The Mole: A Measurement of Matter
... You live in a quantitative world. The grade you got on your last exam, the number of times you heard your favorite song on the radio yesterday, and the cost of a bicycle you would like to own are all important quantities to you. These are quantities that answer questions such as "How much?" or "How ...
... You live in a quantitative world. The grade you got on your last exam, the number of times you heard your favorite song on the radio yesterday, and the cost of a bicycle you would like to own are all important quantities to you. These are quantities that answer questions such as "How much?" or "How ...
Chemistry - RESONANCE PCCP IDEAL for NTSE, IJSO, Olympiads
... Mol wt. or At. wt No. of electrons lost or gained by one molecule of the substance ...
... Mol wt. or At. wt No. of electrons lost or gained by one molecule of the substance ...
The Alkaline Earth Metals (Group 2) - Chemwiki
... have very negative reduction potentials, large amounts of energy are needed to isolate them from their ores. Four of the six group 2 elements— magnesium (Mg), calcium (Ca), strontium (Sr), and barium (Ba)—were first isolated in the early 19th century by Sir Humphry Davy, using a technique similar to ...
... have very negative reduction potentials, large amounts of energy are needed to isolate them from their ores. Four of the six group 2 elements— magnesium (Mg), calcium (Ca), strontium (Sr), and barium (Ba)—were first isolated in the early 19th century by Sir Humphry Davy, using a technique similar to ...
NON METALS- SILICON
... 2. Non metals are chemically active. 3. All non metals, except graphite, are bad conductors of electricity. 4. Carbon, sulphur and phosphorous, which are non metals, exhibit allotropy. ...
... 2. Non metals are chemically active. 3. All non metals, except graphite, are bad conductors of electricity. 4. Carbon, sulphur and phosphorous, which are non metals, exhibit allotropy. ...
1 Assignment 4 Hydrogen – The Unique Element
... many molecular hydrides e.g. boiling points, pKa. It is also important in many biological settings e.g. base-pairing in DNA. Typically, hydrogen bonds have enthalpies of between 20 and 60 kJ mol-1.15 The presence of hydrogen bonds within certain compounds can be detected using molecular structure de ...
... many molecular hydrides e.g. boiling points, pKa. It is also important in many biological settings e.g. base-pairing in DNA. Typically, hydrogen bonds have enthalpies of between 20 and 60 kJ mol-1.15 The presence of hydrogen bonds within certain compounds can be detected using molecular structure de ...
1 Assignment 5 Hydrogen – The Unique Element
... many molecular hydrides e.g. boiling points, pKa. It is also important in many biological settings e.g. base-pairing in DNA. Typically, hydrogen bonds have enthalpies of between 20 and 60 kJ mol-1.15 The presence of hydrogen bonds within certain compounds can be detected using molecular structure de ...
... many molecular hydrides e.g. boiling points, pKa. It is also important in many biological settings e.g. base-pairing in DNA. Typically, hydrogen bonds have enthalpies of between 20 and 60 kJ mol-1.15 The presence of hydrogen bonds within certain compounds can be detected using molecular structure de ...
Booklet Chapter 3
... b. Identify which of two atoms in a polar covalent bond has a partial negative charge and which atom has a partial positive charge. c. Identify which of two atoms in an ionic bond has a negative charge and which atom has a positive charge. d. Given two bonds, determine which of the bonds would be ex ...
... b. Identify which of two atoms in a polar covalent bond has a partial negative charge and which atom has a partial positive charge. c. Identify which of two atoms in an ionic bond has a negative charge and which atom has a positive charge. d. Given two bonds, determine which of the bonds would be ex ...
atomic mass
... • Outer electrons define the radius of the atom. • Electrons, and the nucleus, are much smaller than the atom itself, so most of the atom is ...
... • Outer electrons define the radius of the atom. • Electrons, and the nucleus, are much smaller than the atom itself, so most of the atom is ...
Chemical analysis of 24 dusty (pre-) main
... lines, and illuminate a bright reflection nebula in their immediate vicinity. Later on other observational criteria were proposed. Finkenzeller & Mundt (1984) mentioned that a shared characteristic of Herbig Ae/Be stars is the presence of an IR excess, which is an independent, but more easily detect ...
... lines, and illuminate a bright reflection nebula in their immediate vicinity. Later on other observational criteria were proposed. Finkenzeller & Mundt (1984) mentioned that a shared characteristic of Herbig Ae/Be stars is the presence of an IR excess, which is an independent, but more easily detect ...
