GCE Chemistry SAMs 2009 onwards pdf
... Here are his results Mass of ethanol before experiment Mass of ethanol after experiment Mass of water Temperature of water before experiment Temperature of water after experiment ...
... Here are his results Mass of ethanol before experiment Mass of ethanol after experiment Mass of water Temperature of water before experiment Temperature of water after experiment ...
Practical Assessment of Sanitizers Steve Gray November
... Especially effective on Biofilms Note - it has been shown that bacteria within a Biofilm are up to 1,000x’s more resistant to some sanitizers ...
... Especially effective on Biofilms Note - it has been shown that bacteria within a Biofilm are up to 1,000x’s more resistant to some sanitizers ...
Appendix
... Fireworks are made from pyrotechnics—chemical substances that produce light and smoke when they are ignited. Pyrotechnics are also used in flares, smoke bombs, explosives, and matches. An aerial fireworks device is a rocket made of a cylinder, chemicals inside the cylinder, and fuses attached to the ...
... Fireworks are made from pyrotechnics—chemical substances that produce light and smoke when they are ignited. Pyrotechnics are also used in flares, smoke bombs, explosives, and matches. An aerial fireworks device is a rocket made of a cylinder, chemicals inside the cylinder, and fuses attached to the ...
Chem13-14PrecipABNeut
... The focus of these lessons is to provide methods will help you to solve numeric calculations in first-year chemistry. This is only one part of a course in chemistry, but it is often the most challenging. Problem Notebook: The purchase of a spiral problem notebook is suggested as a place to write you ...
... The focus of these lessons is to provide methods will help you to solve numeric calculations in first-year chemistry. This is only one part of a course in chemistry, but it is often the most challenging. Problem Notebook: The purchase of a spiral problem notebook is suggested as a place to write you ...
General and Inorganic Chemistry
... hydrolysis of acetylsalicylic acid (ASA) ................................................................................. 77 3. VI.3 Calculations ................................................................................................................ 78 7. VII Chemical equilibrium ......... ...
... hydrolysis of acetylsalicylic acid (ASA) ................................................................................. 77 3. VI.3 Calculations ................................................................................................................ 78 7. VII Chemical equilibrium ......... ...
Conceptual Integrated Science—Chapter 13
... B. Nitrogen oxide and nitrogen dioxide from the atmosphere are pulled into the combustion chamber prior to exiting the engine through the exhaust. C. Atmospheric nitrogen and oxygen react with each other within the combustion chamber. D. According to the principle of the conservation of mass, this i ...
... B. Nitrogen oxide and nitrogen dioxide from the atmosphere are pulled into the combustion chamber prior to exiting the engine through the exhaust. C. Atmospheric nitrogen and oxygen react with each other within the combustion chamber. D. According to the principle of the conservation of mass, this i ...
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... directly in your final report as you obtain it. (Data entered on scraps of paper will be confiscated.) Where calculations of data are involved, show an orderly calculation for the first set of data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answe ...
... directly in your final report as you obtain it. (Data entered on scraps of paper will be confiscated.) Where calculations of data are involved, show an orderly calculation for the first set of data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answe ...
File - cpprashanths Chemistry
... Co-ordination compounds impart colour due to the presence of unpaired electrons in the central metal atom or ion. 1/2M The unpaired electrons show d-d transition by jumping between t2g and eg orbitals. 1/2M Q3. Write the structures of the each of the following alcohols. 1)3,3-dimethyl cyclopentanol ...
... Co-ordination compounds impart colour due to the presence of unpaired electrons in the central metal atom or ion. 1/2M The unpaired electrons show d-d transition by jumping between t2g and eg orbitals. 1/2M Q3. Write the structures of the each of the following alcohols. 1)3,3-dimethyl cyclopentanol ...
Complete Set
... dissolve all of the calcium hydroxide? Assume the volume of the nitric acid is small and can be ignored in the calculation of the total volume. If all of the Ca(OH)2 dissolves then [Ca2+(aq)] = 0.0270 M. The [OH(aq)] required to achieve this is given by: Ksp = [Ca2+(aq)][OH-(aq)]2 = (0.0270) × [OH-( ...
