Slide 1 ______
... Atoms are stable (inert) when the outermost shell is complete How to fill the atom’s shells Shell 1 can hold a maximum of 2 electrons ...
... Atoms are stable (inert) when the outermost shell is complete How to fill the atom’s shells Shell 1 can hold a maximum of 2 electrons ...
covalent - Typepad
... 50. The type of bond in Br2 (electronegativity for Br is 2.8) is a. nonpolar covalent. c. pure covalent. b. polar covalent. d. pure ionic. 51. The type of bond in LiCl (electronegativity for Li is 1.0; electronegativity for Cl is 3.2) is a. pure ionic. c. nonpolar covalent. b. polar covalent. d. ion ...
... 50. The type of bond in Br2 (electronegativity for Br is 2.8) is a. nonpolar covalent. c. pure covalent. b. polar covalent. d. pure ionic. 51. The type of bond in LiCl (electronegativity for Li is 1.0; electronegativity for Cl is 3.2) is a. pure ionic. c. nonpolar covalent. b. polar covalent. d. ion ...
View accepted manuscript: Metastable triply charged diatomic
... pump-probe experiment since the rising and falling parts of the pulse act like pre- and post-pulses, respectively, for the high intensity peak part of the pulse. Although diatomic trications have been produced before [7,16], they have not been produced by optical means or in large quantities. Optica ...
... pump-probe experiment since the rising and falling parts of the pulse act like pre- and post-pulses, respectively, for the high intensity peak part of the pulse. Although diatomic trications have been produced before [7,16], they have not been produced by optical means or in large quantities. Optica ...
Coulomb effect in multiphoton ionization of rare
... establishing the calibration. The accuracy in the relative intensity calibration between one given ion curve and the reference He curve is estimated to ±5% (see figure 1). The experimentally measured ion yield versus intensity curves were compared with those predicted by five models: ADK, SFA, PPT, ...
... establishing the calibration. The accuracy in the relative intensity calibration between one given ion curve and the reference He curve is estimated to ±5% (see figure 1). The experimentally measured ion yield versus intensity curves were compared with those predicted by five models: ADK, SFA, PPT, ...
Laser - nptel
... states may be picturised as follows. The three hydrogen atoms are at the vertices of an equilateral triangle which forms the base of a pyramid with the nitrogen atom at the apex of the pyramid. The nitrogen atom may be in two possible positions, either above the hydrgen plane or below it. (Physicall ...
... states may be picturised as follows. The three hydrogen atoms are at the vertices of an equilateral triangle which forms the base of a pyramid with the nitrogen atom at the apex of the pyramid. The nitrogen atom may be in two possible positions, either above the hydrgen plane or below it. (Physicall ...
Cathodoluminescence in the scanning transmission electron
... as non-radiative centres, quenching the emission of the object of interest, but others can be radiative. Colour centres can be highly absorbing and/or emitting, dominating the optical properties of the host matrix in the visible range - consider for example the case of a ruby stone. Colour centres a ...
... as non-radiative centres, quenching the emission of the object of interest, but others can be radiative. Colour centres can be highly absorbing and/or emitting, dominating the optical properties of the host matrix in the visible range - consider for example the case of a ruby stone. Colour centres a ...
Document
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
Synthesis and Characterization of Large Colloidal Silver Particles
... Au, Al, Ni, Cu) have been investigated theoretically as possible candidates for metallodielectric photonic crystals.10-14 Because of its low bulk absorption, silver (Ag) is the most suitable metal to create a CPBG in the visible. Recent calculations of Moroz have shown that a CPBG can even be opened ...
... Au, Al, Ni, Cu) have been investigated theoretically as possible candidates for metallodielectric photonic crystals.10-14 Because of its low bulk absorption, silver (Ag) is the most suitable metal to create a CPBG in the visible. Recent calculations of Moroz have shown that a CPBG can even be opened ...
Powerpoint template for scientific posters (Swarthmore College)
... done. Changing parameters in figure 1, the figure 2 is found. It shows us FSO fade mardgin graph will determine us at which path length with which availability. According to availability we can choose our link distance or we can change our parameters to obtain availability. ...
... done. Changing parameters in figure 1, the figure 2 is found. It shows us FSO fade mardgin graph will determine us at which path length with which availability. According to availability we can choose our link distance or we can change our parameters to obtain availability. ...
Vinnitsa National Pirogov Memorial Medical University Biological
... Did you study chemistry yes/no. For how many years______________ What parts of chemistry did you study: inorganic chemistry yes/no, organic chemistry yes/no. After I had finished to study chemistry ______________ years/months passed. The students who studied chemistry at their countries we ask to re ...
... Did you study chemistry yes/no. For how many years______________ What parts of chemistry did you study: inorganic chemistry yes/no, organic chemistry yes/no. After I had finished to study chemistry ______________ years/months passed. The students who studied chemistry at their countries we ask to re ...
b1.2. generation and propagation of higher order gaussian beams
... Generation of higher order modes are interesting because of the phase singularity and the characteristic intensity distribution. In particular, the dark region is very useful to manipulate particles in single beam optical traps [1]. Large number of vortices available in higher order modes could be a ...
... Generation of higher order modes are interesting because of the phase singularity and the characteristic intensity distribution. In particular, the dark region is very useful to manipulate particles in single beam optical traps [1]. Large number of vortices available in higher order modes could be a ...
