Molecules, Moles and Chemical Equations File
... Step 2: Balancing an equation like this is aided by making some observations. In this case, both carbon and hydrogen appear in only one place on each side of the equation. Our first steps will be to balance these two elements, because there will be no other way to adjust them. Let’s begin with carbo ...
... Step 2: Balancing an equation like this is aided by making some observations. In this case, both carbon and hydrogen appear in only one place on each side of the equation. Our first steps will be to balance these two elements, because there will be no other way to adjust them. Let’s begin with carbo ...
Equilibrium - Clayton State University
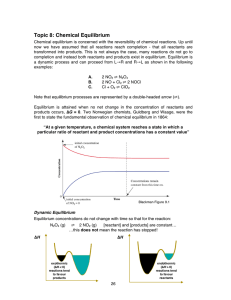
... - Many reactions do not go to completion - Amount of products formed or reactants consumed cannot be predicted from stoichiometry alone - These reactions achieve a condition of equilibrium ...
... - Many reactions do not go to completion - Amount of products formed or reactants consumed cannot be predicted from stoichiometry alone - These reactions achieve a condition of equilibrium ...
Pesticides, Chemical Regulation, and Right-to
... substance when considering potentially “safer” alternatives, as doing so can reveal some of the ambiguities of green chemistry. For example, if a new substance B is marginally less toxic to humans than existing sub ...
... substance when considering potentially “safer” alternatives, as doing so can reveal some of the ambiguities of green chemistry. For example, if a new substance B is marginally less toxic to humans than existing sub ...
Review Unit: Chemistry Review
... know it. We now have cleaner fuel, more durable and safer paints, easy-care clothing, inexpensive fertilizers, life-saving pharmaceuticals, corrosion-resistant tools and machinery, and unusual new materials that we are using in interesting new ways. Much of this innovation has made our lives better ...
... know it. We now have cleaner fuel, more durable and safer paints, easy-care clothing, inexpensive fertilizers, life-saving pharmaceuticals, corrosion-resistant tools and machinery, and unusual new materials that we are using in interesting new ways. Much of this innovation has made our lives better ...
Chapter 18 review
... b. The value of Ks p for the compound formed by the combination of two ions is greater than 1. c. The concentration of one of the ions in solution exceeded the Ks p for that ion in water. d. The product of the concentrations of two ions exceeded the Ks p for the compound formed by the ions. ____ 17. ...
... b. The value of Ks p for the compound formed by the combination of two ions is greater than 1. c. The concentration of one of the ions in solution exceeded the Ks p for that ion in water. d. The product of the concentrations of two ions exceeded the Ks p for the compound formed by the ions. ____ 17. ...
Chapter 6 Table of Contents
... Note how the mol H2 unit cancels, and mol O2 is the new unit introduced. This is an example of a mole-mole calculation, when you start with moles of one substance and convert to moles of another substance by using the balanced chemical equation. The example may seem simple because the numbers are sm ...
... Note how the mol H2 unit cancels, and mol O2 is the new unit introduced. This is an example of a mole-mole calculation, when you start with moles of one substance and convert to moles of another substance by using the balanced chemical equation. The example may seem simple because the numbers are sm ...
7.1 Describing Reactions
... If you examine this equation carefully, you will notice that the number of atoms on the left side does not equal the number of atoms on the right. The equation is not balanced. In order to show that mass is conserved during a reaction, a chemical equation must be balanced. You can balance a chemical ...
... If you examine this equation carefully, you will notice that the number of atoms on the left side does not equal the number of atoms on the right. The equation is not balanced. In order to show that mass is conserved during a reaction, a chemical equation must be balanced. You can balance a chemical ...
Handout - UNT Chemistry
... concentration [say, 0.20 M Zn(NO3)2] and the sample electrode (anode) has the water sample. This cell can be designated as: Zn(s)|Zn2+(xx M)||Zn2+(0.20 M)|Zn(s). The cell potential was measured as +0.078 V. Determine the concentration of Zn2+ in the sample, in mg/L. S6.10. An electrochemical cell is ...
... concentration [say, 0.20 M Zn(NO3)2] and the sample electrode (anode) has the water sample. This cell can be designated as: Zn(s)|Zn2+(xx M)||Zn2+(0.20 M)|Zn(s). The cell potential was measured as +0.078 V. Determine the concentration of Zn2+ in the sample, in mg/L. S6.10. An electrochemical cell is ...