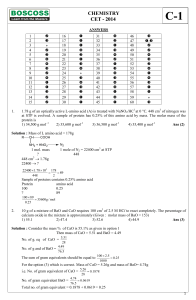
Questions
... These compounds can also be distinguished from one another by the use of concentrated sulphuric acid. (i) ...
... These compounds can also be distinguished from one another by the use of concentrated sulphuric acid. (i) ...
Exam Edge Digital
... be familiar with how overlap of atomic orbitals is used to explain the formation of covalent bonds and be able to distinguish between sigma and pi bonding. You should also know how the number of bonding electron pairs and lone electron pairs influences the shape of covalent molecules. It is essentia ...
... be familiar with how overlap of atomic orbitals is used to explain the formation of covalent bonds and be able to distinguish between sigma and pi bonding. You should also know how the number of bonding electron pairs and lone electron pairs influences the shape of covalent molecules. It is essentia ...
Mineralization of Drugs in Aqueous Medium by Advanced Oxidation
... This procedure is known as anodic oxidation with H2O2 electrogeneration. The electro-Fenton process involves the enhancement of the oxidizing power of the above electrolytic system by adding small amounts of a catalyst like Fe2+, which reacts with electrogenerated H2O2 to yield •OH in solution from ...
... This procedure is known as anodic oxidation with H2O2 electrogeneration. The electro-Fenton process involves the enhancement of the oxidizing power of the above electrolytic system by adding small amounts of a catalyst like Fe2+, which reacts with electrogenerated H2O2 to yield •OH in solution from ...
Discussion Questions
... 50. A 1.00-g sample of an alkaline earth metal chloride is treated with excess silver nitrate. All of the chloride is recovered as 1.38 g of silver chloride. Identify the metal. 51. A mixture contains only NaCl and Al2(SO4)3. A 1.45-g sample of the mixture is dissolved in water, and an excess ...
... 50. A 1.00-g sample of an alkaline earth metal chloride is treated with excess silver nitrate. All of the chloride is recovered as 1.38 g of silver chloride. Identify the metal. 51. A mixture contains only NaCl and Al2(SO4)3. A 1.45-g sample of the mixture is dissolved in water, and an excess ...
QualGroupD
... ions are very stable and prevent the cations from precipitating when oxalate is added even though their oxalates are relatively insoluble. Many complex ions are highly colored and the color of your solution at this stage may help in identification of the cations in your unknown. Testing known soluti ...
... ions are very stable and prevent the cations from precipitating when oxalate is added even though their oxalates are relatively insoluble. Many complex ions are highly colored and the color of your solution at this stage may help in identification of the cations in your unknown. Testing known soluti ...
EXAM IIR - Academics
... HNO3 + KOH H2O + KNO3 H+ + NO3- + K+ + OH- H2O + NO3- + K+ H+ + OH- H2O b. Write the balanced equation for the reaction between Acetic acid and potassium hydroxide. CH3COOH + KOH H2O + CH3COOK CH3COOH + K+ + OH- H2O + CH3COO- + K+ CH3COOH + OH- H2O + CH3COOc. Write the balanced NET IONIC ...
... HNO3 + KOH H2O + KNO3 H+ + NO3- + K+ + OH- H2O + NO3- + K+ H+ + OH- H2O b. Write the balanced equation for the reaction between Acetic acid and potassium hydroxide. CH3COOH + KOH H2O + CH3COOK CH3COOH + K+ + OH- H2O + CH3COO- + K+ CH3COOH + OH- H2O + CH3COOc. Write the balanced NET IONIC ...
CHEMISTRY CET
... The statement that is NOT correct is 1) Van der Waals constant 'a' measures extent of intermolecular attractive forces for real gases. (2) Boyle point depends on the nature of real gas. (3) Compressibility factor measures the deviation of real gas from ideal behaviour. (4) Critical temperature is th ...
... The statement that is NOT correct is 1) Van der Waals constant 'a' measures extent of intermolecular attractive forces for real gases. (2) Boyle point depends on the nature of real gas. (3) Compressibility factor measures the deviation of real gas from ideal behaviour. (4) Critical temperature is th ...
Chemical Reactions and Solution Stoichiometry
... Notice that in Interactive Figure 4.2.1 the water molecules orient themselves so that the oxygen atoms are near the Na+ cations and the hydrogen atoms are near the Cl− anions. This is due to the polar nature of water, a result of uneven electron distribution in water molecules. ( Flashforward to Se ...