Solitons: From Canal Water Waves to Molecular
... “I was observing the motion of a boat which was rapidly drawn along a narrow channel by a pair of horses, when the boat suddenly stopped - not so the mass of water in the channel which it had put in motion; it accumulated round the prow of the vessel in a state of violent agitation, then suddenly le ...
... “I was observing the motion of a boat which was rapidly drawn along a narrow channel by a pair of horses, when the boat suddenly stopped - not so the mass of water in the channel which it had put in motion; it accumulated round the prow of the vessel in a state of violent agitation, then suddenly le ...
East Meck Chemistry
... A scientific method is a systematic approach to answer a ______________________ or study a situation. It starts with _____________________ - noting and recording facts. A _________________________ is a possible explanation for what has been observed. It is an educated _________________ as to the cau ...
... A scientific method is a systematic approach to answer a ______________________ or study a situation. It starts with _____________________ - noting and recording facts. A _________________________ is a possible explanation for what has been observed. It is an educated _________________ as to the cau ...
Review on X-ray Detectors Based on Scintillators and CMOS
... Occasionally, the electron passes very close to an atomic nucleus, where it suffers a deflection, mainly due to the nuclear charge. The interaction of the electron with the atomic nucleus results in a change of its kinetic energy, which results in the emission of a photon in the spectral range of th ...
... Occasionally, the electron passes very close to an atomic nucleus, where it suffers a deflection, mainly due to the nuclear charge. The interaction of the electron with the atomic nucleus results in a change of its kinetic energy, which results in the emission of a photon in the spectral range of th ...
Heteroepitaxy of GaP on Si(100)*
... odd multiples thereof.7 Fortunately, one APD variant often outgrows the other, so that the APDs remain restricted to regions close to the heterointerface.8 At interfaces between singular Si and III–V faces that are polar, such as $100% or $111%, either faceting into nonpolar JVST B - Microelectronic ...
... odd multiples thereof.7 Fortunately, one APD variant often outgrows the other, so that the APDs remain restricted to regions close to the heterointerface.8 At interfaces between singular Si and III–V faces that are polar, such as $100% or $111%, either faceting into nonpolar JVST B - Microelectronic ...
syntheses, structures, and their interconversion
... within a Teflon inlay which was then placed in a stainless steel autoclave. The autoclave has been tightly closed and heated to 150 °C (Ch = Se) or to 130 °C (Ch = Te) for 7 days and was allowed to slowly cool down to room temperature over 1 day. The products were hand-selected under a light microsc ...
... within a Teflon inlay which was then placed in a stainless steel autoclave. The autoclave has been tightly closed and heated to 150 °C (Ch = Se) or to 130 °C (Ch = Te) for 7 days and was allowed to slowly cool down to room temperature over 1 day. The products were hand-selected under a light microsc ...
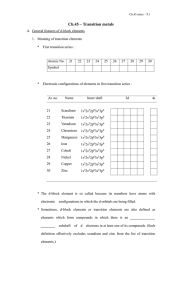
12 U Chem Review
... organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical character ...
... organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical character ...
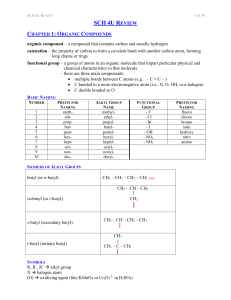
sch4ureview
... organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical character ...
... organic compound – a compound that contains carbon and usually hydrogen catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical character ...
Chemistry I
... "A chemical compound always contains exactly the same proporproportion of elements by mass". E. g. water: oxygen:hydrogen = 8:1 Law of multiple proportions (J. Dalton, 1803): "The proportions of mass of two elements in different compounds are rations of small whole numbers". E.g.: 100 g of carbon re ...
... "A chemical compound always contains exactly the same proporproportion of elements by mass". E. g. water: oxygen:hydrogen = 8:1 Law of multiple proportions (J. Dalton, 1803): "The proportions of mass of two elements in different compounds are rations of small whole numbers". E.g.: 100 g of carbon re ...
Quantum dynamics study of fulvene double bond photoisomerization: The
... the nuclei combined with a trajectory surface hopping algorithm) which showed that different trajectories decay to the ground state at different points on the seam.10 Recently there have been several quantum dynamics studies on two- and three-dimensional surfaces with the goal of separating nuclear ...
... the nuclei combined with a trajectory surface hopping algorithm) which showed that different trajectories decay to the ground state at different points on the seam.10 Recently there have been several quantum dynamics studies on two- and three-dimensional surfaces with the goal of separating nuclear ...
Band Gap Extraction from Individual Two
... further insights into the bonding characteristics, oxidation state of the individual atoms and chemical composition of a single nanosheet. The scattering cross section for core-loss excitations is typically several orders of magnitude less than that for the valence loss excitations;43 hence, to obta ...
... further insights into the bonding characteristics, oxidation state of the individual atoms and chemical composition of a single nanosheet. The scattering cross section for core-loss excitations is typically several orders of magnitude less than that for the valence loss excitations;43 hence, to obta ...
Rutherford backscattering spectrometry
Rutherford backscattering spectrometry (RBS) is an analytical technique used in materials science. Sometimes referred to as high-energy ion scattering (HEIS) spectrometry, RBS is used to determine the structure and composition of materials by measuring the backscattering of a beam of high energy ions (typically protons or alpha particles) impinging on a sample.