Chemistry - Resonance
... The main reasons for this huge number of organic compounds are (i) Catenation : The property of self linking of carbon atoms through covalent bonds to form long straight or branched chains and rings of different sizes is called catenation.Carbon shows maximum catenation in the periodic table due to ...
... The main reasons for this huge number of organic compounds are (i) Catenation : The property of self linking of carbon atoms through covalent bonds to form long straight or branched chains and rings of different sizes is called catenation.Carbon shows maximum catenation in the periodic table due to ...
Atoms, Ions and Molecules
... All atoms of a given element show the same chemical properties. 2. Atoms of different elements have different properties. In an ordinary chemical reaction, no atom of any element disappears or is changed into an atom of another element. 3. Compounds are formed when atoms of two or more elements comb ...
... All atoms of a given element show the same chemical properties. 2. Atoms of different elements have different properties. In an ordinary chemical reaction, no atom of any element disappears or is changed into an atom of another element. 3. Compounds are formed when atoms of two or more elements comb ...
Some basic concepts of chemistry
... The three basic units, i.e., units of mass, length and time are independent units and cannot be derived from any other units, hence they are called fundamental units. The three fundamental units cannot describe all the physical quantities such as temperature, intensity of luminosity, electric curren ...
... The three basic units, i.e., units of mass, length and time are independent units and cannot be derived from any other units, hence they are called fundamental units. The three fundamental units cannot describe all the physical quantities such as temperature, intensity of luminosity, electric curren ...
W. M. White Geochemistry Chapter 10: Cosmochemistry
... we learn about the evolution of the Earth by examining old rocks, we can learn about the evolution of the cosmos by looking at old stars. The old stars of Population II are considerably poorer in heavy elements than are young stars. In particular, Population II stars have a Fe/H ratio typically a fa ...
... we learn about the evolution of the Earth by examining old rocks, we can learn about the evolution of the cosmos by looking at old stars. The old stars of Population II are considerably poorer in heavy elements than are young stars. In particular, Population II stars have a Fe/H ratio typically a fa ...
Chapter One
... that no longer retains any of the characteristics of the element. An atom is therefore the smallest particle that can be used to identify an element. Compounds are substances that contain more than one element combined in fixed proportions. Water, for example, is composed of the elements hydrogen an ...
... that no longer retains any of the characteristics of the element. An atom is therefore the smallest particle that can be used to identify an element. Compounds are substances that contain more than one element combined in fixed proportions. Water, for example, is composed of the elements hydrogen an ...
Metal-poor Stars
... It was long believed that all stars would have a similar chemical composition to the Sun. In the late 1940s, however, some metal lines observed in stars appeared to be unusually weak compared with the Sun. It was first suspected that these stars could be hydrogen-deficient or might have peculiar atm ...
... It was long believed that all stars would have a similar chemical composition to the Sun. In the late 1940s, however, some metal lines observed in stars appeared to be unusually weak compared with the Sun. It was first suspected that these stars could be hydrogen-deficient or might have peculiar atm ...
Elements, Compounds, and Chemical Equations
... To complete TAKS Review Booklet: • For each objective include AT LEAST 10 facts. • Facts may include sentences, formulas, labeled pictures, diagrams, or definitions. • The Review Booklet will be a major grade and is due on Thursday. Come to tutoring to use the study guide to find additional facts! ...
... To complete TAKS Review Booklet: • For each objective include AT LEAST 10 facts. • Facts may include sentences, formulas, labeled pictures, diagrams, or definitions. • The Review Booklet will be a major grade and is due on Thursday. Come to tutoring to use the study guide to find additional facts! ...
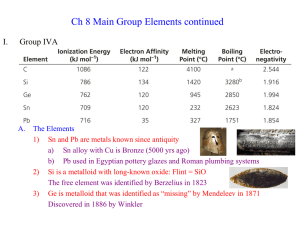
InorgCh8.2
... 3) Ge is metalloid that was identified as “missing” by Mendeleev in 1871 Discovered in 1886 by Winkler ...
... 3) Ge is metalloid that was identified as “missing” by Mendeleev in 1871 Discovered in 1886 by Winkler ...
practice test 4 CHM 112
... 6. The equilibrium 3Fe(s) + C(s) Fe3C(s) is established in a solid solution. For such a solution, one can write an equilibrium constant in the usual way except that here one has concentrations that refer to solids in the solid solution. Determine the equilibrium constant for the formation of cementi ...
... 6. The equilibrium 3Fe(s) + C(s) Fe3C(s) is established in a solid solution. For such a solution, one can write an equilibrium constant in the usual way except that here one has concentrations that refer to solids in the solid solution. Determine the equilibrium constant for the formation of cementi ...