... Notice that in Interactive Figure 4.2.1 the water molecules orient themselves so that the oxygen atoms are near the Na+ cations and the hydrogen atoms are near the Cl− anions. This is due to the polar nature of water, a result of uneven electron distribution in water molecules. ( Flashforward to Se ...
organic problems - St. Olaf College
... 25 Which of the following molecular formulas is reasonable for a stable compound? A) C8H14O2Cl B) C6H14Br2 C) C7H10NF D) C30H54N2Cl 26 What formal charges are present in the molecule C6H5C≡N-O? ( all heavy atoms have a valence shell octet, and C6H5- is a phenyl group) A) N is -1 and C is +1 B) N is ...
... 25 Which of the following molecular formulas is reasonable for a stable compound? A) C8H14O2Cl B) C6H14Br2 C) C7H10NF D) C30H54N2Cl 26 What formal charges are present in the molecule C6H5C≡N-O? ( all heavy atoms have a valence shell octet, and C6H5- is a phenyl group) A) N is -1 and C is +1 B) N is ...
Mechanistic and Computational Studies of Ferroin, Simple Organic
... many months of unpublished work, the initial models were determined to be inadequate and the project underwent a transformation, eschewing the model in an effort to focus on the ‘real deal’ and favoring a new theoretical approach, determined to be capable of examining many different systems as effic ...
... many months of unpublished work, the initial models were determined to be inadequate and the project underwent a transformation, eschewing the model in an effort to focus on the ‘real deal’ and favoring a new theoretical approach, determined to be capable of examining many different systems as effic ...
Recent Developments on the Mechanism and Kinetics
... Esters have played a significant role in daily living and chemical industry, such as plasticizers, fragrance, adhesive and lubricants (Joseph et al., 2005; Mbaraka & Shanks, 2006; Krause et al., 2009; Martínez et al., 2011). The vast majority of esters can be prepared using esterification reaction ...
... Esters have played a significant role in daily living and chemical industry, such as plasticizers, fragrance, adhesive and lubricants (Joseph et al., 2005; Mbaraka & Shanks, 2006; Krause et al., 2009; Martínez et al., 2011). The vast majority of esters can be prepared using esterification reaction ...
Chapter 4 Chemical Reactions and Solution Stoichiometry 4.1
... the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The overall double displacement reaction is represented by the following balanced equation: NaCl(aq ...
... the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The overall double displacement reaction is represented by the following balanced equation: NaCl(aq ...
NO - Blue Devil Chem
... gas causing the column of carbon puff up, just like gases during cooking cause a cake to rise. The name carbohydrate derives from the formula of sugars such as sucrose, C12H22O11 (notice the 2:1 H:O ratio C12(H2O)11) in which the formula appears to be a hydrate of carbon. C12H22O11(s) → 11H2O(g) + 1 ...
... gas causing the column of carbon puff up, just like gases during cooking cause a cake to rise. The name carbohydrate derives from the formula of sugars such as sucrose, C12H22O11 (notice the 2:1 H:O ratio C12(H2O)11) in which the formula appears to be a hydrate of carbon. C12H22O11(s) → 11H2O(g) + 1 ...
Chemistry booklet
... Eg, in Na+ Cℓ- Nao [Ne]3s1 loses one electron to form Na+ [Ne]3so, so it has been oxidised , whilst Clo [1s22s22p5 ] gains one electron to form Cℓ- [1s22s22p6]=[Ne], so it has been reduced, and the two ions have the stable ‘s2p6’ electron configuration. Na+ is said to have an OS (ON) of +1, while Cℓ ...
... Eg, in Na+ Cℓ- Nao [Ne]3s1 loses one electron to form Na+ [Ne]3so, so it has been oxidised , whilst Clo [1s22s22p5 ] gains one electron to form Cℓ- [1s22s22p6]=[Ne], so it has been reduced, and the two ions have the stable ‘s2p6’ electron configuration. Na+ is said to have an OS (ON) of +1, while Cℓ ...
sample chapter
... Na⫹ and Cl⫺ ions are separated from each other. In solution, each Na⫹ ion is surrounded by a number of water molecules orienting their negative ends toward the cation. Similarly, each Cl⫺ ion is surrounded by water molecules with their positive ends oriented toward the anion (Figure 4.2). The proces ...
... Na⫹ and Cl⫺ ions are separated from each other. In solution, each Na⫹ ion is surrounded by a number of water molecules orienting their negative ends toward the cation. Similarly, each Cl⫺ ion is surrounded by water molecules with their positive ends oriented toward the anion (Figure 4.2). The proces ...
Mock Examination (2016/2017) CHEMISTRY PAPER 1 SECTION B
... Chemical property: Oxidation with acidified potassium dichromate solution with heating Only butan-1-ol and butan-2-ol can undergoes oxidation that turn acidified potassium dichromate solution from orange to green, while methylbutan-2-ol cannot. ...
