Multiple Choice Exam Review June 2016
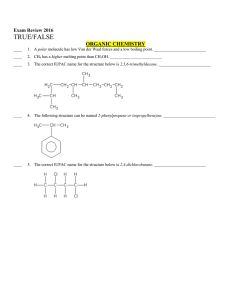
... ____ 34. Catalysts can be used to speed up a reaction. _________________________ ____ 35. The value of the rate constant, k, is valid only for a specific reaction at a specific temperature. _________________________ ____ 36. An ineffective collision is one that has sufficient energy and correct orie ...
... ____ 34. Catalysts can be used to speed up a reaction. _________________________ ____ 35. The value of the rate constant, k, is valid only for a specific reaction at a specific temperature. _________________________ ____ 36. An ineffective collision is one that has sufficient energy and correct orie ...
SCH4U TEXT BOOK
... foods that have been grown without the use of pesticides, herbicides, fertilizers, hormones, and other synthetic chemicals. The original meaning of the word “organic” refers to anything that is or has been alive. In this sense, all vegetables are organic, no matter how they are grown. Organic chemis ...
... foods that have been grown without the use of pesticides, herbicides, fertilizers, hormones, and other synthetic chemicals. The original meaning of the word “organic” refers to anything that is or has been alive. In this sense, all vegetables are organic, no matter how they are grown. Organic chemis ...
Transition Metal-Modified Zirconium Phosphate Electrocatalysts for
... precious metals [15,16]. Significant research has been conducted to improve electrocatalysts through based on precious metals [15,16]. Significant research has been conducted to improve electrocatalysts a number of general strategies, including increasing the number of active sites, increasing the ...
... precious metals [15,16]. Significant research has been conducted to improve electrocatalysts through based on precious metals [15,16]. Significant research has been conducted to improve electrocatalysts a number of general strategies, including increasing the number of active sites, increasing the ...
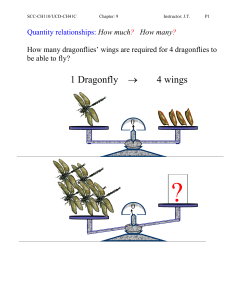
Quantity relationships: How much
... 1.85 × 17.03 = 31.51 g NH3 left A mixture of 5.0 g of H2 (g) and 10.0 g of O2(g) is ignited. Water forms according to the following combination reaction: 2H2(g) +O2(g) → 2H2O(g) Which reactant is limiting? How much water will the reaction produce? ...
... 1.85 × 17.03 = 31.51 g NH3 left A mixture of 5.0 g of H2 (g) and 10.0 g of O2(g) is ignited. Water forms according to the following combination reaction: 2H2(g) +O2(g) → 2H2O(g) Which reactant is limiting? How much water will the reaction produce? ...
Chemistry Final Exam Review
... ____ 10. To convert to molecules you use Avogadro's number. ____ 11. A binary compound is composed of only two elements. ____ 12. Avogadro's number is 2.066 x 1023. ____ 13. Empirical formulas are also called the simplest formula. ____ 14. Empirical formulas show the exact number of each element con ...
... ____ 10. To convert to molecules you use Avogadro's number. ____ 11. A binary compound is composed of only two elements. ____ 12. Avogadro's number is 2.066 x 1023. ____ 13. Empirical formulas are also called the simplest formula. ____ 14. Empirical formulas show the exact number of each element con ...
Chemistry Appendixes
... Scientists ask questions and seek concepts to answer these questions by applying consistent, logical reasoning to describe, explain, and predict observations, and by doing experiments to test hypotheses or predictions from these hypotheses. In this way science progresses using a general model for so ...
... Scientists ask questions and seek concepts to answer these questions by applying consistent, logical reasoning to describe, explain, and predict observations, and by doing experiments to test hypotheses or predictions from these hypotheses. In this way science progresses using a general model for so ...
CHAPTER 21 ELECTROCHEMISTRY: CHEMICAL CHANGE AND
... b) The metal ion B is gaining electrons to form the metal B, so it is displaced. c) The anode is the electrode at which oxidation takes place, so metal A is used as the anode. d) Acid oxidizes metal B and metal B oxidizes metal A, so acid will oxidize metal A and bubbles will form when metal A is pl ...
... b) The metal ion B is gaining electrons to form the metal B, so it is displaced. c) The anode is the electrode at which oxidation takes place, so metal A is used as the anode. d) Acid oxidizes metal B and metal B oxidizes metal A, so acid will oxidize metal A and bubbles will form when metal A is pl ...
Determination of Equilibrium Constants for Reactions between Nitric
... cobalt(II) ammonia system, only penta- and hexaamminecobalt(II) ions were referred to as reactants that react with NO to form nitrosyl by Ford and Lorkovic,20 Asmussen et al.,21 and Gans.22 Besides, it has been reported by Simplicio and Wilkins18 that penta- and hexaamminecobalt(II) nitrates were th ...
... cobalt(II) ammonia system, only penta- and hexaamminecobalt(II) ions were referred to as reactants that react with NO to form nitrosyl by Ford and Lorkovic,20 Asmussen et al.,21 and Gans.22 Besides, it has been reported by Simplicio and Wilkins18 that penta- and hexaamminecobalt(II) nitrates were th ...
