3.3 and 3.4 Non Flow Energy
... The non-flow energy equation can be applied to any non-flow thermodynamic process. We can classify processes depending on the conditions under which they take place, and the type of fluid. Typical processes that are performed on gases are at constant volume, constant pressure, constant temperature ( ...
... The non-flow energy equation can be applied to any non-flow thermodynamic process. We can classify processes depending on the conditions under which they take place, and the type of fluid. Typical processes that are performed on gases are at constant volume, constant pressure, constant temperature ( ...
Lecture_1_ Heat and - Arizona State University
... system with adiabatic walls so that energy change can only occur by doing some form of mechanical work on the system. For a simple hydrostatic system (pdV work term) the change in internal energy is describable in terms of any two of the variables p, V and T since the third is by an equation of stat ...
... system with adiabatic walls so that energy change can only occur by doing some form of mechanical work on the system. For a simple hydrostatic system (pdV work term) the change in internal energy is describable in terms of any two of the variables p, V and T since the third is by an equation of stat ...
Carnot - UniMAP Portal
... • In many systems, steady state is not achieved until some time has elapsed after the system is started or initiated. This initial situation is often identified as a transient state, start-up or warm-up period. • While a dynamic equilibrium occurs when two or more reversible processes occur at the s ...
... • In many systems, steady state is not achieved until some time has elapsed after the system is started or initiated. This initial situation is often identified as a transient state, start-up or warm-up period. • While a dynamic equilibrium occurs when two or more reversible processes occur at the s ...
Chapter 15 Notes - Valdosta State University
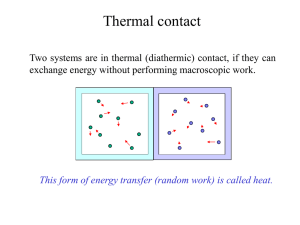
... type of wall. A wall that allows heat flow is called a diathermal wall. One that does not is said to be adiabatic. The state of a system is described by specifying factors that affect the internal energy of the system. In the case of a gas this would be temperature, pressure, volume, and mass. There ...
... type of wall. A wall that allows heat flow is called a diathermal wall. One that does not is said to be adiabatic. The state of a system is described by specifying factors that affect the internal energy of the system. In the case of a gas this would be temperature, pressure, volume, and mass. There ...
Gill_chapter4
... 17a. When a parcel of fluid sinks adiabatically, say into the deep ocean, it experiences increased pressure and therefore warms up. Similarly, when air parcel rises (to mountain top) it experiences less pressure and cools down. In this case, “adiabatic” means that there is no turbulent or molecular ...
... 17a. When a parcel of fluid sinks adiabatically, say into the deep ocean, it experiences increased pressure and therefore warms up. Similarly, when air parcel rises (to mountain top) it experiences less pressure and cools down. In this case, “adiabatic” means that there is no turbulent or molecular ...
Quantum Mechanics_isothermal process An isothermal process is a
... structuredmachines, and even living cells. Various parts of the cycles of some Heat enginesare carried out isothermally and may be approximated by a Carnot cycle. Phase changes, such as melting or evaporation, are also isothermal processes. In Isothermal non flow Process, the work done by compressin ...
... structuredmachines, and even living cells. Various parts of the cycles of some Heat enginesare carried out isothermally and may be approximated by a Carnot cycle. Phase changes, such as melting or evaporation, are also isothermal processes. In Isothermal non flow Process, the work done by compressin ...