Chapter 3 Notes
... Definition – weighted average mass of the atoms in a naturally occurring sample of the ...
... Definition – weighted average mass of the atoms in a naturally occurring sample of the ...
Review Package KCI 2017 Sem 1
... convert kinetic energy to activation energy) each reactant requires a different collision geometry that can be simple or complex 2. TEMPERATURE : an increase in temperature increases the rate of reaction as temperature rises, the reactant particles gain kinetic energy, moving faster, colliding ...
... convert kinetic energy to activation energy) each reactant requires a different collision geometry that can be simple or complex 2. TEMPERATURE : an increase in temperature increases the rate of reaction as temperature rises, the reactant particles gain kinetic energy, moving faster, colliding ...
pp. 18-21
... 3. The following substances are fertilizers that contribute nitrogen to the soil: urea [(NH2)2CO]; ammonium nitrate; ammonium sulfate. Arrange these fertilizers in order of decreasing nitrogen content on a mass percentage basis. a) b) c) d) e) ...
... 3. The following substances are fertilizers that contribute nitrogen to the soil: urea [(NH2)2CO]; ammonium nitrate; ammonium sulfate. Arrange these fertilizers in order of decreasing nitrogen content on a mass percentage basis. a) b) c) d) e) ...
Chapter 17 Thermodynamics: Directionality of Chemical Reactions
... E stays with original atoms in only 3 (of 10) cases: • 70% chance that E will disperse. • More atoms: E concentration is much less likely. ...
... E stays with original atoms in only 3 (of 10) cases: • 70% chance that E will disperse. • More atoms: E concentration is much less likely. ...
B. Gaussian Beam Transformation by a Lens.
... beam parameters and the lens focal length must be known.) 1. Beam expanding, collimating, and focusing. - First expand the laser beam using an objective and spatial filter. - Add a lens of longer focal length to collimate the beam (see Laboratory Technique). - Insert another lens that will be used i ...
... beam parameters and the lens focal length must be known.) 1. Beam expanding, collimating, and focusing. - First expand the laser beam using an objective and spatial filter. - Add a lens of longer focal length to collimate the beam (see Laboratory Technique). - Insert another lens that will be used i ...
PRACTICE EXERCISE - Needham.K12.ma.us
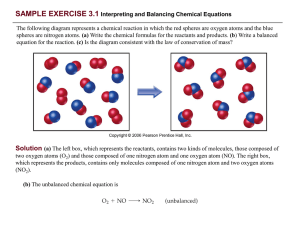
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
... (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each two NO as required by the balanced equation. The right box (products) contains eight NO 2 molecules. The number of NO2 molecules on the right equals the number of NO molecules ...
Field distributions in the crossover from ballistic to diffusive wave
... large number of waveguide modes excited in our sample, and that the deviations would be larger, if only a single mode were excited by the incident wave. The wave generated by the horn excites a number of low order waveguide modes. When the horn is replaced by a wire antenna positioned just in front ...
... large number of waveguide modes excited in our sample, and that the deviations would be larger, if only a single mode were excited by the incident wave. The wave generated by the horn excites a number of low order waveguide modes. When the horn is replaced by a wire antenna positioned just in front ...
1 Mole
... How many moles of O2 are needed to react with 4 moles of sodium? How many moles of O2 are needed to react with 2 moles of sodium? How many moles of Na2O are produced when 2 moles of sodium are used? ...
... How many moles of O2 are needed to react with 4 moles of sodium? How many moles of O2 are needed to react with 2 moles of sodium? How many moles of Na2O are produced when 2 moles of sodium are used? ...
Analysis of a single-atom dipole trap
... this FORT have a very low photon scattering rate and therefore negligible photon recoil heating. Confinement times up to 4 s are achieved with no additional cooling. Because of the small trap volume, only a single atom can be loaded at a time 关21兴. To prove this property the photon statistics of the ...
... this FORT have a very low photon scattering rate and therefore negligible photon recoil heating. Confinement times up to 4 s are achieved with no additional cooling. Because of the small trap volume, only a single atom can be loaded at a time 关21兴. To prove this property the photon statistics of the ...
