Water: The Universal Solvent
... • Water is polar thus having positive & negative partial charges on its ends. ...
... • Water is polar thus having positive & negative partial charges on its ends. ...
5H2O → CuSO4 + 5H2O(g)
... ◦ Solution: homogeneous mixture of two or more substances ◦ Solute: substance present in smaller amount ◦ Solvent: substance present in greater amount ◦ Aqueous solution: solvent is water ...
... ◦ Solution: homogeneous mixture of two or more substances ◦ Solute: substance present in smaller amount ◦ Solvent: substance present in greater amount ◦ Aqueous solution: solvent is water ...
Synthesis of Imidazolium Room-Temperature Ionic
... Department of Chemistry, Texas Christian University, Fort Worth, TX 76129; *[email protected] ...
... Department of Chemistry, Texas Christian University, Fort Worth, TX 76129; *[email protected] ...
Introduction
... Which one is more likely to be pulled apart by water molecules? Electrolytes are ionic and strong acid solutions (e.g., GatoradeTM); Nonelectrolytes are covalent compounds (e.g., sugar); weak electrolytes are in between. ...
... Which one is more likely to be pulled apart by water molecules? Electrolytes are ionic and strong acid solutions (e.g., GatoradeTM); Nonelectrolytes are covalent compounds (e.g., sugar); weak electrolytes are in between. ...
Learning Outcomes for CHEM1001 in 2015
... 4. explain the differences between elements, compounds and mixtures 5. explain the difference between allotropes and the physical state of an element 6. explain what atoms are and how they combine to form compounds 7. appreciate the difference between physical and chemical properties 8. list the par ...
... 4. explain the differences between elements, compounds and mixtures 5. explain the difference between allotropes and the physical state of an element 6. explain what atoms are and how they combine to form compounds 7. appreciate the difference between physical and chemical properties 8. list the par ...
synthesis and potentiometric analysis of - G
... spectrometer «Nicolet Avatar-360" (KBr tablets, measurement error is 0.2 cm-1). 1H NMR spectra were recorded by the spectrometer «Bruker DRX500» of operating frequency of 500 MHz in DMSO-d6 solution relatively to internal standard (TMS) (measurement error is ± 0.10.2 ppm). Melting point was detemine ...
... spectrometer «Nicolet Avatar-360" (KBr tablets, measurement error is 0.2 cm-1). 1H NMR spectra were recorded by the spectrometer «Bruker DRX500» of operating frequency of 500 MHz in DMSO-d6 solution relatively to internal standard (TMS) (measurement error is ± 0.10.2 ppm). Melting point was detemine ...
Bonding
... and 3 unpaired electrons. According to crystal field theory, as the chloride ion approaches the cobalt(II) ion, repulsion between the chloride lone pairs and the metal electrons affects the metal d orbitals differently (the x2-y2 and z2 more than the xy, xz, and yz). There is an energy difference be ...
... and 3 unpaired electrons. According to crystal field theory, as the chloride ion approaches the cobalt(II) ion, repulsion between the chloride lone pairs and the metal electrons affects the metal d orbitals differently (the x2-y2 and z2 more than the xy, xz, and yz). There is an energy difference be ...
2011 Lecture 22: Transport in Bulk Electrolytes
... A subtle consequence of quasi-electroneutrality is that the bulk electric field generally does not satisfy Maxwell’s equations. In particular, in a linear dielectric medium, one should properly impose Poisson’s equation, −ε∇2 φ = ρe (where ε is the permittivity) with an electrostatic boundary condit ...
... A subtle consequence of quasi-electroneutrality is that the bulk electric field generally does not satisfy Maxwell’s equations. In particular, in a linear dielectric medium, one should properly impose Poisson’s equation, −ε∇2 φ = ρe (where ε is the permittivity) with an electrostatic boundary condit ...
Extended Abstract Template
... Solid phase extraction (SPE) is a widely used extraction method in sample preparation and pre-concentration because of high concentration factor. It is normally performed using either cartridge or disc format. However, commercially available SPE sorbent is relatively expensive and SPE process is les ...
... Solid phase extraction (SPE) is a widely used extraction method in sample preparation and pre-concentration because of high concentration factor. It is normally performed using either cartridge or disc format. However, commercially available SPE sorbent is relatively expensive and SPE process is les ...
Electronic Spectroscopy of Transition Metal Ions
... • combine these into S and L which represent the TOTAL spin and orbital angular momentum • there are many different arrangements of electrons in d orbitals so this gives rise to many possible states (RussellSaunders ‘terms’) that represent different energies for the system as a whole eg. d2 ion 1st ...
