AC Circuits
... In this part of the experiment, you need to measure the absorption spectrum of the Rose Bengal solution at different concentrations (Rose Bengal is an organic dye). Prepare a master aqueous solution of molar concentration ~1.25x10-4 M (moles/l) in a large storage vial. Use the plastic pipettes to tr ...
... In this part of the experiment, you need to measure the absorption spectrum of the Rose Bengal solution at different concentrations (Rose Bengal is an organic dye). Prepare a master aqueous solution of molar concentration ~1.25x10-4 M (moles/l) in a large storage vial. Use the plastic pipettes to tr ...
absorbance, a - srmbiotech25
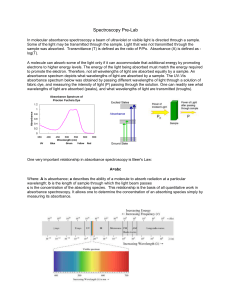
... shifts in the position of a chemical or physical equilibrium involving the absorbing species. • A common example of this behavior is found with acid/base indicators. • Deviations arising from chemical factors can only be observed when concentrations are changed. eg:dichromate ions on dilution. Cr2O7 ...
... shifts in the position of a chemical or physical equilibrium involving the absorbing species. • A common example of this behavior is found with acid/base indicators. • Deviations arising from chemical factors can only be observed when concentrations are changed. eg:dichromate ions on dilution. Cr2O7 ...
Fourier Transform IR Spectroscopy
... • FT – IR can take wavelength readings across the whole IR region simultaneously and smoothly, making this a very rapid technique. • The technique is non-invasive and non-destructive. Its resolution of .125 cm-1 is not spectacular in comparison to other vibrational techniques and it will not give th ...
... • FT – IR can take wavelength readings across the whole IR region simultaneously and smoothly, making this a very rapid technique. • The technique is non-invasive and non-destructive. Its resolution of .125 cm-1 is not spectacular in comparison to other vibrational techniques and it will not give th ...
Spectrophotometry and its Applications in Microbiology
... Certain covalent bonds in molecules are able to absorb energy at particular wavelengths ranging from infrared to ultraviolet. This absorbance is readily detected by using some kind of spectrometer which sends light of a specific wavelength through the sample; if the chemical absorbs energy, then the ...
... Certain covalent bonds in molecules are able to absorb energy at particular wavelengths ranging from infrared to ultraviolet. This absorbance is readily detected by using some kind of spectrometer which sends light of a specific wavelength through the sample; if the chemical absorbs energy, then the ...
Info Note 804: UV-VIS Nomenclature and Units
... A spectrophotometer can be either single beam or double beam. In a single beam instrument, all of the light passes through the sample cell. Io must be measured by removing the sample. This was the earliest design, but is still in common use in both teaching and industrial labs. Samples for UV/Vis sp ...
... A spectrophotometer can be either single beam or double beam. In a single beam instrument, all of the light passes through the sample cell. Io must be measured by removing the sample. This was the earliest design, but is still in common use in both teaching and industrial labs. Samples for UV/Vis sp ...
Ultraviolet–visible spectroscopy

Ultraviolet–visible spectroscopy or ultraviolet-visible spectrophotometry (UV-Vis or UV/Vis) refers to absorption spectroscopy or reflectance spectroscopy in the ultraviolet-visible spectral region. This means it uses light in the visible and adjacent (near-UV and near-infrared [NIR]) ranges. The absorption or reflectance in the visible range directly affects the perceived color of the chemicals involved. In this region of the electromagnetic spectrum, molecules undergo electronic transitions. This technique is complementary to fluorescence spectroscopy, in that fluorescence deals with transitions from the excited state to the ground state, while absorption measures transitions from the ground state to the excited state.