Heat Capacity. Enthalpy. Magnetic Systems.
... For a gas it is straightforward to add energy (via heat) without changing the volume. For other kinds of systems (e.g. solids) or for other situations one more easily keeps the pressure held fixed and the volume may vary during the heating/cooling process. Note that allowing the volume to change mea ...
... For a gas it is straightforward to add energy (via heat) without changing the volume. For other kinds of systems (e.g. solids) or for other situations one more easily keeps the pressure held fixed and the volume may vary during the heating/cooling process. Note that allowing the volume to change mea ...
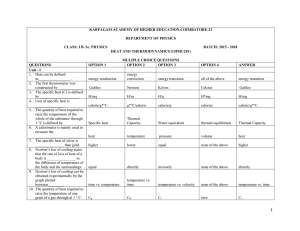
Meteorology Chapter 4 Worksheet 2 Name: Circle the letter that
... T F 24) The saturation vapor pressure is independent of temperature. T F 25) The capacity of air to hold water vapor is temperature dependent T F 26) Relative humidity is "relative" to pressure. T F 27) Relative humidity levels are always higher in St. Louis, MO than Anchorage, AK. T F 28 ...
... T F 24) The saturation vapor pressure is independent of temperature. T F 25) The capacity of air to hold water vapor is temperature dependent T F 26) Relative humidity is "relative" to pressure. T F 27) Relative humidity levels are always higher in St. Louis, MO than Anchorage, AK. T F 28 ...
Exercise No. 1 - People(dot)tuke(dot)
... thermal equilibrium and is the basis for the concept of temperature. The most common enunciation of the zeroth law of thermodynamics is: “If two thermodynamic systems are in thermal equilibrium with a third, they are also in thermal equilibrium with each other.” In other words, the zeroth law says t ...
... thermal equilibrium and is the basis for the concept of temperature. The most common enunciation of the zeroth law of thermodynamics is: “If two thermodynamic systems are in thermal equilibrium with a third, they are also in thermal equilibrium with each other.” In other words, the zeroth law says t ...
Heat and temperature
... • Applications - systems with energy inputs and outputs: heat engines, heat pumps, refrigerators, … • Based upon but not concerned with microscopic details ...
... • Applications - systems with energy inputs and outputs: heat engines, heat pumps, refrigerators, … • Based upon but not concerned with microscopic details ...
ME 204 Thermodynamics I
... control surfaces for various work modes or use the first law or conservation of mass)? (determination of properties from the relation between them) vii) what we have done so far in previous steps, how do we proceed to find whatever it is that is desired? Is a trial-anderror solution necessary? (anot ...
... control surfaces for various work modes or use the first law or conservation of mass)? (determination of properties from the relation between them) vii) what we have done so far in previous steps, how do we proceed to find whatever it is that is desired? Is a trial-anderror solution necessary? (anot ...
Ezio Fornero, The Principle of Equivalence of Heat and
... Although we are discuss only dissipative cycles, the equivalence of heat and work applies to every cyclic process – heat engines function by converting heat into work during an indefinite series of cycles. But what about non-cyclic transformations? The principle of equivalence describes the nature o ...
... Although we are discuss only dissipative cycles, the equivalence of heat and work applies to every cyclic process – heat engines function by converting heat into work during an indefinite series of cycles. But what about non-cyclic transformations? The principle of equivalence describes the nature o ...
Class notes
... to calculate the iH2O we need to know Xi H2O to do this we need something called molar volume 1st the concept of molar volume molar vol = liquid vol of one mole (L/mol) ...
... to calculate the iH2O we need to know Xi H2O to do this we need something called molar volume 1st the concept of molar volume molar vol = liquid vol of one mole (L/mol) ...
**** 1 - apctp
... Heat Capacities for constant volume, gravity and for constant volume, temperature: ...
... Heat Capacities for constant volume, gravity and for constant volume, temperature: ...
Internal Energy, Heat, Enthalpy, and Calorimetry
... What is Enthalpy and How Does it Relate to Internal Energy (E)? If a process takes place at constant pressure (as the majority of processes we study do) and the only work done is this pressure–volume work, we can account for heat flow during the process by measuring the enthalpy of the system E ...
... What is Enthalpy and How Does it Relate to Internal Energy (E)? If a process takes place at constant pressure (as the majority of processes we study do) and the only work done is this pressure–volume work, we can account for heat flow during the process by measuring the enthalpy of the system E ...
The Heat Equation - Rose
... ends of the bar. Now since q = −c1 ρx = −kc1 ux quantifies the rate of heat flow, we typically specify conditions like −kc1 ux (0, t) = ψ0 (t) (the rate heat ENTERS at x = 0) and kc1 ux (L, 0) = ψL (t) (the rate heat ENTERS at x = L) for functions ψ0 and ψL . A very common Neumman boundary condition i ...
... ends of the bar. Now since q = −c1 ρx = −kc1 ux quantifies the rate of heat flow, we typically specify conditions like −kc1 ux (0, t) = ψ0 (t) (the rate heat ENTERS at x = 0) and kc1 ux (L, 0) = ψL (t) (the rate heat ENTERS at x = L) for functions ψ0 and ψL . A very common Neumman boundary condition i ...