... Chemical property: Oxidation with acidified potassium dichromate solution with heating Only butan-1-ol and butan-2-ol can undergoes oxidation that turn acidified potassium dichromate solution from orange to green, while methylbutan-2-ol cannot. ...
Soft X-Ray-Induced Decomposition of Amino Acids: An XPS, Mass
... spectra have complicated asymmetric shapes due to contributions of several functional groups and shake-up satellites.3 For the assignment of the spectral features, it must be taken into account that the form of amino acids that is most stable in the solid state is a zwitterion with a protonated amin ...
... spectra have complicated asymmetric shapes due to contributions of several functional groups and shake-up satellites.3 For the assignment of the spectral features, it must be taken into account that the form of amino acids that is most stable in the solid state is a zwitterion with a protonated amin ...
LABORATORY MANUAL CHEMISTRY 121
... In aqueous solution the green complex trans-dichlorbis (ethylenediamine) cobalt(III) chloride dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction ...
... In aqueous solution the green complex trans-dichlorbis (ethylenediamine) cobalt(III) chloride dissociates into chloride ion and trans-[Co(en)2Cl2]+. This cobalt complex ion then reacts in acid solution to yield a mixture of the cis and trans forms of [Co(en)2(H2O)Cl]2+. The progress of this reaction ...
Benzylamine reacts with nitrous acid to form unstable
... 1. Write the use of quaternary ammonium salts? 2. What product is formed when aniline is first diazotized and then treated with Phenol in alkaline medium? 3. How is phenyl hydrazine prepared from aniline? 4. What is the IUPAC name of a tertiary amine containing one methyl, one ethyl and one n-propyl ...
... 1. Write the use of quaternary ammonium salts? 2. What product is formed when aniline is first diazotized and then treated with Phenol in alkaline medium? 3. How is phenyl hydrazine prepared from aniline? 4. What is the IUPAC name of a tertiary amine containing one methyl, one ethyl and one n-propyl ...
Chemistry 120
... liquids such as acetone, hexane, benzene or ether or water. Water is the most important solvent. The oceans cover ~ ¾ of the surface of the planet and every cell is mainly composed of water. Solutions in water are termed aqueous solutions and species are written as E(aq). ...
... liquids such as acetone, hexane, benzene or ether or water. Water is the most important solvent. The oceans cover ~ ¾ of the surface of the planet and every cell is mainly composed of water. Solutions in water are termed aqueous solutions and species are written as E(aq). ...
analytical chemistry - Львівський національний медичний
... Getting the maximum amount of product from a reaction depends on the proper selection of reaction conditions. By changing these conditions, we can increase or decrease the yield of product. We might change the yield by: 1. Changing concentrations by removing products or adding reactants to the react ...
... Getting the maximum amount of product from a reaction depends on the proper selection of reaction conditions. By changing these conditions, we can increase or decrease the yield of product. We might change the yield by: 1. Changing concentrations by removing products or adding reactants to the react ...
5 organic chemistry: functional groups
... The longest chain contains the OOH group, which means the compound is named as a derivative of octane. Because it is an alcohol, it would be tempting to name it as an octanol. But it contains a CPC double bond, which means it must be an octenol. We now have to indicate that the OOH group is on one e ...
... The longest chain contains the OOH group, which means the compound is named as a derivative of octane. Because it is an alcohol, it would be tempting to name it as an octanol. But it contains a CPC double bond, which means it must be an octenol. We now have to indicate that the OOH group is on one e ...
New liquid absorbents for the removal of CO2 from gas
... acid gases in general using primary amines and amino alcohols as efficient sorption media are limited for this purpose because they do not fit the requirements of regeneration, bio-compatibility and sterilization temperature imposed by the intended applications. Thus, a more complex system has to be ...
... acid gases in general using primary amines and amino alcohols as efficient sorption media are limited for this purpose because they do not fit the requirements of regeneration, bio-compatibility and sterilization temperature imposed by the intended applications. Thus, a more complex system has to be ...
Nature of Materials in Serum That Interfere inthe Glucose Oxidase
... against a buffer solution to remove the glucose from the protein serum matrix (8-10), sq that interfering material of high-molecular-weight is excluded. Because automated methods are most frequently used for routine analysis, the interferences most often discussed are those caused by substances of l ...
... against a buffer solution to remove the glucose from the protein serum matrix (8-10), sq that interfering material of high-molecular-weight is excluded. Because automated methods are most frequently used for routine analysis, the interferences most often discussed are those caused by substances of l ...