Basics of Semiconductors_1
... Depending on the doping material the Fermi level tends to shift like for intrinsic semiconductor it is exactly in the center of the conduction and valence band For n type it shift towards conduction as electrons are in abundance so probability actually shifts towards conduction band On contrary for ...
... Depending on the doping material the Fermi level tends to shift like for intrinsic semiconductor it is exactly in the center of the conduction and valence band For n type it shift towards conduction as electrons are in abundance so probability actually shifts towards conduction band On contrary for ...
File - Mr. Gittermann
... • Electrons: Subatomic particle with a negative charge found in a certain region of space around the nucleus called the electron cloud; kept close to the atom due to the attraction between the opposite charges of the electron and proton ...
... • Electrons: Subatomic particle with a negative charge found in a certain region of space around the nucleus called the electron cloud; kept close to the atom due to the attraction between the opposite charges of the electron and proton ...
Periodic Table Jeopardy
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...
... A substance that cannot be separated or broken down into simpler substances by chemical means. All atoms in this substance have the same atomic #. ...
Define:
... Chemistry Fall Final Exam Review Define: 1. solid 2. liquid 3. gas 4. chemistry 5. heterogenous mixture 6. homogeneous mixture 7. element 8. compound 9. law of conservation of mass 10. atom 11. atomic number 12. mass number 13. isotope 14. atomic mass 15. Hund’s Rule 16. Heisenburg uncertainty princ ...
... Chemistry Fall Final Exam Review Define: 1. solid 2. liquid 3. gas 4. chemistry 5. heterogenous mixture 6. homogeneous mixture 7. element 8. compound 9. law of conservation of mass 10. atom 11. atomic number 12. mass number 13. isotope 14. atomic mass 15. Hund’s Rule 16. Heisenburg uncertainty princ ...
Scott Lascelle Bill Davis Nanomaterials Workshop University of
... gases is then introduced, providing the atoms needed for the coatings. We did not use this method in our research. CHARACTERIZING After these processes had been completed, careful analysis was needed to confirm or deny our success. Several methods were employed, each contributing its own special dat ...
... gases is then introduced, providing the atoms needed for the coatings. We did not use this method in our research. CHARACTERIZING After these processes had been completed, careful analysis was needed to confirm or deny our success. Several methods were employed, each contributing its own special dat ...
Electron
... • Protons are positive • Neutrons are neutral • The # of Protons and Electrons determine the charge of the atom. ...
... • Protons are positive • Neutrons are neutral • The # of Protons and Electrons determine the charge of the atom. ...
Laser Refraction and Diffraction
... bands are formed when the wave satisfies Formula 3. Wavefronts in the plane wave are parallel to each other. A ray composed of this type of wave is either highly directional or possesses a small divergence angle. a sin θ = mλ , m = intengers ...
... bands are formed when the wave satisfies Formula 3. Wavefronts in the plane wave are parallel to each other. A ray composed of this type of wave is either highly directional or possesses a small divergence angle. a sin θ = mλ , m = intengers ...
ChemicalBondingTestAnswers
... The charge on the nucleus – On moving from left to right, the effective nuclear charge increases. The distance of the electron from the nucleus – On moving from left to right in a period, the distance of the electron from the nucleus decreases a little . The number of electrons between the outer ele ...
... The charge on the nucleus – On moving from left to right, the effective nuclear charge increases. The distance of the electron from the nucleus – On moving from left to right in a period, the distance of the electron from the nucleus decreases a little . The number of electrons between the outer ele ...
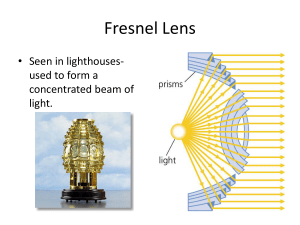
Fresnel Lens
... packets of energy, called photons. • Not a new idea, he was building upon an idea proposed by Planck, that light came in discrete packets. (in fact, Newton proposed a particle like explanation of light centuries earlier). The problem for Planck was his discrete packets were in conflict with the wave ...
... packets of energy, called photons. • Not a new idea, he was building upon an idea proposed by Planck, that light came in discrete packets. (in fact, Newton proposed a particle like explanation of light centuries earlier). The problem for Planck was his discrete packets were in conflict with the wave ...
Molecular Geometry Why?
