Teacher Resource 5: Monoclonal antibodies
... Teachers will need to photocopy and cut out a set of cards for learners to sequence, one set per pair. ...
... Teachers will need to photocopy and cut out a set of cards for learners to sequence, one set per pair. ...
Antibodies - INAYA Medical College
... • Serologic, genetic, biochemical and molecular study of antigens associated with membrane structures on the cellular constituents of the blood ...
... • Serologic, genetic, biochemical and molecular study of antigens associated with membrane structures on the cellular constituents of the blood ...
Antibody
... 3. IgA is produced in 2nd immune response and plays a key role in mucosa immunity area (eg. Respiratory & GI tracts). 4. IgD is a membrane-bound Ag receptor on B cells. 5. IgE have evolved to protect against helminth parasites. ...
... 3. IgA is produced in 2nd immune response and plays a key role in mucosa immunity area (eg. Respiratory & GI tracts). 4. IgD is a membrane-bound Ag receptor on B cells. 5. IgE have evolved to protect against helminth parasites. ...
2 Antibodies - WordPress.com
... B-cells B-cell binds to antigen. B-cell divides by mitosis. Some cells formed are plasma cells – secrete antibodies. Some cells formed are memory cells – remain in blood for a period of time, providing ...
... B-cells B-cell binds to antigen. B-cell divides by mitosis. Some cells formed are plasma cells – secrete antibodies. Some cells formed are memory cells – remain in blood for a period of time, providing ...
No Slide Title
... two different ways, RNA splicing and DNA switch recombination. The signals regulating these changes come from antigen binding to the B cell receptor and antigen specific T cells. During the immune response B cells mutate their immunoglobulin variable regions under the control of T cells and other si ...
... two different ways, RNA splicing and DNA switch recombination. The signals regulating these changes come from antigen binding to the B cell receptor and antigen specific T cells. During the immune response B cells mutate their immunoglobulin variable regions under the control of T cells and other si ...
BLOCK F – Krizia,Kevin,Synnove – Production of Antibodies
... matching antigens. 7. Antibodies are made up of white blood cells, called B lymphocytes or B Cells. Each B Cell carries a different membrane-bound antibody molecule on its surface that serves as a receptor for recognizing a specific antigen. When antigen binds to this receptor, the B cell is stimula ...
... matching antigens. 7. Antibodies are made up of white blood cells, called B lymphocytes or B Cells. Each B Cell carries a different membrane-bound antibody molecule on its surface that serves as a receptor for recognizing a specific antigen. When antigen binds to this receptor, the B cell is stimula ...
antibody antigen interaction
... Antigen-antibody reaction, is a specific chemical interaction between antibodies produced by B cells of the white blood cells and antigens during immune reaction. It is the fundamental reaction in the body by which the body is protected from complex foreign molecules. Biological Aspects of Antibody- ...
... Antigen-antibody reaction, is a specific chemical interaction between antibodies produced by B cells of the white blood cells and antigens during immune reaction. It is the fundamental reaction in the body by which the body is protected from complex foreign molecules. Biological Aspects of Antibody- ...
ELISA Pre and Post Test
... a. first line defenses; b. nonspecific immunities; c. specific immunities; d. both a and b. 3. An antigen is: a. a protein or other molecule that can be separate or found on a pathogen and is foreign to your body; b. a protein made in response to a specific pathogen used to limit pathogen’s growth; ...
... a. first line defenses; b. nonspecific immunities; c. specific immunities; d. both a and b. 3. An antigen is: a. a protein or other molecule that can be separate or found on a pathogen and is foreign to your body; b. a protein made in response to a specific pathogen used to limit pathogen’s growth; ...
Lecture 5 - Andrew.cmu.edu
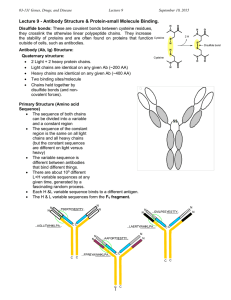
... Disulfide bonds: These are covalent bonds between cysteine residues, they crosslink the otherwise linear polypeptide chains. They increase the stability of proteins and are often found on proteins that function Cysteine outside of cells, such as antibodies. Antibody (Ab, Ig) Structure: Quaternary st ...
... Disulfide bonds: These are covalent bonds between cysteine residues, they crosslink the otherwise linear polypeptide chains. They increase the stability of proteins and are often found on proteins that function Cysteine outside of cells, such as antibodies. Antibody (Ab, Ig) Structure: Quaternary st ...
ANTIBODY PRODUCTION
... Antigens from the ruined pathogen are displayed on the surface of the phagocyte (or B-cell), bound to a membrane called MHC protein. This combination of antigen and MHC attracts the help of a mature, matching Helper T Cell. ...
... Antigens from the ruined pathogen are displayed on the surface of the phagocyte (or B-cell), bound to a membrane called MHC protein. This combination of antigen and MHC attracts the help of a mature, matching Helper T Cell. ...
Antibodies - blobs.org
... and cells to label them as invaders and stop them attacking the body. They are produced by special cells in the immune system called plasma cells, which are mature B lymphocytes. Each antibody is made up of light chains and heavy chains, connected by a disulphide bond. The structure of the antibody ...
... and cells to label them as invaders and stop them attacking the body. They are produced by special cells in the immune system called plasma cells, which are mature B lymphocytes. Each antibody is made up of light chains and heavy chains, connected by a disulphide bond. The structure of the antibody ...
Protection against Disease
... The interlocking of and antibody and antigen could render a toxic antigen harmless if its active region was blocked by an antibody molecule Phagocytes can more easily track down and ingest pathogens if they are immobilised in an AAC The AAC also stimulates the activation of a number of plasma ...
