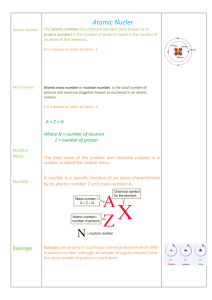
Jeopardy Nuclear Physics
... The time interval during which a nucleus has a 50% probability of decaying is its __________. ANSWER ...
... The time interval during which a nucleus has a 50% probability of decaying is its __________. ANSWER ...
Nuclear Chemistry
... • In nuclear fission, a very heavy nucleus splits into more-stable nuclei of intermediate mass. • Enormous amounts of energy are released. • Nuclear fission can occur spontaneously or when nuclei are bombarded by particles. ...
... • In nuclear fission, a very heavy nucleus splits into more-stable nuclei of intermediate mass. • Enormous amounts of energy are released. • Nuclear fission can occur spontaneously or when nuclei are bombarded by particles. ...
CHAPTER 1 INTRODUCTION 1.1 Preview
... penetrating radiation to nondestructively assess the physical integrity of selected materials and structures. The radiographic image is essentially a two-dimensional shadow display or picture of the intensity distribution of thermal neutrons that have passed through a material object. Although both ...
... penetrating radiation to nondestructively assess the physical integrity of selected materials and structures. The radiographic image is essentially a two-dimensional shadow display or picture of the intensity distribution of thermal neutrons that have passed through a material object. Although both ...
EPDG ILT Template - Nuclear Community
... 3. Explain the production of heat from fission. 4. Describe intrinsic and installed neutron sources and their contribution to source neutron strength over core life. 5. Explain the relationship between neutron flux, microscopic and macroscopic cross-sections; and their effect on neutron reaction rat ...
... 3. Explain the production of heat from fission. 4. Describe intrinsic and installed neutron sources and their contribution to source neutron strength over core life. 5. Explain the relationship between neutron flux, microscopic and macroscopic cross-sections; and their effect on neutron reaction rat ...
Study of Neutron and Gamma Radiation Protective
... on the number of electrons in the absorption material and, therefore, is independent of the atomic number of the absorber material. The Compton scattering coefficient decreases with increasing photon energy, but the rate of decline compared with the photoelectric effect, is much slower. In general, ...
... on the number of electrons in the absorption material and, therefore, is independent of the atomic number of the absorber material. The Compton scattering coefficient decreases with increasing photon energy, but the rate of decline compared with the photoelectric effect, is much slower. In general, ...
Module 6 Chemical Reactions
... controlled. Devices called control rods are inserted into the reactor to absorb some of the neutrons. This reduces the number of reactions that occur and controls the amount of energy the reactor produces. In a nuclear bomb, the chain reaction is uncontrolled. All of the energy is released at once. ...
... controlled. Devices called control rods are inserted into the reactor to absorb some of the neutrons. This reduces the number of reactions that occur and controls the amount of energy the reactor produces. In a nuclear bomb, the chain reaction is uncontrolled. All of the energy is released at once. ...
View Transcript
... And one of the first things that you’ll notice is that the data, for the most part, fall above the line of one neutron per proton. Another way to say that is that, except for hydrogen 1, just a plain old proton, and helium-3, which is 1 proton and 2 neutrons, all are stable nuclei – have 1 or more n ...
... And one of the first things that you’ll notice is that the data, for the most part, fall above the line of one neutron per proton. Another way to say that is that, except for hydrogen 1, just a plain old proton, and helium-3, which is 1 proton and 2 neutrons, all are stable nuclei – have 1 or more n ...
Extra revision sheet quarter 2 Physical science Grade 9
... 15. In the equation, E = mc2, what does the E stand for?............................................... 16. In the equation, E = mc2, what does the m stand for?............................................... 17. In the equation, E=mc2, what does c stand for?.......................................... ...
... 15. In the equation, E = mc2, what does the E stand for?............................................... 16. In the equation, E = mc2, what does the m stand for?............................................... 17. In the equation, E=mc2, what does c stand for?.......................................... ...
