Name
... 32. What are the period, group, and symbol for Tin? period___________group_________symbol__________ 33. What number that is represented on each element of the periodic table shows how many electrons and protons it holds? ____________________________________________________________________ 34. How do ...
... 32. What are the period, group, and symbol for Tin? period___________group_________symbol__________ 33. What number that is represented on each element of the periodic table shows how many electrons and protons it holds? ____________________________________________________________________ 34. How do ...
chapter-7-explore-page-248-protons-neutrons
... Any one of these three quantities can be determined if you know the value of the other two quantities. For example: to determine the mass number of an atom, you must know the number of neutrons and the number of protons in the atom. An isotope often is written with the element name followed by t ...
... Any one of these three quantities can be determined if you know the value of the other two quantities. For example: to determine the mass number of an atom, you must know the number of neutrons and the number of protons in the atom. An isotope often is written with the element name followed by t ...
Chapter 2 Atoms, Molecules, and Ions
... 2.6 Ions and Ionic Compounds • If an electron is added or removed from an atom, then the atom now has a charge, or has become an ion. • Cation = positively charged = loss of electron • Anion = negatively charged = gain of electron • Polyatomic ion – ions that consist of atoms joined as in a molecul ...
... 2.6 Ions and Ionic Compounds • If an electron is added or removed from an atom, then the atom now has a charge, or has become an ion. • Cation = positively charged = loss of electron • Anion = negatively charged = gain of electron • Polyatomic ion – ions that consist of atoms joined as in a molecul ...
Branches of Chemistry
... Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other related compounds. Photochemists investigate the relationship between light and chemical reactions. Physical chemists use the principles of physics to ...
... Nuclear chemists investigate changes that happen in atomic nuclei. Organic chemists study hydrocarbons – compounds of carbon and hydrogen – and other related compounds. Photochemists investigate the relationship between light and chemical reactions. Physical chemists use the principles of physics to ...
Atoms and Elements Class Notes and Class Work
... Introduction to Matter Property: Characteristics matter has. Law: A rule nature seems to follow. It’s been observed regularly. Theory: Tries to explain the law The Particle Theory of Matter 1. Matter is made up of particles (Atoms, molecules, and ions) 2. Pure substances: One kind of matter or parti ...
... Introduction to Matter Property: Characteristics matter has. Law: A rule nature seems to follow. It’s been observed regularly. Theory: Tries to explain the law The Particle Theory of Matter 1. Matter is made up of particles (Atoms, molecules, and ions) 2. Pure substances: One kind of matter or parti ...
Regents Chemistry
... and void : The Greeks! Said matter was made up of particles from four elemental substances : Earth, water, air and fire Dalton’s Postulates (proposed 1808) 1. All elements are composed of indivisible atoms ...
... and void : The Greeks! Said matter was made up of particles from four elemental substances : Earth, water, air and fire Dalton’s Postulates (proposed 1808) 1. All elements are composed of indivisible atoms ...
Packet 5
... Protons – Positive charge, found in the nucleus and have a mass of 1 amu. ( Identify) Neutrons- No charge, found in the nucleus, and have a mass of 1 amu ( Isotopes) Electrons- Negative charge, found in the energy levels outside of the nucleus, have relatively no mass ( Ions) ...
... Protons – Positive charge, found in the nucleus and have a mass of 1 amu. ( Identify) Neutrons- No charge, found in the nucleus, and have a mass of 1 amu ( Isotopes) Electrons- Negative charge, found in the energy levels outside of the nucleus, have relatively no mass ( Ions) ...
The average atomic mass of an element is the sum of the
... Chlorine consists of two major isotopes, one with 18 neutrons (75.77 percent of natural chlorine atoms) and one with 20 neutrons (24.23 percent of natural chlorine atoms). The atomic number of chlorine is 17 (it has 17 protons in its nucleus). To calculate the average mass, first convert the percent ...
... Chlorine consists of two major isotopes, one with 18 neutrons (75.77 percent of natural chlorine atoms) and one with 20 neutrons (24.23 percent of natural chlorine atoms). The atomic number of chlorine is 17 (it has 17 protons in its nucleus). To calculate the average mass, first convert the percent ...
Notes matter energy
... lose electrons to become ions. Your periodic table gives some charges of ions that form from atoms. Ions can be indicated by a superscript in the upper right corner of the element box. For example Na+1 is a sodium ion that forms from a sodium atom losing one negatively charged electron. F−1 is a flu ...
... lose electrons to become ions. Your periodic table gives some charges of ions that form from atoms. Ions can be indicated by a superscript in the upper right corner of the element box. For example Na+1 is a sodium ion that forms from a sodium atom losing one negatively charged electron. F−1 is a flu ...
Any substance that cannot be decomposed into
... carbon isotope of mass 12, which is written carbon-12. This is the isotope of carbon that has in its nucleus 6 neutrons in addition to the 6 protons that all carbon atoms possess. One twelfth of the weight of carbon-12 is defined as the relative atomic weight unit (awu). Sometimes it is also called ...
