aq - Byron High School
... Oxidation Numbers Rules 3. Nonmetals tend to have negative oxidation numbers, although some are positive in certain compounds or ions. c. Fluorine always has an oxidation number of −1. d. The other halogens have an oxidation number of −1 when they are negative; they can have positive oxidation numb ...
... Oxidation Numbers Rules 3. Nonmetals tend to have negative oxidation numbers, although some are positive in certain compounds or ions. c. Fluorine always has an oxidation number of −1. d. The other halogens have an oxidation number of −1 when they are negative; they can have positive oxidation numb ...
Kinetic multi-layer model of aerosol surface and bulk chemistry (KM
... a kinetic model framework (PRA framework) with a double-layer surface concept and universally applicable rate equations and parameters for mass transport and chemical reactions at the gas-particle interface of aerosols and clouds (Pöschl et al., 2007). Ammann and Pöschl (2007) provided first examp ...
... a kinetic model framework (PRA framework) with a double-layer surface concept and universally applicable rate equations and parameters for mass transport and chemical reactions at the gas-particle interface of aerosols and clouds (Pöschl et al., 2007). Ammann and Pöschl (2007) provided first examp ...
ABSTRACT Title of Document:
... hydrophobic liquid is mixed with water. In this case, two liquid phases are formed above a lower critical solution temperature (LCST) due to the negative entropy of mixing. Enthalpically-driven phase separation generally occurs when two liquids, at least moderately different in structure and/or pola ...
... hydrophobic liquid is mixed with water. In this case, two liquid phases are formed above a lower critical solution temperature (LCST) due to the negative entropy of mixing. Enthalpically-driven phase separation generally occurs when two liquids, at least moderately different in structure and/or pola ...
Supplementary Exercise 1B Topic 5
... For the first two chemical cells, metal W is the positive electrode while metals X and Y are the negative electrodes. Therefore metals X and Y form ions more readily than metal W. The Y/W couple gives a higher voltage than the X/W couple. Therefore the difference in the tendency to form ions between ...
... For the first two chemical cells, metal W is the positive electrode while metals X and Y are the negative electrodes. Therefore metals X and Y form ions more readily than metal W. The Y/W couple gives a higher voltage than the X/W couple. Therefore the difference in the tendency to form ions between ...
mass
... Calculating average r.a.m. from isotopes To calculate the average r.a.m. of a mixture of isotopes, multiply the percentage of each isotope( as a decimal) by its relative atomic mass and then add these together. Naturally-occurring bromine is composed of two isotopes: bromine-79 (50.5%) and bromine- ...
... Calculating average r.a.m. from isotopes To calculate the average r.a.m. of a mixture of isotopes, multiply the percentage of each isotope( as a decimal) by its relative atomic mass and then add these together. Naturally-occurring bromine is composed of two isotopes: bromine-79 (50.5%) and bromine- ...
Synthesis and Characterization of Dendrimer Templated Supported
... of bimetallic Pt-Au NPs was also identified.13,21,23,24 Bimetallic Pt-Au NPs within the miscibility gap can also be prepared in zeolites by using the zeolite supercages to arrest particle agglomeration and trap bimetallic NPs.25 Inorganic and organometallic cluster compounds have also been used to s ...
... of bimetallic Pt-Au NPs was also identified.13,21,23,24 Bimetallic Pt-Au NPs within the miscibility gap can also be prepared in zeolites by using the zeolite supercages to arrest particle agglomeration and trap bimetallic NPs.25 Inorganic and organometallic cluster compounds have also been used to s ...
oltmanns-Diffraction and Interference
... sharp top to each peak. For error, just the pixel to pixel difference for position and intensity were taken into account. Having shaper peaks means that we can be more certain about the point we pick. The single slit doesn’t have as many interference points and therefore can’t provide nearly as shar ...
... sharp top to each peak. For error, just the pixel to pixel difference for position and intensity were taken into account. Having shaper peaks means that we can be more certain about the point we pick. The single slit doesn’t have as many interference points and therefore can’t provide nearly as shar ...
AP Chemistry Curriculum Map - Belle Vernon Area School District
... Dalton (atomic theory), Thomson (the electron), Rutherford (the nucleus), and Bohr (planetary model of the atom) and understand how each discovery leads to modern theory. Anchor: CHEM.A.1.1 Identify and describe how observable and measurable properties can be used to classify and describe matter and ...
... Dalton (atomic theory), Thomson (the electron), Rutherford (the nucleus), and Bohr (planetary model of the atom) and understand how each discovery leads to modern theory. Anchor: CHEM.A.1.1 Identify and describe how observable and measurable properties can be used to classify and describe matter and ...
Pore Scale Modeling of Rock Properties and Comparison to Laboratory Measurements
... with feldspar, sulfate, and carbonates, which are usually either isolated or small, also do not contribute to permeability (Nelson, 2000; Wu, 2004). Thus, as long as the microtomography ...
