Amazing Amino Acids
... function properly without amino acids, which are necessary for the brain to both send and receive information. In fact, not only does every cell in the body contain protein, but protein makes up half of the body's dry weight. Protein is found in muscle, bone, cartilage and blood, as well as enzymes ...
... function properly without amino acids, which are necessary for the brain to both send and receive information. In fact, not only does every cell in the body contain protein, but protein makes up half of the body's dry weight. Protein is found in muscle, bone, cartilage and blood, as well as enzymes ...
Amino acids
... • The nonpolar R-group fill up the interior of the folded protein and help give it its 3D shape. • In proteins located in hydrophobic environment, such as a membrane, the nonpolar R-groups are found on the outside surface of the protein, interacting with lipid environment to stabilize protein struct ...
... • The nonpolar R-group fill up the interior of the folded protein and help give it its 3D shape. • In proteins located in hydrophobic environment, such as a membrane, the nonpolar R-groups are found on the outside surface of the protein, interacting with lipid environment to stabilize protein struct ...
Guidelines for Abstract Submission
... encoded by nuclear genes and have specific N-terminal targeting sequences. Although cell compartmentalization enables distinct roles to organelles, they may have overlapping functions, and a given protein may be required to more than one compartment. Dual targeting appears as a strategy to deal with ...
... encoded by nuclear genes and have specific N-terminal targeting sequences. Although cell compartmentalization enables distinct roles to organelles, they may have overlapping functions, and a given protein may be required to more than one compartment. Dual targeting appears as a strategy to deal with ...
Using a Mixture of Probabilistic Decision Trees for Prediction of
... Sequences often diverge and become unrecognizable ...
... Sequences often diverge and become unrecognizable ...
(Figure 1.3) (Figure 1.6)
... (A). Electrostatic surface presentation of the N-terminal BRCT (BRCT-n) of BRCA1 (PDB:1T29) in complex with a phosphoserine peptide (in magenta). The C-terminal BRCT domain is not directly involved in the phosphate binding and therefore has been deleted from this figure for clarity. Positive potenti ...
... (A). Electrostatic surface presentation of the N-terminal BRCT (BRCT-n) of BRCA1 (PDB:1T29) in complex with a phosphoserine peptide (in magenta). The C-terminal BRCT domain is not directly involved in the phosphate binding and therefore has been deleted from this figure for clarity. Positive potenti ...
Amino acids
... • The nonpolar R-group fill up the interior of the folded protein and help give it its 3D shape. • In proteins located in hydrophobic environment, such as a membrane, the nonpolar R-groups are found on the outside surface of the protein, interacting with lipid environment to stabilize protein struct ...
... • The nonpolar R-group fill up the interior of the folded protein and help give it its 3D shape. • In proteins located in hydrophobic environment, such as a membrane, the nonpolar R-groups are found on the outside surface of the protein, interacting with lipid environment to stabilize protein struct ...
Aligning protein sequences by hand
... and Archaea. We look at the multiple sequence alignment, and if we see positions where all the thermostable stable proteins have one type of residue, and our protein another, we may have a site which we could mutate. If one such position is also far away from the active site, and not in an unpleasan ...
... and Archaea. We look at the multiple sequence alignment, and if we see positions where all the thermostable stable proteins have one type of residue, and our protein another, we may have a site which we could mutate. If one such position is also far away from the active site, and not in an unpleasan ...
Nutrimin Prescribing Information
... increases liver test during intravenous nutrition have been reported. The reasons are at present unclear. The underlying disease and the components and their amount in the intravenous feeding regimens have been suggested. Hypersensitivity reactions have been reported. As with all hypertonic infusion ...
... increases liver test during intravenous nutrition have been reported. The reasons are at present unclear. The underlying disease and the components and their amount in the intravenous feeding regimens have been suggested. Hypersensitivity reactions have been reported. As with all hypertonic infusion ...
Gene expression (Protein synthesis )
... The enzyme peptidyltransferase catalyzes the formation of the peptide bond. ...
