Hebden V.2 – Oxidation Numbers
... 1. Write the oxidation and reduction half reactions with their potentials (from the table) 2. E0cell = E0RED - E0ox 3. If you have to reverse the way the reaction is written to show the reduction or oxidation you must reverse the sign on the E0 4. If E0cell is positive = spontaneous 5. If E0cell is ...
... 1. Write the oxidation and reduction half reactions with their potentials (from the table) 2. E0cell = E0RED - E0ox 3. If you have to reverse the way the reaction is written to show the reduction or oxidation you must reverse the sign on the E0 4. If E0cell is positive = spontaneous 5. If E0cell is ...
File
... Accept temperatures in this range. Accept room temperature as an answer if platinum or palladium used. the enthalpy change when (one mole of) the gaseous bond is broken (or formed) / X–Y(g) → X(g) + Y(g) / X(g) + Y(g) → X–Y(g); averaged for the same bond in a number of similar compounds / OWTTE; ...
... Accept temperatures in this range. Accept room temperature as an answer if platinum or palladium used. the enthalpy change when (one mole of) the gaseous bond is broken (or formed) / X–Y(g) → X(g) + Y(g) / X(g) + Y(g) → X–Y(g); averaged for the same bond in a number of similar compounds / OWTTE; ...
Water-Mediated Dimerization of Ubiquitin Ions Captured by
... dependent on the initial presence and ultimate occlusion of water. Although insights about protein hydration and watermediated processes have been gleaned from molecular dynamics simulations, there are few experimental tools for understanding the effects of water on protein structure and protein−prot ...
... dependent on the initial presence and ultimate occlusion of water. Although insights about protein hydration and watermediated processes have been gleaned from molecular dynamics simulations, there are few experimental tools for understanding the effects of water on protein structure and protein−prot ...
Copper Coordination Polymers with Infinite Chloride Ion Channels
... At the same time there are no decisive features, such as O-H..CI hydrogen bonds, in the "non-bondcd" coordination spheres for both chlorine atoms to account for the difference in the Cu-Cl bonds. The CI-.(H-)O contact is rather similar, with 3.25 A for C11 and 3.35 A for CI2['1. The unusual phenomen ...
... At the same time there are no decisive features, such as O-H..CI hydrogen bonds, in the "non-bondcd" coordination spheres for both chlorine atoms to account for the difference in the Cu-Cl bonds. The CI-.(H-)O contact is rather similar, with 3.25 A for C11 and 3.35 A for CI2['1. The unusual phenomen ...
syntheses, structures, and their interconversion
... addition of 0.5 vol% of water to the reaction mixture. Complete absence of water, or other volume ratios than 995 : 5 led to no or lower yields. For us, such hydrate compounds are particularly interesting for their apparent stability in water and thus the potential for subsequent reactions in aqueou ...
... addition of 0.5 vol% of water to the reaction mixture. Complete absence of water, or other volume ratios than 995 : 5 led to no or lower yields. For us, such hydrate compounds are particularly interesting for their apparent stability in water and thus the potential for subsequent reactions in aqueou ...
Unit 13 Stoichiometry (Theoretical)
... Ionic compounds o In 1 mole of Ca(NO3)2 there are 1 mole of calcium ions and 2 moles of nitrate ions o In 1 mole of K2O, the ion ratio is 2 mole K+1 ions: 1 mole of O-2 ions ...
... Ionic compounds o In 1 mole of Ca(NO3)2 there are 1 mole of calcium ions and 2 moles of nitrate ions o In 1 mole of K2O, the ion ratio is 2 mole K+1 ions: 1 mole of O-2 ions ...
2.3 Atomic and Molecular Collisions
... storage conditions and to laser interaction experiments with stored, negatively and positively charged molecules and clusters [1]. The beams for CSR were produced in its ion source area from discharge and sputter ion sources. Aside from its electronic diagnostic elements for stored ions, the CSR was ...
... storage conditions and to laser interaction experiments with stored, negatively and positively charged molecules and clusters [1]. The beams for CSR were produced in its ion source area from discharge and sputter ion sources. Aside from its electronic diagnostic elements for stored ions, the CSR was ...
Specific adsorption of carbonate ions at the zinc oxide/electrolyte
... Practical application is the most important aspect for experimental study of properties of the electrical double layer. Zeta potential measurements give important information concerning the properties of the diffuse part of the edl. Measurements include surface charge, ion adsorption, and zeta poten ...
