This is a heat engine
... 20% and produces an average of 23 kJ of mechanical work per second during operation. Remember: QH = W/e . (a) How much heat input is required, and QH = W/e = 23 kJ/0.20 = 115 kJ (b) How much heat is discharged as waste heat from this engine, per second? QL = (1-e) QH = (0.8) 115 kJ = 92 kJ Copyright ...
... 20% and produces an average of 23 kJ of mechanical work per second during operation. Remember: QH = W/e . (a) How much heat input is required, and QH = W/e = 23 kJ/0.20 = 115 kJ (b) How much heat is discharged as waste heat from this engine, per second? QL = (1-e) QH = (0.8) 115 kJ = 92 kJ Copyright ...
paper - American Society for Engineering Education
... Areas of conceptual misconceptions include: open vs. closed systems, evaluation of properties, state principle, internal energy vs. enthalpy, transient vs. steady state, realizing entropy is a thermodynamic property, reversibility, and correct application of process equations vs. rate equations. A f ...
... Areas of conceptual misconceptions include: open vs. closed systems, evaluation of properties, state principle, internal energy vs. enthalpy, transient vs. steady state, realizing entropy is a thermodynamic property, reversibility, and correct application of process equations vs. rate equations. A f ...
Fragilities of Liquids Predicted from the Random First Order
... by a specific numerical barrier height or fiducial relaxation time will be largely a function of the configurational entropy density there will be another explicit but weak thermodynamic dependence on pressure too. Consistent with Nieuwenhuizen’s recent analysis of the dynamic effects on glass trans ...
... by a specific numerical barrier height or fiducial relaxation time will be largely a function of the configurational entropy density there will be another explicit but weak thermodynamic dependence on pressure too. Consistent with Nieuwenhuizen’s recent analysis of the dynamic effects on glass trans ...
Power laws in finite Ising systems - Journal of Applied Mathematics
... out that the correct infinite system power laws can be observed in the reasonable approximation also in the infinite strip of width L even smaller than 100 lattice spacing provided an appropriate symmetry breaking field is applied. Although in such a case the power-law divergences of the infinite sy ...
... out that the correct infinite system power laws can be observed in the reasonable approximation also in the infinite strip of width L even smaller than 100 lattice spacing provided an appropriate symmetry breaking field is applied. Although in such a case the power-law divergences of the infinite sy ...
Atmospheric Thermodynamics
... reason why temperatures are not additive as well? Why is there not a partial temperature? ...
... reason why temperatures are not additive as well? Why is there not a partial temperature? ...
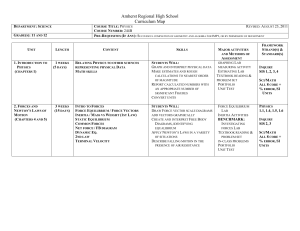
Curriculum Map for Physics - Amherst
... Amherst Regional High School Curriculum Map COURSE TITLE: PHYSICS REVISED: AUGUST 23, 2011 COURSE NUMBER: 244B PRE-REQUISITES (IF ANY): SUCCESSFUL COMPLETION OF GEOMETRY AND ALGEBRA I OR IMP I, OR BY PERMISSION OF DEPARTMENT ...
... Amherst Regional High School Curriculum Map COURSE TITLE: PHYSICS REVISED: AUGUST 23, 2011 COURSE NUMBER: 244B PRE-REQUISITES (IF ANY): SUCCESSFUL COMPLETION OF GEOMETRY AND ALGEBRA I OR IMP I, OR BY PERMISSION OF DEPARTMENT ...
experimental evaluation of heat exchange between water surface
... Additionally all relevant meteorological data are registered on the platform and recorded. The principle of the measuring equipment is that both pools have equal dimensions and that they are exposed to the same meteorological conditions. By means of an electronic control unit the water of the first ...
... Additionally all relevant meteorological data are registered on the platform and recorded. The principle of the measuring equipment is that both pools have equal dimensions and that they are exposed to the same meteorological conditions. By means of an electronic control unit the water of the first ...
About the Guide - American Chemical Society
... We can imagine thermodynamic processes which conserve energy but which never occur in nature. For example, if we bring a hot object into contact with a cold object, we observe that the hot object cools down and the cold object heats up until an equilibrium is reached. The transfer of heat goes from ...
... We can imagine thermodynamic processes which conserve energy but which never occur in nature. For example, if we bring a hot object into contact with a cold object, we observe that the hot object cools down and the cold object heats up until an equilibrium is reached. The transfer of heat goes from ...