... The enzyme peptidyltransferase catalyzes the formation of the peptide bond. ...
85.00 - XBrain
... Proteins are vey sensitive to high temperature or pH extremes. These conditions, which are used to purify ...
... Proteins are vey sensitive to high temperature or pH extremes. These conditions, which are used to purify ...
sample-student-2-Pos.. - NuWrite
... •The Jab1/MPN region has also has been shown to bind ubiquitin. •This criticality of Jab1/MPN and its ubiquitin-binding ability in Prp8 suggest that ubiquitin could regulate splicing through protein-protein interactions involving Prp8. •Characterizing this domain by localizing its critical amino aci ...
... •The Jab1/MPN region has also has been shown to bind ubiquitin. •This criticality of Jab1/MPN and its ubiquitin-binding ability in Prp8 suggest that ubiquitin could regulate splicing through protein-protein interactions involving Prp8. •Characterizing this domain by localizing its critical amino aci ...
BIOCHEMISTRY Medical biochemistry is an essential component of
... glutamate in which side chain COOH groups are amidated. They are classified as neutral amino acids. II- Classification according to polarity of side chain (R): A- Polar amino acids: in which R contains polar hydrophilic group so can forms hydrogen bond with H2O. In those amino acids, R may contain: ...
... glutamate in which side chain COOH groups are amidated. They are classified as neutral amino acids. II- Classification according to polarity of side chain (R): A- Polar amino acids: in which R contains polar hydrophilic group so can forms hydrogen bond with H2O. In those amino acids, R may contain: ...
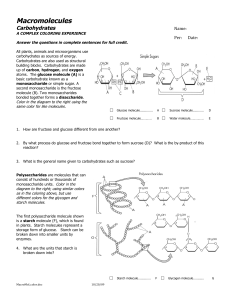
Macromolecule coloring
... called amino acids. A protein may contain as few as ten amino acids, or it may contain thousands. The sequence of amino acids in proteins gives them unique functional characteristics. In the picture below, lightly shade the two amino acids (A) and the peptide bond (D). 1. Which elements are present ...
... called amino acids. A protein may contain as few as ten amino acids, or it may contain thousands. The sequence of amino acids in proteins gives them unique functional characteristics. In the picture below, lightly shade the two amino acids (A) and the peptide bond (D). 1. Which elements are present ...
translation
... 2. The codon a) There are 20 amino acids and 4 nucleotide bases (1) Obviously single nucleotides cannot code for amino acids (2) There is 16 combinations possible for 4 nucleotides read 2 at a time, not enough to code for 20 amino acids (3) There are 64 combinations possible for 4 nucleotides read 3 ...
... 2. The codon a) There are 20 amino acids and 4 nucleotide bases (1) Obviously single nucleotides cannot code for amino acids (2) There is 16 combinations possible for 4 nucleotides read 2 at a time, not enough to code for 20 amino acids (3) There are 64 combinations possible for 4 nucleotides read 3 ...
Zecture #24
... reactions are reversible, proteases can make peptide bonds if the proteins are placed in organic solutions. ‧ Proteases accelerate their rates of reaction as much as 1012 fold! Where does this huge difference come from? o Mostly from binding energy (correct orientation, desolvation, freezing out ent ...
... reactions are reversible, proteases can make peptide bonds if the proteins are placed in organic solutions. ‧ Proteases accelerate their rates of reaction as much as 1012 fold! Where does this huge difference come from? o Mostly from binding energy (correct orientation, desolvation, freezing out ent ...
Amino Acids, Proteins, and Enzymes
... They form about 30% of total body proteins. • There are more than 20 types of collagens, the most common type is collagen I which constitutes about 90% of cell collagens. • Structure of collagen: three helical polypeptide chains (trimeric) twisted around each other forming triplet-helix molecule. • ...
... They form about 30% of total body proteins. • There are more than 20 types of collagens, the most common type is collagen I which constitutes about 90% of cell collagens. • Structure of collagen: three helical polypeptide chains (trimeric) twisted around each other forming triplet-helix molecule. • ...