Demonstration of Optical Rotatory Dispersion of Sucrose
... in the region of wavelength λ, the optical rotation is a sum over such expressions. When the specific rotation of a compound decreases with increasing wavelength as in eq 3 and there are no local extrema in the curve, the optical rotatory dispersion (ORD) curve is called a plain curve. Many articles ...
... in the region of wavelength λ, the optical rotation is a sum over such expressions. When the specific rotation of a compound decreases with increasing wavelength as in eq 3 and there are no local extrema in the curve, the optical rotatory dispersion (ORD) curve is called a plain curve. Many articles ...
AP Chemistry: Bonding Multiple Choice
... (C) Atoms contain protons, neutrons, and electrons. (D) Atoms have a positively charged nucleus surrounded by an electron cloud. (E) No two electrons in one atom can have the same four quantum numbers. 1. Use these answers for questions 1 - 3. (A) O ...
... (C) Atoms contain protons, neutrons, and electrons. (D) Atoms have a positively charged nucleus surrounded by an electron cloud. (E) No two electrons in one atom can have the same four quantum numbers. 1. Use these answers for questions 1 - 3. (A) O ...
notes and handout
... 6) Find the number of lone pair (nonbonding) electrons by subtracting the bonding electrons (#3 above) from the valence electrons (#1 above). Arrange these around the atoms until all of them satisfy the octet rule: Remember, ALL elements EXCEPT hydrogen want eight electrons around them, total. Hydro ...
... 6) Find the number of lone pair (nonbonding) electrons by subtracting the bonding electrons (#3 above) from the valence electrons (#1 above). Arrange these around the atoms until all of them satisfy the octet rule: Remember, ALL elements EXCEPT hydrogen want eight electrons around them, total. Hydro ...
Download PDF
... is frequency shifted with respect to the other, such that the phase of the interference term becomes ∆φ = ∆ωτ + δφ, the interference term will be oscillating at a selected frequency permitting high-fidelity measurements. Since the measured signal is proportional to the product of two fields, one of wh ...
... is frequency shifted with respect to the other, such that the phase of the interference term becomes ∆φ = ∆ωτ + δφ, the interference term will be oscillating at a selected frequency permitting high-fidelity measurements. Since the measured signal is proportional to the product of two fields, one of wh ...
Net ionic equation
... • In the above rxn, Mg(s) loses e-, H+ gains e• Oxidized: atom, molecule, or ion becomes more positively charged. • Reduced: atom, molecule, or ion becomes less positively charged. ...
... • In the above rxn, Mg(s) loses e-, H+ gains e• Oxidized: atom, molecule, or ion becomes more positively charged. • Reduced: atom, molecule, or ion becomes less positively charged. ...
regents chemistry midterm - irondequoit 2014_entire exam w key
... 7. Compared to the charge and mass of a proton, an electron has 1) the same charge and a smaller mass 2) the same charge and the same mass 3) an opposite charge and a smaller mass 4) an opposite charge and the same mass 8. An atom in the excited state can have an electron configuration of 1) 2 - 8 - ...
... 7. Compared to the charge and mass of a proton, an electron has 1) the same charge and a smaller mass 2) the same charge and the same mass 3) an opposite charge and a smaller mass 4) an opposite charge and the same mass 8. An atom in the excited state can have an electron configuration of 1) 2 - 8 - ...
Answers/solutions
... Measure the 3d/4s crossing and we find it occurs at atomic number=22. This means the HF calculations made by Latter either overestimated the energy of 3d or underestimated the energy of 4s, or both.. this means inter-electronic repulsions in 3d calculation were overestimated or that in 4s calculatio ...
... Measure the 3d/4s crossing and we find it occurs at atomic number=22. This means the HF calculations made by Latter either overestimated the energy of 3d or underestimated the energy of 4s, or both.. this means inter-electronic repulsions in 3d calculation were overestimated or that in 4s calculatio ...