... dissolve all of the calcium hydroxide? Assume the volume of the nitric acid is small and can be ignored in the calculation of the total volume. If all of the Ca(OH)2 dissolves then [Ca2+(aq)] = 0.0270 M. The [OH(aq)] required to achieve this is given by: Ksp = [Ca2+(aq)][OH-(aq)]2 = (0.0270) × [OH-( ...
BARIUM NITRATE
... its oxide. In nature, beryllium occurs in several minerals, mostly combined with silica and alumina. The most common minerals are beryl, 3BeO•Al2O3•6SiO2; chrysoberyl, BeO•Al2O3; phenacite, 2BeO•SiO2; and bertrandite, 4BeO•2SiO2•H2O. Also, it is found in trace amounts in the ore feldspar, and in vol ...
... its oxide. In nature, beryllium occurs in several minerals, mostly combined with silica and alumina. The most common minerals are beryl, 3BeO•Al2O3•6SiO2; chrysoberyl, BeO•Al2O3; phenacite, 2BeO•SiO2; and bertrandite, 4BeO•2SiO2•H2O. Also, it is found in trace amounts in the ore feldspar, and in vol ...
Problem Authors - PianetaChimica
... Preparatory Problems, Theoretical contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, prec ...
... Preparatory Problems, Theoretical contaminations. The reaction of sulfuric acid with colemanite takes place in two steps: In the first step colemanite is dissolved in sulfuric acid forming the calcium(II) ion and boric acid. In the second step, calcium sulfate, formed from Ca2+ and SO42− ions, prec ...
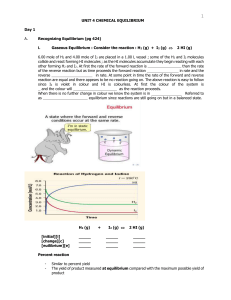
Unit 4 - Chemical Equilibrium
... add an inert gas so that the pressure remains constant - for pressure to remain constant the volume must ______________ ; this will __________ the partial pressure of the reactants and products ; stress is ____________ to relieve stress equ. shifts to the ______________ since there are ________ mole ...
... add an inert gas so that the pressure remains constant - for pressure to remain constant the volume must ______________ ; this will __________ the partial pressure of the reactants and products ; stress is ____________ to relieve stress equ. shifts to the ______________ since there are ________ mole ...
Final Review 3-8 Answers_2
... 14. The different shapes of balloons are possible because air is a gas that a) has neither a fixed shape nor volume b) is highly compressible c) diffused readily d) mixes easily with other gases 15. What volume is occupied by 0.35 mol of helium at SATP? a) 71 L b) 8.7 L c) 0.35 L d) 0.014 L 16. In ...
... 14. The different shapes of balloons are possible because air is a gas that a) has neither a fixed shape nor volume b) is highly compressible c) diffused readily d) mixes easily with other gases 15. What volume is occupied by 0.35 mol of helium at SATP? a) 71 L b) 8.7 L c) 0.35 L d) 0.014 L 16. In ...
equilibrium - eVirtualGuru
... equilibrium constants; • establish a relationship between Kp and K c; • explain various factors that affect the equilibrium state of a reaction; • classify substances as acids or bases according to Arrhenius, Bronsted-Lowry and Lewis concepts; • classify acids and bases as weak or strong in terms of ...
... equilibrium constants; • establish a relationship between Kp and K c; • explain various factors that affect the equilibrium state of a reaction; • classify substances as acids or bases according to Arrhenius, Bronsted-Lowry and Lewis concepts; • classify acids and bases as weak or strong in terms of ...
Now! - Soojeede.com
... Let’s look at citric acid. Citric acid, C6H8O7, is commonly found in everyday products like lemons and limes, and even soft drinks. It is the substance responsible for making the sour taste of these foods and drinks. Citric acid only ionizes a little more than 3% and is therefore classified as a wea ...
... Let’s look at citric acid. Citric acid, C6H8O7, is commonly found in everyday products like lemons and limes, and even soft drinks. It is the substance responsible for making the sour taste of these foods and drinks. Citric acid only ionizes a little more than 3% and is therefore classified as a wea ...
LABORATORY MANUAL FOR CHEMISTRY 102
... the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum v ...
