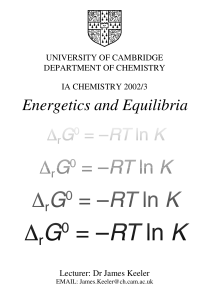Part III: Second Law of Thermodynamics
... (i) The principle of Thomson (Lord Kelvin) states: 'It is impossible by a cyclic process to take heat from a reservoir and to convert it into work without simultaneously transferring heat from a hot to a cold reservoir.' This statement of the second law is related to equilibrium, i.e. work can be ob ...
... (i) The principle of Thomson (Lord Kelvin) states: 'It is impossible by a cyclic process to take heat from a reservoir and to convert it into work without simultaneously transferring heat from a hot to a cold reservoir.' This statement of the second law is related to equilibrium, i.e. work can be ob ...
pages 15
... In many applications of (1.30) the net energy added to a system will cause an increase in temperature. Waste heat from a power plant, for example, will raise the temperature of cooling water drawn into its condenser. The amount of energy needed to raise the temperature of a unit mass of a substance ...
... In many applications of (1.30) the net energy added to a system will cause an increase in temperature. Waste heat from a power plant, for example, will raise the temperature of cooling water drawn into its condenser. The amount of energy needed to raise the temperature of a unit mass of a substance ...
Physics 12c Notes - Caltech Particle Theory Group
... Ph12c, Statistical Mechanics. Administrative details are the same as last term, except that there is one new grader. Same professor, different material. These details are on the course website http://www.theory.caltech.edu/~preskill/ph12c/. Textbook is Thermal Physics by Kittel and Kroemer. In contr ...
... Ph12c, Statistical Mechanics. Administrative details are the same as last term, except that there is one new grader. Same professor, different material. These details are on the course website http://www.theory.caltech.edu/~preskill/ph12c/. Textbook is Thermal Physics by Kittel and Kroemer. In contr ...
lectures in physics - O6U E
... liquid moves upwards and its place is taken by a cold current moving down wards. This is due to the fact that when a portion of liquid near the bottom is heated it expands. Since its mass remains the same, it becomes less dense, and therefore rises. Thus a warm convection current moves upwards and c ...
... liquid moves upwards and its place is taken by a cold current moving down wards. This is due to the fact that when a portion of liquid near the bottom is heated it expands. Since its mass remains the same, it becomes less dense, and therefore rises. Thus a warm convection current moves upwards and c ...
Part II First Law of Thermodynamics
... to the system. If we had defined W as work done by the system, Equation 2-1 would become ∆U = Q- W. • For an isolated system there is no heat or work transferred with the surroundings, thus, by definition W = Q = 0 and therefore ∆U = 0. • The first law of thermodynamics states that this energy diffe ...
... to the system. If we had defined W as work done by the system, Equation 2-1 would become ∆U = Q- W. • For an isolated system there is no heat or work transferred with the surroundings, thus, by definition W = Q = 0 and therefore ∆U = 0. • The first law of thermodynamics states that this energy diffe ...
CE3503 Expectations – Equilibrium Reactions that proceed to
... Reactions that proceed to equilibrium slowly (hours, e.g. BOD exertion, to decades, radioisotope decay) require a kinetic approach as introduced during our discussion of kinetics, reactors and mass balance. Those that proceed more rapidly (milliseconds, e.g. dissociation of strong acid, to minutes, ...
... Reactions that proceed to equilibrium slowly (hours, e.g. BOD exertion, to decades, radioisotope decay) require a kinetic approach as introduced during our discussion of kinetics, reactors and mass balance. Those that proceed more rapidly (milliseconds, e.g. dissociation of strong acid, to minutes, ...
Gibbs Free Energy and the Chemical Potential
... magnitude is limited by the value of the Helmholtz Free Energy (Figure 2.4). For any spontaneous process in which a system evolves from one state to another at constant temperature, a portion of the decrease in the internal energy (TdS) cannot be used to do work because of the requirement that the t ...
... magnitude is limited by the value of the Helmholtz Free Energy (Figure 2.4). For any spontaneous process in which a system evolves from one state to another at constant temperature, a portion of the decrease in the internal energy (TdS) cannot be used to do work because of the requirement that the t ...