... carbon isotope of mass 12, which is written carbon-12. This is the isotope of carbon that has in its nucleus 6 neutrons in addition to the 6 protons that all carbon atoms possess. One twelfth of the weight of carbon-12 is defined as the relative atomic weight unit (awu). Sometimes it is also called ...
Chem 115 POGIL Worksheet
... fundamental particles combine in forming the atom. This energy, called the binding energy, can be calculated with Einstein’s equation, E = mc2, where m is the mass converted into energy, and c is the speed of light in a vacuum. 18. What information about an element is provided in the box for that el ...
... fundamental particles combine in forming the atom. This energy, called the binding energy, can be calculated with Einstein’s equation, E = mc2, where m is the mass converted into energy, and c is the speed of light in a vacuum. 18. What information about an element is provided in the box for that el ...
Chapter 1
... B. Now for Some Neutrons C. Building Bigger Atoms D. Protons and Atomic Number *Notes-The number of protons in the nucleus of an atom give the element its _____________________. (also the number of electrons) ...
... B. Now for Some Neutrons C. Building Bigger Atoms D. Protons and Atomic Number *Notes-The number of protons in the nucleus of an atom give the element its _____________________. (also the number of electrons) ...
Dalton`s Atomic Theory
... • In chemical reactions (rxns), atoms are _________, _________, or _________. ...
... • In chemical reactions (rxns), atoms are _________, _________, or _________. ...
Document
... Democritus and the Atom section and explain how the student in figure 3 is illustrating Democritus’s ...
... Democritus and the Atom section and explain how the student in figure 3 is illustrating Democritus’s ...
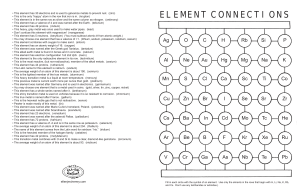
element connections
... • This element has 30 electrons and is used to galvanize metals to prevent rust. (zinc) • This is the only “happy” atom in the row that iron is in. (krypton) • This element is in the same row as silver and the same column as nitrogen. (antimony) • This element has a valence of 4 and was named after ...
... • This element has 30 electrons and is used to galvanize metals to prevent rust. (zinc) • This is the only “happy” atom in the row that iron is in. (krypton) • This element is in the same row as silver and the same column as nitrogen. (antimony) • This element has a valence of 4 and was named after ...
Ch 2 Atoms and Molecules
... • One amu is equivalent to: • A proton is 1.0073 amu and a neutron is 1.0087 amu • Angstroms (Å), which are 10^-10 m, are used to measure diameter ...
... • One amu is equivalent to: • A proton is 1.0073 amu and a neutron is 1.0087 amu • Angstroms (Å), which are 10^-10 m, are used to measure diameter ...
Atomic Number
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
... • Atoms are the building blocks of all materials • An atom is made of 3 parts: – Protons and Neutrons are in the nucleus (center) – Electrons orbit around the nucleus ...
Test #1 Study Guide
... grams mean different things across different elements, it is a way to standardize the amount of a substance. One mole of an element is equal to its atomic number Avagadro’s number indicates the amount of particles, molecules, etc. in a mole. There are always 6.022 x 10^23 particles in one mole of a ...
... grams mean different things across different elements, it is a way to standardize the amount of a substance. One mole of an element is equal to its atomic number Avagadro’s number indicates the amount of particles, molecules, etc. in a mole. There are always 6.022 x 10^23 particles in one mole of a ...
Atoms - SWThornton
... Two very important tenets ◦ Law of Conservation of Mass ◦ Law of Definite Proportions ...
... Two very important tenets ◦ Law of Conservation of Mass ◦ Law of Definite Proportions ...
Review for Chapter 2
... Review for Chapter 2: Atoms, Molecules, and Ions 1. Dalton’s Atomic Theory says: • Matter is composed of tiny, indivisible particles called “atoms”. • All atoms of the same element are identical. • Compounds contain atoms of different elements combined in whole-number ratios. • Atoms are combined or ...
... Review for Chapter 2: Atoms, Molecules, and Ions 1. Dalton’s Atomic Theory says: • Matter is composed of tiny, indivisible particles called “atoms”. • All atoms of the same element are identical. • Compounds contain atoms of different elements combined in whole-number ratios. • Atoms are combined or ...
Lesson 1 & 2 Periodic table trends and formation
... - some elements were out of order therefore modern table is arranged in Atomic Number Meyer recognised Mendeleev’s work and both where awarded The Davy medal for Chemistry in 1882. ...
... - some elements were out of order therefore modern table is arranged in Atomic Number Meyer recognised Mendeleev’s work and both where awarded The Davy medal for Chemistry in 1882. ...
Atomic History - Wylie High School Advanced Chemistry
... – How would you explain bullets ricocheting off to the left or right…. • The bullets hit “something” that was massive enough to deflect it. This was a very suprising finding and showed evidence for a MASSIVE part of the atom. The current model, the “Plum Pudding” model did not account for this data ...
... – How would you explain bullets ricocheting off to the left or right…. • The bullets hit “something” that was massive enough to deflect it. This was a very suprising finding and showed evidence for a MASSIVE part of the atom. The current model, the “Plum Pudding” model did not account for this data ...