Stability of Matter
... In Section 3.2, we will pull out the e-dependence6 from VC . Note 2.2.1. Let us collect some properties of (2.9) and (2.10). (i) The direction of the Coulomb force is determined by the relative sign of q and Q. If their signs are the same, the force is repulsive. Otherwise, it is attractive. (ii) Th ...
... In Section 3.2, we will pull out the e-dependence6 from VC . Note 2.2.1. Let us collect some properties of (2.9) and (2.10). (i) The direction of the Coulomb force is determined by the relative sign of q and Q. If their signs are the same, the force is repulsive. Otherwise, it is attractive. (ii) Th ...
VIII
... causing a greater decrease of -potential. So, if an electrolyte, having bivalent negative ions will be applied for causing of coagulation in this sole, its coagulating ability will be much greater, than of an electrolyte, containing univalent negative ions. Now we can formulate Shultze-Hardy’s law: ...
... causing a greater decrease of -potential. So, if an electrolyte, having bivalent negative ions will be applied for causing of coagulation in this sole, its coagulating ability will be much greater, than of an electrolyte, containing univalent negative ions. Now we can formulate Shultze-Hardy’s law: ...
Abstraction and its Limits: Finding Space For Novel Explanation
... another theory. When we properly understand the role of abstraction, we appreciate that explanatory value may be irreducible, even where theoretical reduction is possible. Particular kinds of complex relationships between quantities make the abstraction techniques of the higher level theory opaque f ...
... another theory. When we properly understand the role of abstraction, we appreciate that explanatory value may be irreducible, even where theoretical reduction is possible. Particular kinds of complex relationships between quantities make the abstraction techniques of the higher level theory opaque f ...
Water Vapor and Mechanical Work: A Comparison of
... cycle often overestimates the conversion of internal energy into kinetic energy in the presence of water vapor. To better assess the role of water vapor, a novel thermodynamic cycle is introduced here: the steam cycle. A steam cycle transports water from a moist source to a dry sink; in doing so, it ...
... cycle often overestimates the conversion of internal energy into kinetic energy in the presence of water vapor. To better assess the role of water vapor, a novel thermodynamic cycle is introduced here: the steam cycle. A steam cycle transports water from a moist source to a dry sink; in doing so, it ...
Enthalpy, Entropy, Mollier Diagram and Steam
... Most engineers understand the role units play in definition and verification of the engineering concepts, principles, equations and analytical techniques. Therefore, most thermodynamic concepts, principles and computational procedures covered in this text are punctuated with proper units. In additio ...
... Most engineers understand the role units play in definition and verification of the engineering concepts, principles, equations and analytical techniques. Therefore, most thermodynamic concepts, principles and computational procedures covered in this text are punctuated with proper units. In additio ...
Arbitrary Steering of Multiple Particles Independently in an Electro-Osmotically Driven Microfluidic System
... to queue particles, the properties of these particles can then be measured, and downstream actuators can be used to shunt particles of type one to one chamber and type two to another chamber [5]. Individual particles can also be steered by optical ...
... to queue particles, the properties of these particles can then be measured, and downstream actuators can be used to shunt particles of type one to one chamber and type two to another chamber [5]. Individual particles can also be steered by optical ...
The Kinetic Theory of Gases
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
Document
... We can see from the plots that the Poisson distribution is well defined for large values of N, in actuallity the Binomial distribution becomes the Poisson distribution for large value of N. Problem # 4 The Meaning of Never It has been said that “six monkeys, set to strum unintellegently on typewrite ...
... We can see from the plots that the Poisson distribution is well defined for large values of N, in actuallity the Binomial distribution becomes the Poisson distribution for large value of N. Problem # 4 The Meaning of Never It has been said that “six monkeys, set to strum unintellegently on typewrite ...
The Kinetic Theory of Gases - Department of Applied Physics
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
... n Chapter 19 we discussed the properties of an ideal gas, using such macroscopic variables as pressure, volume, and temperature. We shall now show that such large-scale properties can be described on a microscopic scale, where matter is treated as a collection of molecules. Newton’s laws of motion a ...
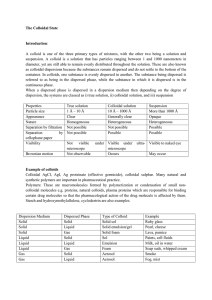
The Colloidal State Introduction: A colloid is one of the three primary
... each other and rotating in opposite directions at a very high speed. The suspension passing through these rotating discs is exposed to a powerful shearing force and the suspended particles are apart to yield particles of colloidal size. Colloid mill are widely used in the industrial preparation of p ...
... each other and rotating in opposite directions at a very high speed. The suspension passing through these rotating discs is exposed to a powerful shearing force and the suspended particles are apart to yield particles of colloidal size. Colloid mill are widely used in the industrial preparation of p ...