Ezio Fornero, Kinetic Theory
... between covolume and number of molecules contained in a given gas amount; in one mole there are about 6.0221023 particles. Therefore the space apparently fitted with an ideal gas is almost completely empty and can be strongly reduced by increasing pressure. 5. The collisions of the molecules agains ...
... between covolume and number of molecules contained in a given gas amount; in one mole there are about 6.0221023 particles. Therefore the space apparently fitted with an ideal gas is almost completely empty and can be strongly reduced by increasing pressure. 5. The collisions of the molecules agains ...
Statistical Physics Exam - FU Berlin
... (b) Determine the sign of the heat and work transfers and change in internal energy for each leg and for the cycle as a whole. Leg 1-2: quasi-static adiabatic compression ...
... (b) Determine the sign of the heat and work transfers and change in internal energy for each leg and for the cycle as a whole. Leg 1-2: quasi-static adiabatic compression ...
Thermodynamic Cycles
... Reversible process can be approximated if a process is performed in a series of small steps. Heat transfer may be considered reversible if it occurs due to a small temperature difference between the system and its surroundings. A temperature difference of 0.00001 °F (degrees Fahrenheit) appears to ...
... Reversible process can be approximated if a process is performed in a series of small steps. Heat transfer may be considered reversible if it occurs due to a small temperature difference between the system and its surroundings. A temperature difference of 0.00001 °F (degrees Fahrenheit) appears to ...
IV. Adiabatic Processes
... IV. Adiabatic Processes If a material undergoes a change in its physical state (e.g., its pressure, volume, or temperature) without any heat being added to it or withdrawn from it, the change is said to be adiabatic. Suppose that the initial state of a material is represented by the point A on the ...
... IV. Adiabatic Processes If a material undergoes a change in its physical state (e.g., its pressure, volume, or temperature) without any heat being added to it or withdrawn from it, the change is said to be adiabatic. Suppose that the initial state of a material is represented by the point A on the ...
Energetics and Equilibria
... saw that some processes could be classed as natural or spontaneous. Such processes take place on their own without any intervention from us. The reverse of such processes do not happen spontaneously, but we can force them to happen by intervening. We saw that it is the Second Law of Thermodynamics w ...
... saw that some processes could be classed as natural or spontaneous. Such processes take place on their own without any intervention from us. The reverse of such processes do not happen spontaneously, but we can force them to happen by intervening. We saw that it is the Second Law of Thermodynamics w ...
unit 61: engineering thermodynamics
... piston or by allowing the pressure to change as it flows through a system such as a turbine. For ease of understanding, let us consider the change as occurring inside a cylinder. The process is best explained with a pressure - volume graph. When the volume changes, the pressure and temperature may a ...
... piston or by allowing the pressure to change as it flows through a system such as a turbine. For ease of understanding, let us consider the change as occurring inside a cylinder. The process is best explained with a pressure - volume graph. When the volume changes, the pressure and temperature may a ...
Energy and the First Law of Thermodynamics
... Internal Energy, E, and State Functions • The internal energy (E) of a system is the sum of all the kinetic and potential energies for every particle in the system. ...
... Internal Energy, E, and State Functions • The internal energy (E) of a system is the sum of all the kinetic and potential energies for every particle in the system. ...
The Ensembles
... In section 2.4 we described the thermodynamics of an isolated system in which U, V and the number of particles N had to be constant. The appropriate thermodynamic function to describe the isolated system was the entropy, S = S(U, V, N ). Since the independent variables of S are constant for an isola ...
... In section 2.4 we described the thermodynamics of an isolated system in which U, V and the number of particles N had to be constant. The appropriate thermodynamic function to describe the isolated system was the entropy, S = S(U, V, N ). Since the independent variables of S are constant for an isola ...
Nature of the anomalies in the supercooled liquid state of the mW
... molecules in the high-temperature liquid, and the fraction f4L < 1 of four-coordinated molecules in the low-temperature liquid. Both f4H and f4L are estimated by an extrapolation of the fraction f4 to high and low temperature. Below Ti , liquid mW water cannot be equilibrated without crystallization ...
... molecules in the high-temperature liquid, and the fraction f4L < 1 of four-coordinated molecules in the low-temperature liquid. Both f4H and f4L are estimated by an extrapolation of the fraction f4 to high and low temperature. Below Ti , liquid mW water cannot be equilibrated without crystallization ...