... Practical application is the most important aspect for experimental study of properties of the electrical double layer. Zeta potential measurements give important information concerning the properties of the diffuse part of the edl. Measurements include surface charge, ion adsorption, and zeta poten ...
Electric conductivity of Cu (NO ) 2∙3 Н2 О solutions in
... Keywords: electrodeposition, dimethylsulphoxide, crystalohydrate of nitrate of copper (II), conductometry, electrolyte. The water solution of copper (II) nitrate acidified by nitric acid is of interest as electrolyte of coppering, carried out at the raised density (till 100 А∙dm–2). The received cov ...
... Keywords: electrodeposition, dimethylsulphoxide, crystalohydrate of nitrate of copper (II), conductometry, electrolyte. The water solution of copper (II) nitrate acidified by nitric acid is of interest as electrolyte of coppering, carried out at the raised density (till 100 А∙dm–2). The received cov ...
Topic 1: Quantitative Chemistry
... 4.1.5: State that transition elements can form more than one ion. 4.1.6: Predict whether a compound of two elements would be ionic from the position of the elements in the periodic table or negativity values.4.1.7: State the formula of common polyatomic ions formed by non-metals in periods 2 and 3. ...
... 4.1.5: State that transition elements can form more than one ion. 4.1.6: Predict whether a compound of two elements would be ionic from the position of the elements in the periodic table or negativity values.4.1.7: State the formula of common polyatomic ions formed by non-metals in periods 2 and 3. ...
New Coordination Compounds of Copper(II) with
... Analysis of IR vibrational spectra: The spectra of the coordination compounds of copper(II) with guanidinopyrimidines are very complicated and the entire assignment of all the absorption bands is impossible. However, a careful study might lead to some important structural conclusions. The frequencie ...
... Analysis of IR vibrational spectra: The spectra of the coordination compounds of copper(II) with guanidinopyrimidines are very complicated and the entire assignment of all the absorption bands is impossible. However, a careful study might lead to some important structural conclusions. The frequencie ...
Chapter 9: Non-aqueous media
... As we discuss the properties and uses of some non-aqueous solvents, we must keep in mind that the extent to which nonaqueous solvents can be used is limited by the fact that many are highly reactive. Quantitative data are scarce for non-aqueous media, and, in solvents of relative permittivity lower ...
... As we discuss the properties and uses of some non-aqueous solvents, we must keep in mind that the extent to which nonaqueous solvents can be used is limited by the fact that many are highly reactive. Quantitative data are scarce for non-aqueous media, and, in solvents of relative permittivity lower ...
Міністерство охорони здоров`я України
... Solubility depends on the nature of solvent: the higher dielectric conductivity of the liquid the better solvent is. For example, urine, alba, blood have higher dielectric conductivity then water. So, biological liquids and tissues are good solvents for biologically active compounds, molecules of wh ...
... Solubility depends on the nature of solvent: the higher dielectric conductivity of the liquid the better solvent is. For example, urine, alba, blood have higher dielectric conductivity then water. So, biological liquids and tissues are good solvents for biologically active compounds, molecules of wh ...
Unit 10: Solutions Text Notes from Zumdahl, Zumdahl, DeCoste
... 6. Substances present in smaller amounts are called _____solutes_______. ...
... 6. Substances present in smaller amounts are called _____solutes_______. ...
Iodine Clock Reaction and Photochemical Reduction
... appearance of blue colour after each addition until the time of a appearance of blue colour becomes 5-6 times the initial time. Shake the reaction mixture throughout the addition of Na2 S2 O 3 solution. For rapid mixing, an electric motor with glass stirrer may also be arranged in the flask. In orde ...
... appearance of blue colour after each addition until the time of a appearance of blue colour becomes 5-6 times the initial time. Shake the reaction mixture throughout the addition of Na2 S2 O 3 solution. For rapid mixing, an electric motor with glass stirrer may also be arranged in the flask. In orde ...
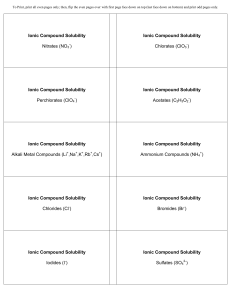
Ionic Compound Solubility Nitrates (NO3 ) Ionic Compound
... Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
... Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
Studies Regarding the Nickel Electrodeposition from
... hard or impossible to be obtained in classical aqueous solutions or to apply these coating layers with a suitable adherence on water sensitive metallic substrates such as Al, Mg, Ti and their alloys, stainless steels, other alloys containing high contents of refractory or rare earth metals (Endres e ...