... the experimentally determined exponents for each species. (The overall order of the reaction is equal to the sum of x + y + z +... .) The term k is known as the rate constant for the reaction. Usually, when a reaction is initiated, the rate (known as the initial rate) is found to be at its maximum v ...
AP Chemistry Notes and Worksheets 2014
... o It stated that when two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole ...
... o It stated that when two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole ...
A Dictionary of the New Chymical Nomenclature
... Benzoat of alumine Benzoat of ammoniac Benzoat of antimony Benzoat of arsenic Benzoat of barytes Benzoat of bismuth Benzoat of cobalt Benzoat of copper Benzoat of gold Benzoat of iron Benzoat of lead Benzoat of lime Benzoat of magnesia Benzoat of manganese [p. 115] Benzoat of mercury Benzoat of moly ...
... Benzoat of alumine Benzoat of ammoniac Benzoat of antimony Benzoat of arsenic Benzoat of barytes Benzoat of bismuth Benzoat of cobalt Benzoat of copper Benzoat of gold Benzoat of iron Benzoat of lead Benzoat of lime Benzoat of magnesia Benzoat of manganese [p. 115] Benzoat of mercury Benzoat of moly ...
teaching and learning materials - UNESDOC
... INTRODUCTION All over the world, science educators declare that practical experiences are an essential part of learning science. However, in many countries these experiences are not provided in the majority of their primary and secondary schools. There are several reasons for this: cost, safety, wa ...
... INTRODUCTION All over the world, science educators declare that practical experiences are an essential part of learning science. However, in many countries these experiences are not provided in the majority of their primary and secondary schools. There are several reasons for this: cost, safety, wa ...
5. Coenzyme HAD+ is derived
... training. Chemistry is a basic science and a powerful tool for studying and learning processes in living systems. Therefore, medical students must thoroughly understand the basic ideas, laws and methods of this science. Program expected to consider the foundations of the most important topics of the ...
... training. Chemistry is a basic science and a powerful tool for studying and learning processes in living systems. Therefore, medical students must thoroughly understand the basic ideas, laws and methods of this science. Program expected to consider the foundations of the most important topics of the ...
Organic Chemistry Organic Chemistry
... are related to these recognizable combinations, called functional groups. These functional groups determine whether the molecules are readily soluble in polar or non-polar solvents, whether they have high or low melting and boiling points, and whether they readily react with other molecules. So, if ...
... are related to these recognizable combinations, called functional groups. These functional groups determine whether the molecules are readily soluble in polar or non-polar solvents, whether they have high or low melting and boiling points, and whether they readily react with other molecules. So, if ...
Grossmont College Chemistry 141 Laboratory Manual 6th Edition
... mathematical theories; So it is often ignored although, it is frequently found to be orders of magnitude larger than the random errors. In fact, many published papers have later been show to be incorrect by amounts far greater than the claimed limits of error. ...
... mathematical theories; So it is often ignored although, it is frequently found to be orders of magnitude larger than the random errors. In fact, many published papers have later been show to be incorrect by amounts far greater than the claimed limits of error. ...
organonitrogen compounds i. amines
... Figure 23-4 lnfrared spectra of cyclohexanamine and N-methylbenzenamine (N-methylaniline) ...
... Figure 23-4 lnfrared spectra of cyclohexanamine and N-methylbenzenamine (N-methylaniline) ...
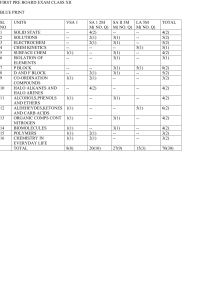
Class-XII, Summer assignment
... 3) A 5% solution of cane sugar (mol. wt. =342) is isotonic with 1% solution of a substance X. Calculate the molecular mass of X. (2 M) 4) During osmosis, Mention the flow of solvent through a semipermeable membrane. (1 M) 5) A solution containing 10 g per dm3of urea (molecular mass = 60 g mol–1) is ...
... 3) A 5% solution of cane sugar (mol. wt. =342) is isotonic with 1% solution of a substance X. Calculate the molecular mass of X. (2 M) 4) During osmosis, Mention the flow of solvent through a semipermeable membrane. (1 M) 5) A solution containing 10 g per dm3of urea (molecular mass = 60 g mol–1) is ...