15equil1pp
... Simply states “If the concentrations of all the substances present at equilibrium are raised to the power of the number of moles they appear in the equation, the product of the concentrations of the products divided by the product of the concentrations of the reactants is a constant, provided the te ...
... Simply states “If the concentrations of all the substances present at equilibrium are raised to the power of the number of moles they appear in the equation, the product of the concentrations of the products divided by the product of the concentrations of the reactants is a constant, provided the te ...
Lab announcements – 2 lab quiz week before spring break
... Most chemical reactions do not go to completion. chemical equilibrium – two opposing reactions occur simultaneously at the same rate ‘equilibrium’ doesn’t necessarily mean ‘equal’ amounts of reactants and products – in fact, it usually doesn’t. Equilibrium constant – measure of this balance aA + Kc ...
... Most chemical reactions do not go to completion. chemical equilibrium – two opposing reactions occur simultaneously at the same rate ‘equilibrium’ doesn’t necessarily mean ‘equal’ amounts of reactants and products – in fact, it usually doesn’t. Equilibrium constant – measure of this balance aA + Kc ...
Chapter 15. Chemical Equilibrium
... However, if we start with just ammonia and no nitrogen or hydrogen, the reaction will proceed and N2 and H2 will be produced until equilibrium is achieved. No matter what the starting composition of reactants and products is, the equilibrium mixture contains the same relative concentrations of react ...
... However, if we start with just ammonia and no nitrogen or hydrogen, the reaction will proceed and N2 and H2 will be produced until equilibrium is achieved. No matter what the starting composition of reactants and products is, the equilibrium mixture contains the same relative concentrations of react ...
Stoichiometry and the Mole - 2012 Book Archive
... pound of flour, and 1 pound of sugar. (That’s why it’s called “pound cake.”) If you have 4 pounds of butter, how many pounds of sugar, flour, and eggs do you need? You would need 4 pounds each of sugar, flour, and eggs. Now suppose you have 1.00 g H2. If the chemical reaction follows the balanced ch ...
... pound of flour, and 1 pound of sugar. (That’s why it’s called “pound cake.”) If you have 4 pounds of butter, how many pounds of sugar, flour, and eggs do you need? You would need 4 pounds each of sugar, flour, and eggs. Now suppose you have 1.00 g H2. If the chemical reaction follows the balanced ch ...
Physical Science Standards
... At Level 2, the student is able to SPI interpret a distance-time graph for velocity or a velocity-time graph for acceleration, given the appropriate graph. TPI collect data and construct, analyze, and interpret graphs pertaining to distance, speed, velocity, and time. SPI solve application problems ...
... At Level 2, the student is able to SPI interpret a distance-time graph for velocity or a velocity-time graph for acceleration, given the appropriate graph. TPI collect data and construct, analyze, and interpret graphs pertaining to distance, speed, velocity, and time. SPI solve application problems ...
Chemistry 12 Worksheet 2-3 Calculations Involving the
... given that the partial pressure of each substance at equilibrium is as follows: Partial Pressure of A2 = 20.0 kPa, Partial Pressure of B 2 = 30.0 kPa, Partial Pressure of ...
... given that the partial pressure of each substance at equilibrium is as follows: Partial Pressure of A2 = 20.0 kPa, Partial Pressure of B 2 = 30.0 kPa, Partial Pressure of ...
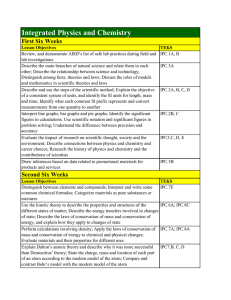
Integrated Physics and Chemistry
... common chemical formulas; Categorize materials as pure substances or mixtures Use the kinetic theory to describe the properties and structures of the different states of matter; Describe the energy transfers involved in changes of state; Describe the laws of conservation of mass and conservation of ...
... common chemical formulas; Categorize materials as pure substances or mixtures Use the kinetic theory to describe the properties and structures of the different states of matter; Describe the energy transfers involved in changes of state; Describe the laws of conservation of mass and conservation of ...
Chemical Equilibrium - 2012 Book Archive
... • The Keq and KP expressions are formulated as amounts of products divided by amounts of reactants; each amount (either a concentration or a pressure) is raised to the power of its coefficient in the balanced chemical equation. • Solids and liquids do not appear in the expression for the equilibrium ...
... • The Keq and KP expressions are formulated as amounts of products divided by amounts of reactants; each amount (either a concentration or a pressure) is raised to the power of its coefficient in the balanced chemical equation. • Solids and liquids do not appear in the expression for the equilibrium ...
LaBrake, Fundamentals Diagnostic Questions
... 45. How many moles of glucose are there in 2.4088 × 1024 molecules of glucose? a) 4 moles b) 2 moles c) 6.0221 × 1023 moles d) 1.4506 × 1048 moles e) insufficient information to answer 46. Calculate the molar mass of copper (II) nitrate, Cu(NO3)2. a) 187.57 g·mol-1 b) 93.56 g·mol-1 c) 125.56 g·mol- ...
... 45. How many moles of glucose are there in 2.4088 × 1024 molecules of glucose? a) 4 moles b) 2 moles c) 6.0221 × 1023 moles d) 1.4506 × 1048 moles e) insufficient information to answer 46. Calculate the molar mass of copper (II) nitrate, Cu(NO3)2. a) 187.57 g·mol-1 b) 93.56 g·mol-1 c) 125.56 g·mol- ...
Chapter 14 Review
... A. Increasing the system volume shifts the equilibrium to the right. B. Increasing the temperature shifts the equilibrium to the right. C. A catalyst speeds up the approach to equilibrium and shifts the position of equilibrium to the right. D. Decreasing the total pressure of the system shifts the e ...
... A. Increasing the system volume shifts the equilibrium to the right. B. Increasing the temperature shifts the equilibrium to the right. C. A catalyst speeds up the approach to equilibrium and shifts the position of equilibrium to the right. D. Decreasing the total pressure of the system shifts the e ...
Predissociation dynamics of lithium iodide
... atomic states but not on the resultant molecular spin; they thus are exactly the same for the low and high-spin states dissociating into the same atoms. The spin-orbit couplings between the nonrelativistic (S − Λ) states are calculated using the MRCI method with the spin-orbit Hamiltonian ĤSO in th ...
... atomic states but not on the resultant molecular spin; they thus are exactly the same for the low and high-spin states dissociating into the same atoms. The spin-orbit couplings between the nonrelativistic (S − Λ) states are calculated using the MRCI method with the spin-orbit Hamiltonian ĤSO in th ...