Chemical Equations and Stoichiometry
... When it comes to chemistry it is also not practical (or reliable) to count grains. It would be even less practical to count molecules. So we use the idea of a bag or a mole which is about 6.02 X 1023 particles (atoms or molecules) of the substance were talking about. Right now that number is not too ...
... When it comes to chemistry it is also not practical (or reliable) to count grains. It would be even less practical to count molecules. So we use the idea of a bag or a mole which is about 6.02 X 1023 particles (atoms or molecules) of the substance were talking about. Right now that number is not too ...
MidtermReview2012
... the think skills required to accurately answer questions. If you need more practice problems for an objective, start by reviewing your old tests! Ask for more! Why Study Like This? This packet will guide you to spend the MOST amount of your time focusing on the 1s. Once you understand that stuff, mo ...
... the think skills required to accurately answer questions. If you need more practice problems for an objective, start by reviewing your old tests! Ask for more! Why Study Like This? This packet will guide you to spend the MOST amount of your time focusing on the 1s. Once you understand that stuff, mo ...
chemistry
... By observing the laws of chemical combination, John Dalton proposed an atomic theory of matter. The main points of Dalton’s atomic theory are as follows: (i) Matter is made up of extremely small, indivisible particles called atoms. (ii) Atom is the smallest particle that takes part in chemical react ...
... By observing the laws of chemical combination, John Dalton proposed an atomic theory of matter. The main points of Dalton’s atomic theory are as follows: (i) Matter is made up of extremely small, indivisible particles called atoms. (ii) Atom is the smallest particle that takes part in chemical react ...
Chapter 3 Notes
... that contains as many atoms, molecules, ions, or other elementary units as the number of atoms in 12.01 g C. The number is 6.02 × 1023, or Avogadro's number. ...
... that contains as many atoms, molecules, ions, or other elementary units as the number of atoms in 12.01 g C. The number is 6.02 × 1023, or Avogadro's number. ...
Group 1: The Alkali Metals
... Answers 1. Sodium has a higher boiling point because it has a larger atomic radius that Francium. Greater atomic radius means a bigger molecule thus having a higher boiling point. 2. False. Group 1 and 2 form ionic hydrides. P block forms molecular hydride and Nitrogen is in P block. ...
... Answers 1. Sodium has a higher boiling point because it has a larger atomic radius that Francium. Greater atomic radius means a bigger molecule thus having a higher boiling point. 2. False. Group 1 and 2 form ionic hydrides. P block forms molecular hydride and Nitrogen is in P block. ...
Note Sheets and Sample Problems
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
... Chemical Foundations Chemistry: An Overview • Matter – takes up space, has mass, exhibits inertia - composed of atoms only 100 or so different types - Water made up of one oxygen and two hydrogen atoms - Pass an electric current through it to separate the two types of atoms and they rearrange to bec ...
SOME BASIC CONCEPTS OF CHEMISTRY NOTES
... mass of one atom relative to another by experimental means. Hydrogen being the lightest atom was assigned a mass of 1(without any units) and other elements were assigned masses relative to it. In the present system, the atomic mass of an element is given relative to the mass of ...
... mass of one atom relative to another by experimental means. Hydrogen being the lightest atom was assigned a mass of 1(without any units) and other elements were assigned masses relative to it. In the present system, the atomic mass of an element is given relative to the mass of ...
Abundance of the chemical elements

The abundance of a chemical element measures how common is the element relative to all other elements in a given environment. Abundance is measured in one of three ways: by the mass-fraction (the same as weight fraction); by the mole-fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases); or by the volume-fraction. Volume-fraction is a common abundance measure in mixed gases such as planetary atmospheres, and is similar in value to molecular mole-fraction for gas mixtures at relatively low densities and pressures, and ideal gas mixtures. Most abundance values in this article are given as mass-fractions. For example, the abundance of oxygen in pure water can be measured in two ways: the mass fraction is about 89%, because that is the fraction of water's mass which is oxygen. However, the mole-fraction is 33.3333...% because only 1 atom of 3 in water, H2O, is oxygen. As another example, looking at the mass-fraction abundance of hydrogen and helium in both the Universe as a whole and in the atmospheres of gas-giant planets such as Jupiter, it is 74% for hydrogen and 23-25% for helium; while the (atomic) mole-fraction for hydrogen is 92%, and for helium is 8%, in these environments. Changing the given environment to Jupiter's outer atmosphere, where hydrogen is diatomic while helium is not, changes the molecular mole-fraction (fraction of total gas molecules), as well as the fraction of atmosphere by volume, of hydrogen to about 86%, and of helium to 13%.