Physical Science Standards
... 4.1 investigate the properties and behaviors of waves. 4.2 explore and explain the nature of sound and light energy. 4.3 examine the applications and effects of heat energy. 4.4 probe the fundamental principles and applications of electricity. 4.5 distinguish between nuclear fission and nuclear fusi ...
... 4.1 investigate the properties and behaviors of waves. 4.2 explore and explain the nature of sound and light energy. 4.3 examine the applications and effects of heat energy. 4.4 probe the fundamental principles and applications of electricity. 4.5 distinguish between nuclear fission and nuclear fusi ...
Solutions for Chapter 8 End-of-Chapter Problems
... solution with concentration of sucrose greater than 0.5% [process (ii)]. Some of the water inside the cell will move by osmosis through the semipermeable membrane into the solution that is more concentrated in sucrose. Since water leaves the cell, the concentration of the sucrose in the cell would i ...
... solution with concentration of sucrose greater than 0.5% [process (ii)]. Some of the water inside the cell will move by osmosis through the semipermeable membrane into the solution that is more concentrated in sucrose. Since water leaves the cell, the concentration of the sucrose in the cell would i ...
A thermodynamic model for the prediction of phase equilibria and
... sedimentary rocks. There have been many experimental work on the solubility of gypsum and anhydrite in aqueous solutions and their phase transitions under different temperatures and pressures (Hulett and Allen, 1902; Partridge and White, 1929; Hill, 1937; Booth and Bidwell, 1950; Madgin and Swales, 1 ...
... sedimentary rocks. There have been many experimental work on the solubility of gypsum and anhydrite in aqueous solutions and their phase transitions under different temperatures and pressures (Hulett and Allen, 1902; Partridge and White, 1929; Hill, 1937; Booth and Bidwell, 1950; Madgin and Swales, 1 ...
For viewing on a computer screen (colored
... 4.3.1 Carnot engines and Carnot cycles . . . . . . . . . . . . . . . 4.3.2 The equivalence of the Clausius and Kelvin–Planck statements 4.3.3 The efficiency of a Carnot engine . . . . . . . . . . . . . . . 4.3.4 Thermodynamic temperature . . . . . . . . . . . . . . . . . . 4.4 The Second Law for Rev ...
... 4.3.1 Carnot engines and Carnot cycles . . . . . . . . . . . . . . . 4.3.2 The equivalence of the Clausius and Kelvin–Planck statements 4.3.3 The efficiency of a Carnot engine . . . . . . . . . . . . . . . 4.3.4 Thermodynamic temperature . . . . . . . . . . . . . . . . . . 4.4 The Second Law for Rev ...
(Physics) Question Bank - North Maharashtra University
... 15 Prove that there are four points collinear with the centre of gravity of a compound pendulum about which its times of oscillations are equal, hence obtain the length of an equivalent simple pendulum. 16 A uniform bar of length 96 cm oscillates like a compound pendulum about the horizontal axis, p ...
... 15 Prove that there are four points collinear with the centre of gravity of a compound pendulum about which its times of oscillations are equal, hence obtain the length of an equivalent simple pendulum. 16 A uniform bar of length 96 cm oscillates like a compound pendulum about the horizontal axis, p ...
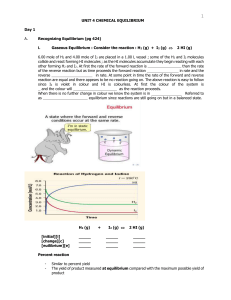
Unit 4 - Chemical Equilibrium
... _ _ _ _ _ _ _ _ _ _. This means the products can react together and turn back into the _ _ _ _ _ _ _ _ reactants. In other words, the reaction can go _ _ _ _ ways. When a reversible reaction is set up in a _ _ _ _ _ _ container, the forward reaction happens much faster than the reverse reaction at f ...
... _ _ _ _ _ _ _ _ _ _. This means the products can react together and turn back into the _ _ _ _ _ _ _ _ reactants. In other words, the reaction can go _ _ _ _ ways. When a reversible reaction is set up in a _ _ _ _ _ _ container, the forward reaction happens much faster than the reverse reaction at f ...
