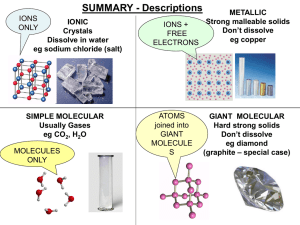
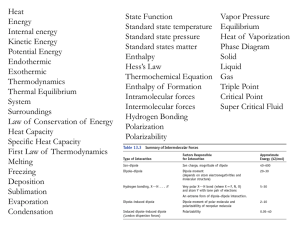
Chem 101 notes review
... The Nucleus: Hund’s Rule states that each orbital will be filled singly Build by adding the required number of protons before pairing begins. The singly filled orbitals will have (the atomic number) and neutrons (the mass of the atom) ...
... The Nucleus: Hund’s Rule states that each orbital will be filled singly Build by adding the required number of protons before pairing begins. The singly filled orbitals will have (the atomic number) and neutrons (the mass of the atom) ...
Ch 8 AP Practice
... (B) H2O (C) CH4 (D) C2H4 (E) PH3 3. The molecule with only one double bond 4. The molecule with the largest dipole moment 5. The molecule that has trigonal pyramidal geometry 53. According to the VSEPR model, the progressive decrease in the bond angles in the series of molecules CH4, NH3, and H2O is ...
... (B) H2O (C) CH4 (D) C2H4 (E) PH3 3. The molecule with only one double bond 4. The molecule with the largest dipole moment 5. The molecule that has trigonal pyramidal geometry 53. According to the VSEPR model, the progressive decrease in the bond angles in the series of molecules CH4, NH3, and H2O is ...
Nano-transistors Sensitive to Vibrations in a Single Molecule
... Molecular Transistors Molecular devices might enable the manipulation of single electrons on the smallest possible device length scales. A cross disciplinary group at Cornell has demonstrated for the first time a transistor device that reaches the ultimate limit in which an electron hops on and off ...
... Molecular Transistors Molecular devices might enable the manipulation of single electrons on the smallest possible device length scales. A cross disciplinary group at Cornell has demonstrated for the first time a transistor device that reaches the ultimate limit in which an electron hops on and off ...