a solution with a pH of 4.5 than a solution with a pH of a) 5.5? b) 6.5? 28) ...
CHEMISTRY 1710 - Practice Exam #2 (KATZ)
... water bath at 99°C. The barometric pressure is 753 torr. If the mass of the liquid retained in the flask is 1.362 g, what is its molar mass? a. ...
... water bath at 99°C. The barometric pressure is 753 torr. If the mass of the liquid retained in the flask is 1.362 g, what is its molar mass? a. ...
Net ionic equation
... Reactions of acids and bases •Neutralization: acid + base are mixed: HNO3(aq) + KOH(aq) ??? •Salt = ionic compound cation from base anion from acid. •Neutralization of acid with metal hydroxide produces water and a salt. •Acids + carbonates = CO2 and H2O ...
... Reactions of acids and bases •Neutralization: acid + base are mixed: HNO3(aq) + KOH(aq) ??? •Salt = ionic compound cation from base anion from acid. •Neutralization of acid with metal hydroxide produces water and a salt. •Acids + carbonates = CO2 and H2O ...
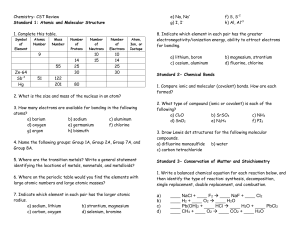
Chemistry- CST Review
... 10. A 500 mL air sample at a temperature of -50 °C has a pressure of 1.3 atm. What will be the new pressure if the temperature is raised to 102 °C and the volume expands to 700 mL? ...
... 10. A 500 mL air sample at a temperature of -50 °C has a pressure of 1.3 atm. What will be the new pressure if the temperature is raised to 102 °C and the volume expands to 700 mL? ...
Biology Fall Semester Test 1 Study Guide
... Isotopes are atoms of the same element with the same number of protons and If an atom contains 11 protons and 12 neutrons, its atomic number is Which of the following terms describes a substance formed by the combination of two or more elements in definite proportions? A covalent bond is formed as t ...
... Isotopes are atoms of the same element with the same number of protons and If an atom contains 11 protons and 12 neutrons, its atomic number is Which of the following terms describes a substance formed by the combination of two or more elements in definite proportions? A covalent bond is formed as t ...
PH

In chemistry, pH (/piːˈeɪtʃ/) is a numeric scale used to specify the acidity or alkalinity of an aqueous solution. It is the negative of the logarithm to base 10 of the activity of the hydrogen ion. Solutions with a pH less than 7 are acidic and solutions with a pH greater than 7 are alkaline or basic. Pure water is neutral, being neither an acid nor a base. Contrary to popular belief, the pH value can be less than 0 or greater than 14 for very strong acids and bases respectively.pH measurements are important in medicine, biology, chemistry, agriculture, forestry, food science, environmental science, oceanography, civil engineering, chemical engineering, nutrition, water treatment & water purification, and many other applications. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.Primary pH standard values are determined using a concentration cell with transference, by measuring the potential difference between a hydrogen electrode and a standard electrode such as the silver chloride electrode.The pH of aqueous solutions can be measured with a glass electrode and a pH meter, or indicator.pH is the negative of the logarithm to base 10 of the activity of the (solvated) hydronium ion, more often (albeit somewhat inaccurately) expressed as the measure of the hydronium ion concentration.The rest of this article uses the technically correct word ""base"" and its inflections in place of ""alkaline"", which specifically refers to a base dissolved in water, and its inflections.