Gene Normalization - Computational Bioscience Program
... – Search the literature for evidence supporting the prediction ...
... – Search the literature for evidence supporting the prediction ...
Protein – Protein Interactions
... Composed of sequences of amino acids – Variations of 20 primary/basic amino acids ...
... Composed of sequences of amino acids – Variations of 20 primary/basic amino acids ...
Practice Multiple Choice questions
... 3. Which bases form hydrogen bonds between strands of DNA? A) Adenine forms two hydrogen bonds with uracil and cytosine forms three hydrogen bonds with guanine B) Adenine forms three hydrogen bonds with uracil and cytosine forms three hydrogen bonds with guanine C) Adenine forms three hydrogen bonds ...
... 3. Which bases form hydrogen bonds between strands of DNA? A) Adenine forms two hydrogen bonds with uracil and cytosine forms three hydrogen bonds with guanine B) Adenine forms three hydrogen bonds with uracil and cytosine forms three hydrogen bonds with guanine C) Adenine forms three hydrogen bonds ...
Scientists clarify structural basis for biosynthesis of mysterious 21st
... Central to this multi-step process is a Sec-specific transfer RNA (tRNASec) with an unusual structure that enables it to hijack the “stop codon” UGA to allow incorporation of selenocysteine during protein synthesis. In earlier work, the researchers identified features of tRNASec that differentiate i ...
... Central to this multi-step process is a Sec-specific transfer RNA (tRNASec) with an unusual structure that enables it to hijack the “stop codon” UGA to allow incorporation of selenocysteine during protein synthesis. In earlier work, the researchers identified features of tRNASec that differentiate i ...
Gene Ontology (GO)
... classical homology modeling. Most importantly, an example of the correct structure must exist in the structural database that is being screened. If not, the method will fail. The quality of the model is limited by the extent of actual structural similarity between the template and the probe structur ...
... classical homology modeling. Most importantly, an example of the correct structure must exist in the structural database that is being screened. If not, the method will fail. The quality of the model is limited by the extent of actual structural similarity between the template and the probe structur ...
Answer
... The fat substitute Olestra contains a sucrose backbone with six to eight fatty acids attached. How is this different from a naturally occurring fat? Answer: Naturally occurring fats contain a glycerol and three fatty acids. Explanation: This question tested your knowledge of the structure of a fat ...
... The fat substitute Olestra contains a sucrose backbone with six to eight fatty acids attached. How is this different from a naturally occurring fat? Answer: Naturally occurring fats contain a glycerol and three fatty acids. Explanation: This question tested your knowledge of the structure of a fat ...
15 Tacks and a 4 Foot Toober
... • And pairing green tacks that form disulfide bonds, • And keeping all of the polar white tacks on the surface of the protein. After everyone has folded their toober as best they can, the teacher can point out: • Every toober had a different random sequence of tacks (amino acids) and therefore each ...
... • And pairing green tacks that form disulfide bonds, • And keeping all of the polar white tacks on the surface of the protein. After everyone has folded their toober as best they can, the teacher can point out: • Every toober had a different random sequence of tacks (amino acids) and therefore each ...
Amino Acids
... of the Amino acids they mention – competition for the most got! Amino acids are the monomers that make up proteins. All Proteins (also called polypeptides) are made up from the 20 different amino acids. ...
... of the Amino acids they mention – competition for the most got! Amino acids are the monomers that make up proteins. All Proteins (also called polypeptides) are made up from the 20 different amino acids. ...
Teaching Notes
... A3. The P66 and P51 chains interact with each other. The Nevirapine is bound to the P66 chain, at the back of the polymerase active site. Q4. What protein and non-protein components does this structure (PDB ID 2hmi) contain? A4. The structure includes the P66 and P51 RT proteins. In addition there a ...
... A3. The P66 and P51 chains interact with each other. The Nevirapine is bound to the P66 chain, at the back of the polymerase active site. Q4. What protein and non-protein components does this structure (PDB ID 2hmi) contain? A4. The structure includes the P66 and P51 RT proteins. In addition there a ...