... with feldspar, sulfate, and carbonates, which are usually either isolated or small, also do not contribute to permeability (Nelson, 2000; Wu, 2004). Thus, as long as the microtomography ...
B.Sc. in Physics - Masinde Muliro University of Science and
... supervisor will agree on the topics. The student will make at least two presentations of about 40 minutes’ duration during the semester. A written report has to be presented to the tutor a week before the presentation dates. At least three lecturers of the department will compose the Presentation ex ...
... supervisor will agree on the topics. The student will make at least two presentations of about 40 minutes’ duration during the semester. A written report has to be presented to the tutor a week before the presentation dates. At least three lecturers of the department will compose the Presentation ex ...
Construction and operation - the UQ Bose
... trapping and evaporation in optical dipole traps and the experimental realisation. In the future this experiment will be extended by a fast scanning dipole trap to apply arbitrary time averaged potentials of more complex geometries, such as toroidal traps to study the effects of superfluidity in a pe ...
... trapping and evaporation in optical dipole traps and the experimental realisation. In the future this experiment will be extended by a fast scanning dipole trap to apply arbitrary time averaged potentials of more complex geometries, such as toroidal traps to study the effects of superfluidity in a pe ...
Metal Nanoparticles for Microscopy and Spectroscopy
... 3.2 The Optical Response of Bulk Metals The optical response of metal nanostructures is primarily determined by the metal’s conduction electrons, which react collectively to external perturbations such as the applied electric field of a light wave. In this chapter, we are concerned with small particl ...
... 3.2 The Optical Response of Bulk Metals The optical response of metal nanostructures is primarily determined by the metal’s conduction electrons, which react collectively to external perturbations such as the applied electric field of a light wave. In this chapter, we are concerned with small particl ...
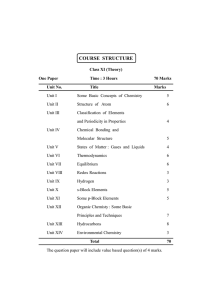
COURSE STRUCTURE
... as well as by the photoelectric effect. Heisenberg’s uncertainty Principle states that ‘‘It is impossible to measure simultaneously the position and momentum of a microscopic particle with absolute accuracy. If one of them is measured with greater accuracy, the other becomes less accurate. The produ ...
... as well as by the photoelectric effect. Heisenberg’s uncertainty Principle states that ‘‘It is impossible to measure simultaneously the position and momentum of a microscopic particle with absolute accuracy. If one of them is measured with greater accuracy, the other becomes less accurate. The produ ...
Full text in DIVA
... the sample reaches the focal plane, Eq. 共4兲 reaches a maximum value. As the sample continues to be moved in the positive z-direction, Eq. 共4兲 becomes small again, since the focus of the laser beam now is inside the sample, and SH light generated before and after focus are phase shifted by radians ...
... the sample reaches the focal plane, Eq. 共4兲 reaches a maximum value. As the sample continues to be moved in the positive z-direction, Eq. 共4兲 becomes small again, since the focus of the laser beam now is inside the sample, and SH light generated before and after focus are phase shifted by radians ...
Subject Area Standard Area Organizing Category Grade Level
... CHEM.A.2.1.1: Describe the evolution of atomic theory leading to the current model of the atom based on the works of Dalton, Thomson, Rutherford, and Bohr. ...
... CHEM.A.2.1.1: Describe the evolution of atomic theory leading to the current model of the atom based on the works of Dalton, Thomson, Rutherford, and Bohr. ...
supplementary information
... ordered surface domains. In general, the small (100) and (111) facets spontaneously formed on the single-crystal sphere have larger ordered domains or less surface defects than the corresponding large ...
... ordered surface domains. In general, the small (100) and (111) facets spontaneously formed on the single-crystal sphere have larger ordered domains or less surface defects than the corresponding large ...
The O 1s and V 2p X-ray Absorption Spectra of Vanadium Oxides
... include explic tly tlie dominant 2p core-leve1 electrons. In this case, tlie spectra present strong multiplet effects which are caused by relatively large Coulomb and exchange interactions. The spectra can be explained by atomic-like calculations projected on the appropriate crystal-field. 't'he ato ...
... include explic tly tlie dominant 2p core-leve1 electrons. In this case, tlie spectra present strong multiplet effects which are caused by relatively large Coulomb and exchange interactions. The spectra can be explained by atomic-like calculations projected on the appropriate crystal-field. 't'he ato ...
Version A
... 27. Which of the following sets of quantum numbers (n, , m , ms) best describes the valence electron of highest energy in a ground-state gallium atom (atomic number 31) ? A) 4, 0, 0, ½ B) 4, 0, 1, ½ ...
... 27. Which of the following sets of quantum numbers (n, , m , ms) best describes the valence electron of highest energy in a ground-state gallium atom (atomic number 31) ? A) 4, 0, 0, ½ B) 4, 0, 1, ½ ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.