... is based on the premise that electrons around a central atom repel each other. Electron domains are areas of high electron density such as bonds (single, double or triple) and lone-pairs of electrons. In simple terms VSEPR means that all electron bonding domains and electron nonbonding domains aroun ...
... is based on the premise that electrons around a central atom repel each other. Electron domains are areas of high electron density such as bonds (single, double or triple) and lone-pairs of electrons. In simple terms VSEPR means that all electron bonding domains and electron nonbonding domains aroun ...
Unit 2 Practice Exam exam_2p_08_matter
... 42. Why do atomic radii increase dramatically with each additional row of the periodic table? a. atomic nuclei become increasingly attractive as more protons are added. b. another energy level is utilized by the electrons. c. the energy required to remove an electron is reduced by shielding of inter ...
... 42. Why do atomic radii increase dramatically with each additional row of the periodic table? a. atomic nuclei become increasingly attractive as more protons are added. b. another energy level is utilized by the electrons. c. the energy required to remove an electron is reduced by shielding of inter ...
Chapter-2 - Shodhganga
... are used to modulate the intensity of the CRT then a channeling pattern is obtained. Backscattered electron patterns can be obtained by setting the specimen so that the electrons strike the surface at a glancing angle. Electrons, which are scattered through less than about 90 and subsequently, Bragg ...
... are used to modulate the intensity of the CRT then a channeling pattern is obtained. Backscattered electron patterns can be obtained by setting the specimen so that the electrons strike the surface at a glancing angle. Electrons, which are scattered through less than about 90 and subsequently, Bragg ...
Computer simulated thermal energy atomic
... wave function is 5% in both directions (Fig. 2A). Using the same atomic beam the lattice constant is increased in the model computations for exploring the eect on the scattering due to the relation between transfer width and the lattice constant. The resolution is low when the transfer width become ...
... wave function is 5% in both directions (Fig. 2A). Using the same atomic beam the lattice constant is increased in the model computations for exploring the eect on the scattering due to the relation between transfer width and the lattice constant. The resolution is low when the transfer width become ...
CHAPTER 5 NOTES – ELECTRONS IN ATOMS
... • c = λν • The wavelength and frequency of light are inversely proportional to each other • c = speed of light (3E8 m/s or 3 E10 cm/s) • When atoms absorb energy, electrons move into higher energy levels, and these electrons lose energy by emitting light when they return to lower energy levels. ...
... • c = λν • The wavelength and frequency of light are inversely proportional to each other • c = speed of light (3E8 m/s or 3 E10 cm/s) • When atoms absorb energy, electrons move into higher energy levels, and these electrons lose energy by emitting light when they return to lower energy levels. ...
Semester Exam Practice Questions
... a. shape of the electron cloud c. spin of the electron b. size of the electron cloud d. intensity of charge of the electron cloud 19. In the wave-mechanical view of atomic structure, the pathway or position of an electron is best represented as __________. a. a circular orbit with a specific diamete ...
... a. shape of the electron cloud c. spin of the electron b. size of the electron cloud d. intensity of charge of the electron cloud 19. In the wave-mechanical view of atomic structure, the pathway or position of an electron is best represented as __________. a. a circular orbit with a specific diamete ...
1 - M*W
... gas. Which of the following represents the reactants in this reaction? a) Magnesium and magnesium chloride b) Hydrochloric acid and hydrogen gas c) Magnesium and hydrochloric acid d) Magnesium chloride and hydrogen gas 50) The equation E=mc2 shows that a) Chemical reactions are either exothermic or ...
... gas. Which of the following represents the reactants in this reaction? a) Magnesium and magnesium chloride b) Hydrochloric acid and hydrogen gas c) Magnesium and hydrochloric acid d) Magnesium chloride and hydrogen gas 50) The equation E=mc2 shows that a) Chemical reactions are either exothermic or ...
4 - College of Arts and Sciences
... Formula Of Reactants and Products • IONIC – ONLY Ions Are Shown • NET IONIC – SPECTATOR Ions Are Removed From Ionic Equation • SPECTATOR Ions – Ions that do not take part in the chemical reaction ...
... Formula Of Reactants and Products • IONIC – ONLY Ions Are Shown • NET IONIC – SPECTATOR Ions Are Removed From Ionic Equation • SPECTATOR Ions – Ions that do not take part in the chemical reaction ...
document
... breaks down into its parts. 2. Ion C B. A bond that is formed by sharing electrons. 3. Charge D C. A charged atom. D. The number of electrons an 4. Covalent Bond B element is willing to gain, lose, or share to form compounds. 5. Ionic Bond I E. States that all elements want either a full outer shell ...