Evaporation–glow discharge hybrid source for plasma immersion
... usually immersed in the plasma and then biased by the high negative-pulsed voltage. The electrons are immediately repelled and the sheath dynamically expanded during the pulse. Ions are accelerated through the sheath and implanted into the surface of the substrate. PIII eliminates the need of beam r ...
... usually immersed in the plasma and then biased by the high negative-pulsed voltage. The electrons are immediately repelled and the sheath dynamically expanded during the pulse. Ions are accelerated through the sheath and implanted into the surface of the substrate. PIII eliminates the need of beam r ...
Effective mass of electron in monolayer graphene: Electron
... Si-face chemo-mechanically polished substrates. All the substrates were obtained from a single 4-in. wafer to avoid the influence of large variations of unintentional off-cut from wafer to wafer. Three samples were grown during each growth run to have similar graphene. This was mainly to facilitate ...
... Si-face chemo-mechanically polished substrates. All the substrates were obtained from a single 4-in. wafer to avoid the influence of large variations of unintentional off-cut from wafer to wafer. Three samples were grown during each growth run to have similar graphene. This was mainly to facilitate ...
Fall 2013 Final practice questions w/o solution
... 8. Which statement best explains why the electron affinity of B is less positive than that of Li? A) B has electrons in p orbitals, so it is lower in energy. B) Li does not want to gain an electron because it already has a half-full s orbital. C) B is a smaller atom, so its valence electrons are mo ...
... 8. Which statement best explains why the electron affinity of B is less positive than that of Li? A) B has electrons in p orbitals, so it is lower in energy. B) Li does not want to gain an electron because it already has a half-full s orbital. C) B is a smaller atom, so its valence electrons are mo ...
Structure of atoms
... Scientists have discovered 118 different substances that are made of one type of atom only – such substances (e.g. hydrogen, oxygen, carbon, gold) are called elements. An atom is the smallest part of an element that can take part in a chemical reaction. Atoms are so small that it would take 4 billio ...
... Scientists have discovered 118 different substances that are made of one type of atom only – such substances (e.g. hydrogen, oxygen, carbon, gold) are called elements. An atom is the smallest part of an element that can take part in a chemical reaction. Atoms are so small that it would take 4 billio ...
AP Chemistry Unit 1 Essential Questions Screencast 1
... 3. When converting units with a power what is important to remember? Screencast 1-7 Basic Atomic Structure, Isotopes and Mass Spectroscopy 1. What are the 3 subatomic particles and where are they located inside the atom? 2. What are the relative masses of the 3 particles? 3. What is the atomic mass? ...
... 3. When converting units with a power what is important to remember? Screencast 1-7 Basic Atomic Structure, Isotopes and Mass Spectroscopy 1. What are the 3 subatomic particles and where are they located inside the atom? 2. What are the relative masses of the 3 particles? 3. What is the atomic mass? ...
Degenerate Fermi Gases
... present for any value of the interaction also for small negative scattering length: COOPER PAIR FORMATION and BCS-superfluidity In 3D in vacuum an attractive potential has to be “attractive enough” to bound a pair of (red-blue) atoms. In 2D and 1D any attractive potential make it due to the differen ...
... present for any value of the interaction also for small negative scattering length: COOPER PAIR FORMATION and BCS-superfluidity In 3D in vacuum an attractive potential has to be “attractive enough” to bound a pair of (red-blue) atoms. In 2D and 1D any attractive potential make it due to the differen ...
Quantum Random Bit Generation using Energy Fluctuations in
... According to these criteria, we selected diamond for construction of a prototype device. Diamond has a high Raman gain, a large Raman shift, and a short phonon decay time of 3.5 ps, making it an excellent material for QRBG. Bulk diamond was easily interfaced with pump pulses from our available laser ...
... According to these criteria, we selected diamond for construction of a prototype device. Diamond has a high Raman gain, a large Raman shift, and a short phonon decay time of 3.5 ps, making it an excellent material for QRBG. Bulk diamond was easily interfaced with pump pulses from our available laser ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.