... The interlocking of and antibody and antigen could render a toxic antigen harmless if its active region was blocked by an antibody molecule Phagocytes can more easily track down and ingest pathogens if they are immobilised in an AAC The AAC also stimulates the activation of a number of plasma ...
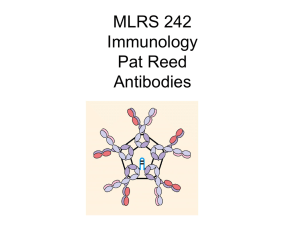
BMT 242 Immunology
... • 5 different isotypes identified: based on type of heavy chain: G,D,E,M,A • Human light chains: 60% kappa (K) chains, 40% lambda (L) chains ...
... • 5 different isotypes identified: based on type of heavy chain: G,D,E,M,A • Human light chains: 60% kappa (K) chains, 40% lambda (L) chains ...
Crystal Structures of Shark Ig New Antigen Receptor Variable
... Sharks are the most primitive animals to have an advanced adaptive immune system. Their long evolutionary history (~400 million years) is reflected in a diverse array of shark antibodies, including the unique IgNAR (Ig new antigen receptor) isotype. IgNARs are heavy chain homodimers, there is no ass ...
... Sharks are the most primitive animals to have an advanced adaptive immune system. Their long evolutionary history (~400 million years) is reflected in a diverse array of shark antibodies, including the unique IgNAR (Ig new antigen receptor) isotype. IgNARs are heavy chain homodimers, there is no ass ...
Antibody
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shape protein produced by plasma cells that is used by the immune system to identify and neutralize pathogens such as bacteria and viruses. The antibody recognizes a unique molecule of the harmful agent, called an antigen, via the variable region. Each tip of the ""Y"" of an antibody contains a paratope (analogous to a lock) that is specific for one particular epitope (similarly analogous to a key) on an antigen, allowing these two structures to bind together with precision. Using this binding mechanism, an antibody can tag a microbe or an infected cell for attack by other parts of the immune system, or can neutralize its target directly (for example, by blocking a part of a microbe that is essential for its invasion and survival). The ability of an antibody to communicate with the other components of the immune system is mediated via its Fc region (located at the base of the ""Y""), which contains a conserved glycosylation site involved in these interactions. The production of antibodies is the main function of the humoral immune system.Antibodies are secreted by cells of the adaptive immune system (B cells), and more specifically, differentiated B cells called plasma cells. Antibodies can occur in two physical forms, a soluble form that is secreted from the cell, and a membrane-bound form that is attached to the surface of a B cell and is referred to as the B cell receptor (BCR). The BCR is found only on the surface of B cells and facilitates the activation of these cells and their subsequent differentiation into either antibody factories called plasma cells or memory B cells that will survive in the body and remember that same antigen so the B cells can respond faster upon future exposure. In most cases, interaction of the B cell with a T helper cell is necessary to produce full activation of the B cell and, therefore, antibody generation following antigen binding. Soluble antibodies are released into the blood and tissue fluids, as well as many secretions to continue to survey for invading microorganisms.Antibodies are glycoproteins belonging to the immunoglobulin superfamily; the terms antibody and immunoglobulin are often used interchangeably. Though strictly speaking, an antibody is not the same as an immunoglobulin; B cells can produce two types of immunoglobulins - surface immunoglobulins, which are B cell receptors; and secreted immunoglobulins, which are antibodies. So antibodies are one of two classes of immunoglobulins. Antibodies are typically made of basic structural units—each with two large heavy chains and two small light chains. There are several different types of antibody heavy chains based on five different types of crystallisable fragments (Fc) that may be attached to the antigen-binding fragments. The five different types of Fc regions allow antibodies to be grouped into five isotypes. Each Fc region of a particular antibody isotype is able to bind to its specific Fc Receptor (except for IgD, which is essentially the BCR), thus allowing the antigen-antibody complex to mediate different roles depending on which FcR it binds. The ability of an antibody to bind to its corresponding FcR is further modulated by the structure of the glycan(s) present at conserved sites within its Fc region. The ability of antibodies to bind to FcRs helps to direct the appropriate immune response for each different type of foreign object they encounter. For example, IgE is responsible for an allergic response consisting of mast cell degranulation and histamine release. IgE's Fab paratope binds to allergic antigen, for example house dust mite particles, while its Fc region binds to Fc receptor ε. The allergen-IgE-FcRε interaction mediates allergic signal transduction to induce conditions such as asthma. Though the general structure of all antibodies is very similar, a small region at the tip of the protein is extremely variable, allowing millions of antibodies with slightly different tip structures, or antigen-binding sites, to exist. This region is known as the hypervariable region. Each of these variants can bind to a different antigen. This enormous diversity of antibody paratopes on the antigen-binding fragments allows the immune system to recognize an equally wide variety of antigens. The large and diverse population of antibody paratope is generated by random recombination events of a set of gene segments that encode different antigen-binding sites (or paratopes), followed by random mutations in this area of the antibody gene, which create further diversity. This recombinational process that produces clonal antibody paratope diversity is called V(D)J or VJ recombination. Basically, the antibody paratope is polygenic, made up of three genes, V, D, and J. Each paratope locus is also polymorphic, such that during antibody production, one allele of V, one of D, and one of J is chosen. These gene segments are then joined together using random genetic recombination to produce the paratope. The regions where the genes are randomly recombined together is the hyper variable region used to recognise different antigens on a clonal basis. Antibody genes also re-organize in a process called class switching that changes the one type of heavy chain Fc fragment to another, creating a different isotype of the antibody that retains the antigen-specific variable region. This allows a single antibody to be used by different types of Fc receptors, expressed on different parts of the immune system.