... hard or impossible to be obtained in classical aqueous solutions or to apply these coating layers with a suitable adherence on water sensitive metallic substrates such as Al, Mg, Ti and their alloys, stainless steels, other alloys containing high contents of refractory or rare earth metals (Endres e ...
Critical Assessment of the Formation of Ionic-Liquid - PATh
... [C4mim]Cl, and distinct inorganic salts. The authors6 have shown that ATPS with [C4mim]Cl can be formed by the adequate addition of alkaline aqueous solutions of the following salts: K3PO4, KOH, K2HPO4, NaOH, K2CO3, and Na2HPO4. Nevertheless, when using acidic or neutral salts, such as KH2PO4, (NH4) ...
... [C4mim]Cl, and distinct inorganic salts. The authors6 have shown that ATPS with [C4mim]Cl can be formed by the adequate addition of alkaline aqueous solutions of the following salts: K3PO4, KOH, K2HPO4, NaOH, K2CO3, and Na2HPO4. Nevertheless, when using acidic or neutral salts, such as KH2PO4, (NH4) ...
New Liquid Crystalline Tolanes from (-)
... The triple bond between two aromatic rings is interesting, because it contributes to the polarizability, linearity and rigidity of the nal molecules. These factors are important and determine many physical properties, such as thermal stability and viscosity in liquid crystals. The presence of the n ...
... The triple bond between two aromatic rings is interesting, because it contributes to the polarizability, linearity and rigidity of the nal molecules. These factors are important and determine many physical properties, such as thermal stability and viscosity in liquid crystals. The presence of the n ...
PowerPoint Chapter 8
... • Negative Aspects of Brønsted-Lowry Definitions – Cannot classify isolated substances as acids (generate H3O+ in water), bases (generate OH in water), or neither. The same substance can sometimes be an acid and sometimes a base. – Does not allow predictions of (1) whether substances will react ...
... • Negative Aspects of Brønsted-Lowry Definitions – Cannot classify isolated substances as acids (generate H3O+ in water), bases (generate OH in water), or neither. The same substance can sometimes be an acid and sometimes a base. – Does not allow predictions of (1) whether substances will react ...
effect of inorganic ions on the oxidation of dichlorvos insecticide with
... This study analyzes the oxidation of dichlorvos with Fenton‘s reagent in solutions containing various ions. Results show that the larger the added amount of ferrous ions, the higher the elimination rate of dichlorvos and the oxidization rate after the addition of ferric ions is far smaller than that ...
... This study analyzes the oxidation of dichlorvos with Fenton‘s reagent in solutions containing various ions. Results show that the larger the added amount of ferrous ions, the higher the elimination rate of dichlorvos and the oxidization rate after the addition of ferric ions is far smaller than that ...
Calculating the conductivity of natural waters
... empirical equation directly relating measured conductivity to salinity in Crater Lake. This was done by fitting a polynomial to simultaneous measurements of conductivity and temperature from two dilutions of lake water over a range of temperatures (similar to the method used for seawater). Wüest et ...
... empirical equation directly relating measured conductivity to salinity in Crater Lake. This was done by fitting a polynomial to simultaneous measurements of conductivity and temperature from two dilutions of lake water over a range of temperatures (similar to the method used for seawater). Wüest et ...
Ionic compound

In chemistry, an ionic compound is a chemical compound in which ions are held together in a structure by electrostatic forces termed ionic bonds. The positively charged ions are called cations and the negatively charged ions are called anions. These can be simple ions such as the sodium (Na+) and chloride (Cl−) in sodium chloride, or polyatomic species such as the carbonate ion (CO32−) in calcium carbonate. Individual ions within an ionic compound usually have multiple nearest neighbours, so are not considered to be part of molecules, but instead part of a continuous three-dimensional network, usually in a crystalline structure.Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.Ionic compounds without the acidic hydrogen ion (H+), or the basic ions hydroxide (OH−) or oxide (O2−), are also known as salts and can be formed by acid-base reactions. Ionic compounds containing hydrogen ions are classified as acids and compounds containing hydroxide or oxide ions are classified as bases.