Section 19.1 Radioactivity
... Using the Nucleus as a Source of Energy A. Nuclear Energy • Two types of nuclear processes can produce energy – Combining 2 light nuclei to form a heavier nucleus fusion – Splitting a heavy nucleus into 2 nuclei with smaller mass numbers - fission ...
... Using the Nucleus as a Source of Energy A. Nuclear Energy • Two types of nuclear processes can produce energy – Combining 2 light nuclei to form a heavier nucleus fusion – Splitting a heavy nucleus into 2 nuclei with smaller mass numbers - fission ...
Physics HW Chapters 383940 (Due May 23, Test May 28)
... a. electrons are always ejected. b. electrons may be ejected. c. protons are always ejected. d. protons may be ejected. e. none of the above 10. The number of electrons ejected in the photoelectric effect depends on the light beam's a. amplitude. b. speed. c. frequency. d. intensity. e. wavelength. ...
... a. electrons are always ejected. b. electrons may be ejected. c. protons are always ejected. d. protons may be ejected. e. none of the above 10. The number of electrons ejected in the photoelectric effect depends on the light beam's a. amplitude. b. speed. c. frequency. d. intensity. e. wavelength. ...
An Unifying Basis for all the Nuclear Reactions Karunakar
... Even though we generate enormous amounts nuclear energy for the benefit of the world, we haven't understood the underlying physical processes which are the basis for these nuclear reactions. Expanded and collapsed forms of an object are not the same. Compact form of the object contains more energy t ...
... Even though we generate enormous amounts nuclear energy for the benefit of the world, we haven't understood the underlying physical processes which are the basis for these nuclear reactions. Expanded and collapsed forms of an object are not the same. Compact form of the object contains more energy t ...
File
... EB = mDc2 where c2 = 931.5 MeV/u EB = (0.030377 u)(931.5 MeV/u) = 28.3 MeV A total of 28.3 MeV is required To tear apart the nucleons from the He-4 atom. ...
... EB = mDc2 where c2 = 931.5 MeV/u EB = (0.030377 u)(931.5 MeV/u) = 28.3 MeV A total of 28.3 MeV is required To tear apart the nucleons from the He-4 atom. ...
Worksheet - Rudds Classroom
... How many neutrons does it have? _________________________ 6. Which element has 19 protons? ___________________________ How many neutrons does it have? _________________________ 7. Which element has 79 protons? ___________________________ How many neutrons does it have? _________________________ 8. W ...
... How many neutrons does it have? _________________________ 6. Which element has 19 protons? ___________________________ How many neutrons does it have? _________________________ 7. Which element has 79 protons? ___________________________ How many neutrons does it have? _________________________ 8. W ...
12_physics_notes_ch13_nuclei
... It is a device in which a nuclear chain reaction is initiated, maintained and controlled. • Nuclear Fusion: It is the process of fusion of two smaller nuclei into a heavier nucleus with the liberation of large amount of energy. • Critical size and Critical Mass: a) The size of the fissionable materi ...
... It is a device in which a nuclear chain reaction is initiated, maintained and controlled. • Nuclear Fusion: It is the process of fusion of two smaller nuclei into a heavier nucleus with the liberation of large amount of energy. • Critical size and Critical Mass: a) The size of the fissionable materi ...
Chapter 9 Nuclear Radiation
... E is the energy released, m is the mass lost, and c is the speed of light, 3.0 108 m/s. Using this equation, a small amount of mass is multiplied by the speed of light squared, resulting in a large amount of energy. Fission of 1 g U-235 produces same energy as 3 tons of coal. ...
... E is the energy released, m is the mass lost, and c is the speed of light, 3.0 108 m/s. Using this equation, a small amount of mass is multiplied by the speed of light squared, resulting in a large amount of energy. Fission of 1 g U-235 produces same energy as 3 tons of coal. ...
Chemistry Lecture No.4______By : Asst. Lect. Tariq-H-AL
... The radiation from radioactive isotopes and x-rays can form ions in matter by knocking electrons off the atoms and molecules in its path. For this reason, it is called ionizing radiation. The chief effects of radiation on living systems are due to these ionization reactions. Repeated exposure to low ...
... The radiation from radioactive isotopes and x-rays can form ions in matter by knocking electrons off the atoms and molecules in its path. For this reason, it is called ionizing radiation. The chief effects of radiation on living systems are due to these ionization reactions. Repeated exposure to low ...