The dimer model - Brown math department
... proportional to the product of its edge weights. When G is infinite, this definition will of course not work. For an infinite graph G, a probability measure on X is a Gibbs measure if it is a weak limit of Boltzmann measures on a sequence of finite subgraphs of G filling out G. By this we mean, for ...
... proportional to the product of its edge weights. When G is infinite, this definition will of course not work. For an infinite graph G, a probability measure on X is a Gibbs measure if it is a weak limit of Boltzmann measures on a sequence of finite subgraphs of G filling out G. By this we mean, for ...
Chapter 1 Principles of Probability
... A more mechanical approach to either part (a) or this part is to write out all the possible circumstances. Rejection and acceptance at H are mutually exclusive. Their probabilities add to one. The same for the other two schools. Therefore all possible circumstances are taken into account by adding t ...
... A more mechanical approach to either part (a) or this part is to write out all the possible circumstances. Rejection and acceptance at H are mutually exclusive. Their probabilities add to one. The same for the other two schools. Therefore all possible circumstances are taken into account by adding t ...
Thermodynamics Theory + Questions.0001
... Fig. A quasi q – static process one the pressure of the gass will displace the piston gradually. It is quasista atic. • On the other hand if we remove all a the weights at once th he piston will be kicked up p by the gas pressure. (This is un nrestrained expansion) but b we don’tt consider th hat th ...
... Fig. A quasi q – static process one the pressure of the gass will displace the piston gradually. It is quasista atic. • On the other hand if we remove all a the weights at once th he piston will be kicked up p by the gas pressure. (This is un nrestrained expansion) but b we don’tt consider th hat th ...
IZ3416361639
... Consider electrons in an isotropic, nonparabolic conduction band whose equilibrium Fermi distribution function is f0 (k) in the absence of electric field. Note the equilibrium distribution f0 (k) is isotropic in k space but is perturbed when an electric field is applied. If the electric field is sma ...
... Consider electrons in an isotropic, nonparabolic conduction band whose equilibrium Fermi distribution function is f0 (k) in the absence of electric field. Note the equilibrium distribution f0 (k) is isotropic in k space but is perturbed when an electric field is applied. If the electric field is sma ...
Electrostatic turbulence in tokamaks on transport time scales
... equation for each species, along with Maxwell's equations. To obtain such a closed hybrid description the expressions for the species heat fluxes and viscous stress tensors, as well as the interspecies energy and momentum exchanges, should be written in terms of velocity moments of and require the l ...
... equation for each species, along with Maxwell's equations. To obtain such a closed hybrid description the expressions for the species heat fluxes and viscous stress tensors, as well as the interspecies energy and momentum exchanges, should be written in terms of velocity moments of and require the l ...
Thermodynamics and Statistical Mechanics
... Maxwell’s equations for electromagnetic fields. We can then analyze the system by solving these equations, either exactly or approximately. In thermodynamics, we have no problem formulating the governing equations. The motions of atoms and molecules are described exactly by the laws of quantum mecha ...
... Maxwell’s equations for electromagnetic fields. We can then analyze the system by solving these equations, either exactly or approximately. In thermodynamics, we have no problem formulating the governing equations. The motions of atoms and molecules are described exactly by the laws of quantum mecha ...
Thermodynamic Properties of a single component fluid PV UH += TS
... Since we have 8 different variables (T, P, V, U, H, S, G, & A) and three ways they can be entered into X, Y, and Z, we have a total of 83 = 512 different partial derivatives. The Bridgeman Tables can give us any of these partials, although some are not as useful as others. The Bridgeman Tables requi ...
... Since we have 8 different variables (T, P, V, U, H, S, G, & A) and three ways they can be entered into X, Y, and Z, we have a total of 83 = 512 different partial derivatives. The Bridgeman Tables can give us any of these partials, although some are not as useful as others. The Bridgeman Tables requi ...
H-theorem

In classical statistical mechanics, the H-theorem, introduced by Ludwig Boltzmann in 1872, describes the tendency to increase in the quantity H (defined below) in a nearly-ideal gas of molecules. As this quantity H was meant to represent the entropy of thermodynamics, the H-theorem was an early demonstration of the power of statistical mechanics as it claimed to derive the second law of thermodynamics—a statement about fundamentally irreversible processes—from reversible microscopic mechanics.The H-theorem is a natural consequence of the kinetic equation derived by Boltzmann that has come to be known as Boltzmann's equation. The H-theorem has led to considerable discussion about its actual implications, with major themes being: What is entropy? In what sense does Boltzmann's quantity H correspond to the thermodynamic entropy? Are the assumptions (such as the Stosszahlansatz described below) behind Boltzmann's equation too strong? When are these assumptions violated?↑