Section 1
... We can picture the hydrogen atom — the simplest of all atoms with one electron and one proton in the nucleus — by considering a pea placed in the centre of a football pitch, to represent the nucleus with its proton. On this scale the electron will revolve in a circular orbit round the goalposts. Bet ...
... We can picture the hydrogen atom — the simplest of all atoms with one electron and one proton in the nucleus — by considering a pea placed in the centre of a football pitch, to represent the nucleus with its proton. On this scale the electron will revolve in a circular orbit round the goalposts. Bet ...
Ozone decomposition
... The proposed mechanism leads to formation of oxygen atoms in ground and excited state. This cycle of decomposition is called the cycle of “residual oxygen”. The photodissociation of ozone (11, 12) takes place under the action of solar radiation with a wavelength of less than 1134 nm. Decomposition o ...
... The proposed mechanism leads to formation of oxygen atoms in ground and excited state. This cycle of decomposition is called the cycle of “residual oxygen”. The photodissociation of ozone (11, 12) takes place under the action of solar radiation with a wavelength of less than 1134 nm. Decomposition o ...
HOTS Worksheet
... Ans. (i) Glucose does not give Schiff‘s Test although it contains aldehyde group. (ii) Glucose does not form crystaline product with NaHSO3. Q. 6. B-complex is an often prescribed Vitamin. What is complex about it ? What is its usefulness ? Ans. It is a type of Vitamin which contains B1, B2, B6 and ...
... Ans. (i) Glucose does not give Schiff‘s Test although it contains aldehyde group. (ii) Glucose does not form crystaline product with NaHSO3. Q. 6. B-complex is an often prescribed Vitamin. What is complex about it ? What is its usefulness ? Ans. It is a type of Vitamin which contains B1, B2, B6 and ...
Improved Synthesis of Seven-Coordinate Molybdenum( I I) and
... [W(CN-t-C4H&I]I. A solution of 0.234 g (0.92 "01) of iodine dissolved in 10 mL of methanol was added dropwise with stirring to a solution containing 0.484 g (0.94 mmol) of W(CN-tc4&)&0)3 dissolved in 25 mL of methanol. After the addition was complete, 0.26 g (3.1 "01) of tert-butyl isocyanidewas add ...
... [W(CN-t-C4H&I]I. A solution of 0.234 g (0.92 "01) of iodine dissolved in 10 mL of methanol was added dropwise with stirring to a solution containing 0.484 g (0.94 mmol) of W(CN-tc4&)&0)3 dissolved in 25 mL of methanol. After the addition was complete, 0.26 g (3.1 "01) of tert-butyl isocyanidewas add ...
1.09 MB / 64 pages
... pairs on the oxygen of ethanol, and another H bond between the –OH group’s hydrogen and a nonbonding electron pair on the oxygen in water. The water molecules have other nonbonding pairs on oxygen and covalently bonded hydrogen, all of which are capable of extending the network of hydrogen bonds. In ...
... pairs on the oxygen of ethanol, and another H bond between the –OH group’s hydrogen and a nonbonding electron pair on the oxygen in water. The water molecules have other nonbonding pairs on oxygen and covalently bonded hydrogen, all of which are capable of extending the network of hydrogen bonds. In ...
Section 1.3 - The Student Room
... The formation of a compound from its elements may be an exothermic reaction (DHf negative) or an endothermic reaction (DHf positive). However, energy is liberated whenever a substance burns, so combustion reactions are always exothermic (DHc negative). ...
... The formation of a compound from its elements may be an exothermic reaction (DHf negative) or an endothermic reaction (DHf positive). However, energy is liberated whenever a substance burns, so combustion reactions are always exothermic (DHc negative). ...
9278654 PS/Chemistry Ja03 - Dolgeville Central School
... 5 Which event must always occur for a chemical reaction to take place? (1) formation of a precipitate (2) formation of a gas (3) effective collisions between reacting particles (4) addition of a catalyst to the reaction system P.S./Chem.–Jan. ’03 ...
... 5 Which event must always occur for a chemical reaction to take place? (1) formation of a precipitate (2) formation of a gas (3) effective collisions between reacting particles (4) addition of a catalyst to the reaction system P.S./Chem.–Jan. ’03 ...
5 Steps
... Copyright © 2010, 2008, 2004 by The McGraw-Hill Companies, Inc. All rights reserved. Except as permitted under the United States Copyright Act of 1976, no part of this publication may be reproduced or distributed in any form or by any means, or stored in a database or retrieval system, without the ...
... Copyright © 2010, 2008, 2004 by The McGraw-Hill Companies, Inc. All rights reserved. Except as permitted under the United States Copyright Act of 1976, no part of this publication may be reproduced or distributed in any form or by any means, or stored in a database or retrieval system, without the ...
Water splitting

Water splitting is the general term for a chemical reaction in which water is separated into oxygen and hydrogen. Efficient and economical water splitting would be a key technology component of a hydrogen economy. Various techniques for water splitting have been issued in water splitting patents in the United States. In photosynthesis, water splitting donates electrons to power the electron transport chain in photosystem II.