... breaks down into its parts. 2. Ion C B. A bond that is formed by sharing electrons. 3. Charge D C. A charged atom. D. The number of electrons an 4. Covalent Bond B element is willing to gain, lose, or share to form compounds. 5. Ionic Bond I E. States that all elements want either a full outer shell ...
Characterization techniques for nanotechnology
... products. The precise control of nanoparticle size, size distribution, dispersion at nanolevel and deposition on textile substrate needs sophisticated characterization techniques, such as particle size analyzer, electron microscopy (SEM/TEM/HRTEM), atomic force microscopy, X-ray diffraction, Raman s ...
... products. The precise control of nanoparticle size, size distribution, dispersion at nanolevel and deposition on textile substrate needs sophisticated characterization techniques, such as particle size analyzer, electron microscopy (SEM/TEM/HRTEM), atomic force microscopy, X-ray diffraction, Raman s ...
Dr. Dagotto Explains Recent Surface State Discovery in Nature
... Santander-Syro and colleagues’ results is that a new conducting, two-dimensional electron system on a familiar and popular substrate is now available by simply cleaving it — or, more generally, by producing oxygen vacancies right at its surface. This simple approach is an attractive alternative to t ...
... Santander-Syro and colleagues’ results is that a new conducting, two-dimensional electron system on a familiar and popular substrate is now available by simply cleaving it — or, more generally, by producing oxygen vacancies right at its surface. This simple approach is an attractive alternative to t ...
PROJECT TEM
... For a 200 kV microscope, with partly corrected spherical aberrations ("to the third order") and a Cs value of 1 µm, a theoretical cut-off value might be 1/qmax = 42 pm. The same microscope without a corrector would have Cs = 0.5 mm and thus a 200-pm cut-off Practically, the spherical aberrations are ...
... For a 200 kV microscope, with partly corrected spherical aberrations ("to the third order") and a Cs value of 1 µm, a theoretical cut-off value might be 1/qmax = 42 pm. The same microscope without a corrector would have Cs = 0.5 mm and thus a 200-pm cut-off Practically, the spherical aberrations are ...
Modulation Transfer Function
... where C(s) is the object contrast and C 0 (s0 ) the image contrast. A value of 1 means that frequency is perfectly transferred to the image plane, which is the best possible outcome in a passive system. A value of 0 means that frequency will not appear at all in the image. The object- and image-spac ...
... where C(s) is the object contrast and C 0 (s0 ) the image contrast. A value of 1 means that frequency is perfectly transferred to the image plane, which is the best possible outcome in a passive system. A value of 0 means that frequency will not appear at all in the image. The object- and image-spac ...
Chemistry ~ Fall Final Review
... 7. Explain how to determine the # of protons, neutrons and electrons from given information about atomic mass, atomic number, and the charge on the atom (ion). ...
... 7. Explain how to determine the # of protons, neutrons and electrons from given information about atomic mass, atomic number, and the charge on the atom (ion). ...
Surface contribution to giant magnetoresistance in Fe/Cr/Fe films K. W
... tion to GMR coming from surfaces, interfaces as well as from inner layers and to compare their influence on the total GMR of the Fe/Cr/Fe sample. Our considerations are based on the relation between the current of electrons moving in the sample in plane of its surface under the influence of an elect ...
... tion to GMR coming from surfaces, interfaces as well as from inner layers and to compare their influence on the total GMR of the Fe/Cr/Fe sample. Our considerations are based on the relation between the current of electrons moving in the sample in plane of its surface under the influence of an elect ...
Low-energy electron diffraction

Low-energy electron diffraction (LEED) is a technique for the determination of the surface structure of single-crystalline materials by bombardment with a collimated beam of low energy electrons (20–200 eV) and observation of diffracted electrons as spots on a fluorescent screen.LEED may be used in one of two ways: Qualitatively, where the diffraction pattern is recorded and analysis of the spot positions gives information on the symmetry of the surface structure. In the presence of an adsorbate the qualitative analysis may reveal information about the size and rotational alignment of the adsorbate unit cell with respect to the substrate unit cell. Quantitatively, where the intensities of diffracted beams are recorded as a function of incident electron beam energy to generate the so-called I-V curves. By comparison with theoretical curves, these may provide accurate information on atomic positions on the surface at hand.↑