... • combine these into S and L which represent the TOTAL spin and orbital angular momentum • there are many different arrangements of electrons in d orbitals so this gives rise to many possible states (RussellSaunders ‘terms’) that represent different energies for the system as a whole eg. d2 ion 1st ...
Chapter 4 Notes: Types of Reactions & Solution
... slightly negative H pulls less hard on e-s, so the H end of the molecule is slightly positive This makes H2O a POLAR molecule (oppositely ...
... slightly negative H pulls less hard on e-s, so the H end of the molecule is slightly positive This makes H2O a POLAR molecule (oppositely ...
System International Base Units
... Combine the name of the cation and anion while dropping the words “cation” and “anion.” o MgF2 Magnesium cation and fluoride anion make magnesium fluoride o MgSO4 Magnesium cation and sulfate anion make magnesium sulfate o Fe(ClO3)3 Iron (III) cation and chlorate anion make iron (III) chlorate Nam ...
... Combine the name of the cation and anion while dropping the words “cation” and “anion.” o MgF2 Magnesium cation and fluoride anion make magnesium fluoride o MgSO4 Magnesium cation and sulfate anion make magnesium sulfate o Fe(ClO3)3 Iron (III) cation and chlorate anion make iron (III) chlorate Nam ...
System International Base Units
... Combine the name of the cation and anion while dropping the words “cation” and “anion.” o MgF2 Magnesium cation and fluoride anion make magnesium fluoride o MgSO4 Magnesium cation and sulfate anion make magnesium sulfate o Fe(ClO3)3 Iron (III) cation and chlorate anion make iron (III) chlorate Nam ...
... Combine the name of the cation and anion while dropping the words “cation” and “anion.” o MgF2 Magnesium cation and fluoride anion make magnesium fluoride o MgSO4 Magnesium cation and sulfate anion make magnesium sulfate o Fe(ClO3)3 Iron (III) cation and chlorate anion make iron (III) chlorate Nam ...
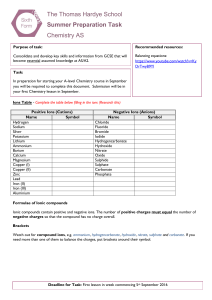
The Thomas Hardye School Summer Preparation Task Chemistry AS
... 13. Sodium hydroxide + Copper(II) sulphate Sodium sulphate + Copper(II) hydroxide 14. Sodium hydroxide + Phosphoric acid Sodium phosphate + Water 15. Sodium hydroxide + Sulphuric acid Sodium sulphate + Water Acid Reactions - Complete the word equations below ...
... 13. Sodium hydroxide + Copper(II) sulphate Sodium sulphate + Copper(II) hydroxide 14. Sodium hydroxide + Phosphoric acid Sodium phosphate + Water 15. Sodium hydroxide + Sulphuric acid Sodium sulphate + Water Acid Reactions - Complete the word equations below ...
Review Ch 4 - mvhs
... Sulfur trioxide gas is added to excess water. Solid sodium oxide is added to water. Solid calcium oxide is mixed with sulfer trioxide gas. Carbon disulfide vapor is burned in excess oxygen. Electric current is passed through water. Ethanol(C2H5OH) is completely burned in air. Solid zinc reacts with ...
... Sulfur trioxide gas is added to excess water. Solid sodium oxide is added to water. Solid calcium oxide is mixed with sulfer trioxide gas. Carbon disulfide vapor is burned in excess oxygen. Electric current is passed through water. Ethanol(C2H5OH) is completely burned in air. Solid zinc reacts with ...
Saturday Study Session 1 1st Class Reactions
... F2 + 2NaBr 2NaF + Br2 molecular F2 + 2Br- 2F- + Br2 – Solutions of Hydrofluoric acid is added to ammonium hydroxide HF + NH4OH NH4F + H2O molecular HF + NH4OH NH4 + + F- + H2O ionic – Butane is burned in Air 2C4H10 + 13O2 8CO2 + 10H2O molecular and ionic ...
... F2 + 2NaBr 2NaF + Br2 molecular F2 + 2Br- 2F- + Br2 – Solutions of Hydrofluoric acid is added to ammonium hydroxide HF + NH4OH NH4F + H2O molecular HF + NH4OH NH4 + + F- + H2O ionic – Butane is burned in Air 2C4H10 + 13O2 8CO2 + 10H2O molecular and ionic ...
