technical report 91 -32
... oxidation of the reducing compounds in the experimental system, glove boxes, filled with nitrogen or a mixture of nitrogen and carbon dioxide have to be used. In such a system, the residual atmospheric oxygen concentration is in the range of 1 ppm, corresponding to an oxygen concentration in solutio ...
... oxidation of the reducing compounds in the experimental system, glove boxes, filled with nitrogen or a mixture of nitrogen and carbon dioxide have to be used. In such a system, the residual atmospheric oxygen concentration is in the range of 1 ppm, corresponding to an oxygen concentration in solutio ...
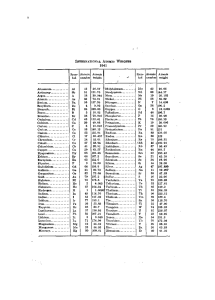
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... PREFACE TO THE THIRD EDITION This revision introduces many new experiments and revises others in an attempt to keep abreast of the rapid developments in physical chemistry. Some of the former experiments have been eliminated or expanded because they have found their way into earlier courses and are ...
... PREFACE TO THE THIRD EDITION This revision introduces many new experiments and revises others in an attempt to keep abreast of the rapid developments in physical chemistry. Some of the former experiments have been eliminated or expanded because they have found their way into earlier courses and are ...
Atmospheric evolution in the Precambrian: Constraints from water
... The correlation of the transitions of the three gases (O2, CO2 and CH4) through ages are emphasized especially in the GOE, because in the GOE, Po2 increased and PCH4 decreased and in the vicinity of the GOE, there were possibly global scale multiple glaciations (e.g., Evans et al., 1997) where green ...
... The correlation of the transitions of the three gases (O2, CO2 and CH4) through ages are emphasized especially in the GOE, because in the GOE, Po2 increased and PCH4 decreased and in the vicinity of the GOE, there were possibly global scale multiple glaciations (e.g., Evans et al., 1997) where green ...
chemistry - Brilliant Public School Sitamarhi
... The well known mineral fluorite is chemically calcium fluoride. It is known that in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell ...
... The well known mineral fluorite is chemically calcium fluoride. It is known that in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell ...
5 Steps to a 5 AP Chemistry, 2008-2009 Edition
... Not too long ago, you enrolled in AP Chemistry. A curiosity about chemistry, encouragement from a respected teacher, or the simple fact that it was a requirement may have been your motivation. No matter what the reason, you find yourself flipping through a book, which promises to help you culminate ...
... Not too long ago, you enrolled in AP Chemistry. A curiosity about chemistry, encouragement from a respected teacher, or the simple fact that it was a requirement may have been your motivation. No matter what the reason, you find yourself flipping through a book, which promises to help you culminate ...
National German Competition and Problems of the IChO
... From hearsay Eileen knows that salty water is particularly healthy. She wants to raise the mass content of chloride in the pool water to 1%. 1 kg of pure salt costs €1.24. The pool has a base area of 5 m x 6 m and is filled up to 1.6 m. The density of the water delivered by the company amounts to 1 ...
... From hearsay Eileen knows that salty water is particularly healthy. She wants to raise the mass content of chloride in the pool water to 1%. 1 kg of pure salt costs €1.24. The pool has a base area of 5 m x 6 m and is filled up to 1.6 m. The density of the water delivered by the company amounts to 1 ...
Sam P. de Visser,* Jan-Uwe Rohde,* Yong
... transfer to substrates, leading to C H hydroxylation, C C epoxidation, aromatic hydroxylation, and heteroatom oxidation [8,9]. The CYP 450s contain a central heme active site that is linked to the protein via a thiolate bridge from a cysteinate residue [10,11]. The catalytic cycle of the CYP 450s st ...
... transfer to substrates, leading to C H hydroxylation, C C epoxidation, aromatic hydroxylation, and heteroatom oxidation [8,9]. The CYP 450s contain a central heme active site that is linked to the protein via a thiolate bridge from a cysteinate residue [10,11]. The catalytic cycle of the CYP 450s st ...
Study Guide for Content Mastery - Student Edition
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
coordination compounds
... oxygen transport to the tissues of the body. Permanent exchange of substances to the environment enables to the body maintain a certain level of concentration of the compounds involved in the equilibrium of the complexation processes, providing metal-ligand homeostasis. In addition, complex compound ...
... oxygen transport to the tissues of the body. Permanent exchange of substances to the environment enables to the body maintain a certain level of concentration of the compounds involved in the equilibrium of the complexation processes, providing metal-ligand homeostasis. In addition, complex compound ...