Ch 16 Power Point
... energy of the particles in a sample of matter. • The greater the kinetic energy of the particles in a sample, the hotter it feels. • For calculations in thermochemistry, the Celsius and Kelvin temperature scales are used. Celsius and Kelvin temperatures are related by the following equation. K = 273 ...
... energy of the particles in a sample of matter. • The greater the kinetic energy of the particles in a sample, the hotter it feels. • For calculations in thermochemistry, the Celsius and Kelvin temperature scales are used. Celsius and Kelvin temperatures are related by the following equation. K = 273 ...
Minimum Learning Competencies - Ministry of Education, Ethiopia
... Explain the MOT & describe molecular orbital using atomic orbital & bonding & anti-bonding orbital; Write the electron configuration of simple molecules using molecular orbital model; Define bond order of some simple molecules & their stability using bond order; Describe the Valence Shell Electron P ...
... Explain the MOT & describe molecular orbital using atomic orbital & bonding & anti-bonding orbital; Write the electron configuration of simple molecules using molecular orbital model; Define bond order of some simple molecules & their stability using bond order; Describe the Valence Shell Electron P ...
sch103manual - university of nairobi staff profiles
... any of the three states of matter: Solids, liquid or gas. Water for example, exists in the solid state as ice, liquid state as water and in the gaseous state as steam. The physical properties of a substance often depend on the state of the substance. In this section, we will review the states of mat ...
... any of the three states of matter: Solids, liquid or gas. Water for example, exists in the solid state as ice, liquid state as water and in the gaseous state as steam. The physical properties of a substance often depend on the state of the substance. In this section, we will review the states of mat ...
Fulltext: english,
... ii) The behaviour of the ligand in solution. Hardly any attempts have been made to establish it. It is currently assumed that the monomer is predominant in solution iii) The composition of the complex needs to be established particularly when dealing with calixarene receptors where it is often found ...
... ii) The behaviour of the ligand in solution. Hardly any attempts have been made to establish it. It is currently assumed that the monomer is predominant in solution iii) The composition of the complex needs to be established particularly when dealing with calixarene receptors where it is often found ...
Herbert Ipser, Olga P. Semenova, Regina
... = 15.189(2) Å, b = 8.799(1) Å, c = 13.602(2) Å and = 123.83(1)°. For the entire data set of 3706 independent reflections residual values are R = 0.0461 and Rw = 0.0789. The structure was found to be isotypic to Pb9Pd13 with In and Sb on the Pb-sites. The existence of a further ternary compound, wh ...
... = 15.189(2) Å, b = 8.799(1) Å, c = 13.602(2) Å and = 123.83(1)°. For the entire data set of 3706 independent reflections residual values are R = 0.0461 and Rw = 0.0789. The structure was found to be isotypic to Pb9Pd13 with In and Sb on the Pb-sites. The existence of a further ternary compound, wh ...
Energy Changes in Chemical Reactions
... In each case, there is energy involved in the change. Energy (in the form of heat) must be supplied to melt ice, whereas energy (in the form of heat and light) is produced by the explosive combination of hydrogen and oxygen gases. In fact, every change that matter undergoes is accompanied by either ...
... In each case, there is energy involved in the change. Energy (in the form of heat) must be supplied to melt ice, whereas energy (in the form of heat and light) is produced by the explosive combination of hydrogen and oxygen gases. In fact, every change that matter undergoes is accompanied by either ...
Characterization of nanopores by standard enthalpy and entropy of
... Numerous theories have been developed for the determination of pore size distribution in adsorbent materials. The calculated pore size distribution is sensitive to the model selected for adsorption in slits, spheres, or cylinders and cannot be independently verified by experimental methods. Physical ...
... Numerous theories have been developed for the determination of pore size distribution in adsorbent materials. The calculated pore size distribution is sensitive to the model selected for adsorption in slits, spheres, or cylinders and cannot be independently verified by experimental methods. Physical ...