SO2 SO3 - mrkearsley.com - The Web Site of Mr. Kearsley
... Oxidation numbers are useful for: 1) naming compounds 2) writing formulas ...
... Oxidation numbers are useful for: 1) naming compounds 2) writing formulas ...
ELECTROLYTES: NONELECTROLYTES
... Ions tend to stay in solution where they can conduct a current rather than re-forming a solid. The reason for this is the polar nature of the water molecule… Positive ions associate with the negative end of the water dipole (oxygen). Negative ions associate with the positive end of the water dipole ...
... Ions tend to stay in solution where they can conduct a current rather than re-forming a solid. The reason for this is the polar nature of the water molecule… Positive ions associate with the negative end of the water dipole (oxygen). Negative ions associate with the positive end of the water dipole ...
CHEMICAL REACTIONS
... • Compounds that do not dissolve in water or only a small fraction dissolves in water thus producing a small amount of ions. • These cmps include insoluble salts, weak acids and weak bases. • When a solid insoluble salt is added to water, most of it sinks to the bottom of the beaker as a solid. ...
... • Compounds that do not dissolve in water or only a small fraction dissolves in water thus producing a small amount of ions. • These cmps include insoluble salts, weak acids and weak bases. • When a solid insoluble salt is added to water, most of it sinks to the bottom of the beaker as a solid. ...
Name_______________________ Answers to Final Exam Study
... 3. Which of the following is not an example of a physical change? A) water boiling C) lead melting B) sugar dissolving D) food souring 4. Which of these steps in the digestive process is a physical change? A) Saliva changing carbohydrates to sugars B) The hormone insulin metabolizing C) Enzymes brea ...
... 3. Which of the following is not an example of a physical change? A) water boiling C) lead melting B) sugar dissolving D) food souring 4. Which of these steps in the digestive process is a physical change? A) Saliva changing carbohydrates to sugars B) The hormone insulin metabolizing C) Enzymes brea ...
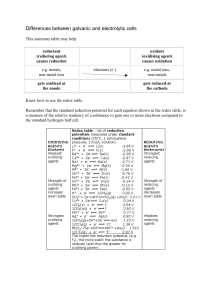
Differences between galvanic and electrolytic cells
... solution, so you might like to try this experiment using a dilute solution and then repeat it with a more concentrated solution. ...
... solution, so you might like to try this experiment using a dilute solution and then repeat it with a more concentrated solution. ...
chapter 4 review: types of chemical reactions and
... is dissolved and the sulfur is converted to the sulfate ion. Barium nitrate is added which causes the sulfate to precipitate out as BaSO4. The original sample has a mass of 3.187 g. The dried BaSO4 has a mass of 2.005 g. What is the percent of sulfur in the original ore? ...
... is dissolved and the sulfur is converted to the sulfate ion. Barium nitrate is added which causes the sulfate to precipitate out as BaSO4. The original sample has a mass of 3.187 g. The dried BaSO4 has a mass of 2.005 g. What is the percent of sulfur in the original ore? ...
Chapter 4 Reactions in Aqueous Solution 4.1 Aqueous Solutions
... • Reactions that result in the formation of precipitate, insoluble solid that separates from the solution • Often involve ionic compounds AgNO3(aq) + 2NaBr(aq) d AgBr(s) + NaNO 3(aq) ...
... • Reactions that result in the formation of precipitate, insoluble solid that separates from the solution • Often involve ionic compounds AgNO3(aq) + 2NaBr(aq) d AgBr(s) + NaNO 3(aq) ...
SCH4C Exam Review Assignment Kathleen Fall 2014
... 12. Is the bond between Cl-O polar covalent or non-polar covalent? Show your calculation. ...
... 12. Is the bond between Cl-O polar covalent or non-polar covalent? Show your calculation. ...
Ionic compound

In chemistry, an ionic compound is a chemical compound in which ions are held together in a structure by electrostatic forces termed ionic bonds. The positively charged ions are called cations and the negatively charged ions are called anions. These can be simple ions such as the sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic species such as the carbonate ion (CO32−) in calcium carbonate. Individual ions within an ionic compound usually have multiple nearest neighbours, so are not considered to be part of molecules, but instead part of a continuous three-dimensional network, usually in a crystalline structure.Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.Ionic compounds without the acidic hydrogen ion (H+), or the basic ions hydroxide (OH−) or oxide (O2−), are also known as salts and can be formed by acid-base reactions. Ionic compounds containing hydrogen ions are classified as acids and compounds containing hydroxide or oxide ions are classified as bases.