Atomic Number - Physical Science
... • Earth receives a small amount of energy as thermal energy and light • As the Sun ages, the hydrogen nuclei are used up as they are converted into helium • It is estimated that the Sun has enough hydrogen to keep reacting for another 5 billion years ...
... • Earth receives a small amount of energy as thermal energy and light • As the Sun ages, the hydrogen nuclei are used up as they are converted into helium • It is estimated that the Sun has enough hydrogen to keep reacting for another 5 billion years ...
Fusion Video Workbook.Final - General Atomics Fusion Education
... The physics of a fusion device also make it inherently safer than a fission reactor. If something goes wrong in a fusion device, the plasma just cools down and the reaction stops. Chernobyl type accidents are not possible with a fusion power plant. ...
... The physics of a fusion device also make it inherently safer than a fission reactor. If something goes wrong in a fusion device, the plasma just cools down and the reaction stops. Chernobyl type accidents are not possible with a fusion power plant. ...
Physics 3 - Westmount High School
... This physicist was trained to be a nuclear physicist. S/he worked on the Manhattan Project during WWII, the secret project that built the first atomic bombs. One of the uranium isotopes (Uranium235, which has a mass of 235 atomic mass units) is a useful chemical element to split into lighter element ...
... This physicist was trained to be a nuclear physicist. S/he worked on the Manhattan Project during WWII, the secret project that built the first atomic bombs. One of the uranium isotopes (Uranium235, which has a mass of 235 atomic mass units) is a useful chemical element to split into lighter element ...
Radioactive decay of nucleus
... Energy is released because mass decreases, mass defect, and the difference is converted to energy. Momentum is conserved(linear) malpha x valpha = mdaughter x vdaughter 2. Beta-negative particles decay, β They are a stream of electrons released from nucleus Some beta particles are able to penetra ...
... Energy is released because mass decreases, mass defect, and the difference is converted to energy. Momentum is conserved(linear) malpha x valpha = mdaughter x vdaughter 2. Beta-negative particles decay, β They are a stream of electrons released from nucleus Some beta particles are able to penetra ...
An Unifying Basis for all the Nuclear Reactions
... It is widely believed that the source of Sun's energy is the fusion of plasma, the hydrogen nuclei, into lighter elements. The amount of mass that was deficit in the reaction was equated to the amount of energy released. Plasma is a densely packed group of protons, completely different from the gase ...
... It is widely believed that the source of Sun's energy is the fusion of plasma, the hydrogen nuclei, into lighter elements. The amount of mass that was deficit in the reaction was equated to the amount of energy released. Plasma is a densely packed group of protons, completely different from the gase ...
Nuclear Chemistry
... An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus. This particle, which consists of two protons and two neutrons, has a net positive charge. Although ...
... An alpha particle is simply a helium nuclei (He) which is ejected with high energy from an unstable nucleus. This particle, which consists of two protons and two neutrons, has a net positive charge. Although ...
Atomic Nuclei - RAJEEV Classes
... a)It is the phenomenon of spontaneous disintegration of the nucleus of an atom with the emission of one or more radiations like α-particles, β-particles or γ-rays. b)The substances which spontaneously emit penetratingradiation are called radioactive ...
... a)It is the phenomenon of spontaneous disintegration of the nucleus of an atom with the emission of one or more radiations like α-particles, β-particles or γ-rays. b)The substances which spontaneously emit penetratingradiation are called radioactive ...
Nickel-Hydrogen Cold Fusion by Intermediate Rydberg State of
... 2. Theoretical base of the objections against cold fusion Why nuclear fusion is considered impossible at accessible temperatures? The quantum mechanical (QM) models of atoms are based on the Rutherford-Bohr planetary model of hydrogen. The nuclear size is determined by scattering experiments, while ...
... 2. Theoretical base of the objections against cold fusion Why nuclear fusion is considered impossible at accessible temperatures? The quantum mechanical (QM) models of atoms are based on the Rutherford-Bohr planetary model of hydrogen. The nuclear size is determined by scattering experiments, while ...