Modern Chemistry
... the answer as 0.571429. a. Is the setup for calculating density correct? b. How many significant figures should the answer contain? 4. It was shown in the text that in a value such as 4000 g, the precision of the number is uncertain. The zeros may or may not be significant. a. Suppose that the mass ...
... the answer as 0.571429. a. Is the setup for calculating density correct? b. How many significant figures should the answer contain? 4. It was shown in the text that in a value such as 4000 g, the precision of the number is uncertain. The zeros may or may not be significant. a. Suppose that the mass ...
Chemistry (Revised)
... (b) Hydrogen sulfide gas produced from iron(II) sulfide is often contaminated with hydrogen gas and gaseous arsenic compounds. (i) Arsenic(III) sulfide is an orange-yellow powder which is insoluble in water. Below 310 °C it can sublime, turning from a solid to a gas. Name the type of bonding and ...
... (b) Hydrogen sulfide gas produced from iron(II) sulfide is often contaminated with hydrogen gas and gaseous arsenic compounds. (i) Arsenic(III) sulfide is an orange-yellow powder which is insoluble in water. Below 310 °C it can sublime, turning from a solid to a gas. Name the type of bonding and ...
Preparatory Problems of the 40th IChO - IChO-2016
... Identify the substances A-K and write down all the equations 1-14. ...
... Identify the substances A-K and write down all the equations 1-14. ...
2013 - SQA
... (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of iron(II) sulfide reacts with hydrochloric acid. Your diagram should be labelled and should show the names of any ...
... (i) Hydrogen sulfide gas is very soluble in water. Draw a diagram to show an assembled apparatus that could be used to measure the volume of H2S gas produced when a sample of iron(II) sulfide reacts with hydrochloric acid. Your diagram should be labelled and should show the names of any ...
Experiment 7: Determination of the concentration of a solution of an
... All results sheets for a particular laboratory must be handed in at the end of that session; students who do not do so will be deemed to have been absent, with possible subsequent DP implications. ...
... All results sheets for a particular laboratory must be handed in at the end of that session; students who do not do so will be deemed to have been absent, with possible subsequent DP implications. ...
chapter 5 gases
... same whether the reaction takes place in one step or in a series of steps. The law enables us to determine the standard enthalpy of formation of a compound from its elements by an indirect route when direct combination of the elements is not feasible. Using Hess’s law, the standard enthalpy change f ...
... same whether the reaction takes place in one step or in a series of steps. The law enables us to determine the standard enthalpy of formation of a compound from its elements by an indirect route when direct combination of the elements is not feasible. Using Hess’s law, the standard enthalpy change f ...
2nd Semester Practice Chemistry Final 2009
... 14. An ideal gas is an imaginary gas a. not made of particles. b. that conforms to all of the assumptions of the kinetic theory. c. whose particles have zero mass. d. made of motionless particles. 15. An NaOH solution contains 1.90 mol of NaOH, and its concentration is 0.555 M. What is its volume? ...
... 14. An ideal gas is an imaginary gas a. not made of particles. b. that conforms to all of the assumptions of the kinetic theory. c. whose particles have zero mass. d. made of motionless particles. 15. An NaOH solution contains 1.90 mol of NaOH, and its concentration is 0.555 M. What is its volume? ...
General and Inorganic Chemistry – Laboratory Techniques
... Knowledge of students on Chemistry at the beginning of their graduate studies is rather different. Most of the students do not have proper laboratory expertise. This educational experience prompted the faculty of the institute to compile an educational material that can help students to make themsel ...
... Knowledge of students on Chemistry at the beginning of their graduate studies is rather different. Most of the students do not have proper laboratory expertise. This educational experience prompted the faculty of the institute to compile an educational material that can help students to make themsel ...
Stoichiometric Calculations
... Example: When 10 grams of hydrogen react with 3.4 moles of nitrogen gas to make ammonia, which substance would be the limiting reactant? N2(g) + 3H2(g) --> 2NH3(g) Step 1: Convert all values to moles. 10 g H2 x 1 mol H2 = 5 mol H2 2 g H2 Initial Amounts = 5 mol H2, 3.4 mol N2 ...
... Example: When 10 grams of hydrogen react with 3.4 moles of nitrogen gas to make ammonia, which substance would be the limiting reactant? N2(g) + 3H2(g) --> 2NH3(g) Step 1: Convert all values to moles. 10 g H2 x 1 mol H2 = 5 mol H2 2 g H2 Initial Amounts = 5 mol H2, 3.4 mol N2 ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.