Chapter 2 Matter and Components F11 110
... 3. For Type II metals with only two common oxidation states an older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root na ...
... 3. For Type II metals with only two common oxidation states an older, Latin system was once used; while it is not employed very often it is useful to know some simple rules regarding it. It is sometimes called the “-ous/-ic” system, where the LOWER charged cation will be denoted by the latin root na ...
Acid-Base Equilibria - Riverside Local Schools
... acids and bases by their characteristic properties. Acids have a sour taste (for example, citric acid in lemon juice) and cause certain dyes to change color (for example, litmus turns red on contact with acids). Indeed, the word acid comes from the Latin word acidus, meaning sour or tart. Bases, in ...
... acids and bases by their characteristic properties. Acids have a sour taste (for example, citric acid in lemon juice) and cause certain dyes to change color (for example, litmus turns red on contact with acids). Indeed, the word acid comes from the Latin word acidus, meaning sour or tart. Bases, in ...
Chem13-14PrecipABNeut
... with extensive memory during tests. If a type of calculator is specified for your course, buy two if possible. When one becomes broken or lost, you will have a familiar backup if the bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calcu ...
... with extensive memory during tests. If a type of calculator is specified for your course, buy two if possible. When one becomes broken or lost, you will have a familiar backup if the bookstore is sold out later in the term. If no type of calculator is specified for your course, any inexpensive calcu ...
REACTIONS IN AQUEOUS SOLUTION
... Not all substances that dissolve in water make the resulting solution conducting. Imagine preparing two aqueous solutions—one by dissolving a teaspoon of table salt (sodium chloride) in a cup of water and the other by dissolving a teaspoon of table sugar (sucrose) in a cup of water (! FIGURE 4.2). B ...
... Not all substances that dissolve in water make the resulting solution conducting. Imagine preparing two aqueous solutions—one by dissolving a teaspoon of table salt (sodium chloride) in a cup of water and the other by dissolving a teaspoon of table sugar (sucrose) in a cup of water (! FIGURE 4.2). B ...
Chapter 4 Aqueous Reactions and Solution Stoichiometry
... The solubility of a substance at a particular temperature is the amount of that substance that can be dissolved in a given quantity of solvent at that temperature. • A substance with a solubility of less than 0.01 mol/L is regarded as being insoluble. Aqueous Reactions ...
... The solubility of a substance at a particular temperature is the amount of that substance that can be dissolved in a given quantity of solvent at that temperature. • A substance with a solubility of less than 0.01 mol/L is regarded as being insoluble. Aqueous Reactions ...
Magic of Chemical Reactions 2. - mt
... 1. Potassium ferrocyanide, (molecular formula is K 4[Fe(CN)6].3H2O) is a yellow coloured compound. 2. If this compound gets exposed to sunlight then it will undergo decomposition reaction. 3. Therefore, potassium ferrocyanide is stored in dark coloured bottles and kept away from sunlight. Iron artic ...
... 1. Potassium ferrocyanide, (molecular formula is K 4[Fe(CN)6].3H2O) is a yellow coloured compound. 2. If this compound gets exposed to sunlight then it will undergo decomposition reaction. 3. Therefore, potassium ferrocyanide is stored in dark coloured bottles and kept away from sunlight. Iron artic ...
Use the following answers for questions 1
... 5. Is used to etch glass chemically 6. Is used extensively for the production of fertilizers 7. Has amphoteric properties 63. Which of the following characteristics is common to elemental sulfur, chlorine, nitrogen, and carbon? (A) They are gaseous elements at room temperature. (B) They have oxides ...
... 5. Is used to etch glass chemically 6. Is used extensively for the production of fertilizers 7. Has amphoteric properties 63. Which of the following characteristics is common to elemental sulfur, chlorine, nitrogen, and carbon? (A) They are gaseous elements at room temperature. (B) They have oxides ...
Appendix
... The elements of Group 1 are important to a person’s diet and body maintenance because they form ionic compounds. These compounds are present in the body as solutions of the ions. All ions carry an electric charge, so they are electrolyte solutes. Two of the most important electrolyte solutes found i ...
... The elements of Group 1 are important to a person’s diet and body maintenance because they form ionic compounds. These compounds are present in the body as solutions of the ions. All ions carry an electric charge, so they are electrolyte solutes. Two of the most important electrolyte solutes found i ...
Lab Manual Quantitative Analytical Method
... 4. Burette for the NaOH Clean, rinse well (with deionized water), and dry three 125-mL or 250-mL conical flasks. Measure between 0.20 – 0.25 g of potassium hydrogen phthalate into each. conical flask. Record Clean, rinse well (with deionized water), and dry three 125-mL or 250-mL Conical flasks. Mea ...
... 4. Burette for the NaOH Clean, rinse well (with deionized water), and dry three 125-mL or 250-mL conical flasks. Measure between 0.20 – 0.25 g of potassium hydrogen phthalate into each. conical flask. Record Clean, rinse well (with deionized water), and dry three 125-mL or 250-mL Conical flasks. Mea ...
chemical reaction equation - parmod cobra insititution.
... 2AgNO3(aq) + Cu(s) Cu(NO 3)2(aq) + 2Ag(s) (silver nitrate) (copper) (copper nitrate) ...
... 2AgNO3(aq) + Cu(s) Cu(NO 3)2(aq) + 2Ag(s) (silver nitrate) (copper) (copper nitrate) ...
CHE 1402 Lab Manual
... 1 How does the pressure of an ideal gas at constant volume change as the temperature increases? 2 How does the volume of an ideal gas at constant temperature change as the pressure increases? 3 How does the volume of an ideal gas at constant temperature and pressure change as the number of molecules ...
... 1 How does the pressure of an ideal gas at constant volume change as the temperature increases? 2 How does the volume of an ideal gas at constant temperature change as the pressure increases? 3 How does the volume of an ideal gas at constant temperature and pressure change as the number of molecules ...
CHAPTER 4 - Myschoolpages.com
... – formation of an insoluble solid – formation of either a soluble weak electrolyte or soluble nonelectrolyte – formation of a gas which escapes from solution Chapt. 4.2 Chem 106, Prof. J.T. Spencer ...
... – formation of an insoluble solid – formation of either a soluble weak electrolyte or soluble nonelectrolyte – formation of a gas which escapes from solution Chapt. 4.2 Chem 106, Prof. J.T. Spencer ...
Form 3 Chem. Term 1 Notes.FINAL.
... To investigate the rate of diffusion of Hydrogen Chloride and Ammonia Clamp a long glass tube horizontally with clamps. At the same time plug one end of the long tube with cotton wool soaked in concentrated Ammonia gas solution and the other end with another cotton wool soaked in concentrated hydroc ...
... To investigate the rate of diffusion of Hydrogen Chloride and Ammonia Clamp a long glass tube horizontally with clamps. At the same time plug one end of the long tube with cotton wool soaked in concentrated Ammonia gas solution and the other end with another cotton wool soaked in concentrated hydroc ...
Lab #3 – A Microscale Study of Chemical Changes
... A precipitate is a solid that separates upon mixing solutions. Which reaction produced a very bright yellow precipitate? ...
... A precipitate is a solid that separates upon mixing solutions. Which reaction produced a very bright yellow precipitate? ...
Kinetics and Mechanism of Uncatalyzed and Ag (I) Catalyzed
... sulphuric acid medium and sulphato complexes, such as CeSO42+, Ce(SO4)2 and Ce(SO4)32- have been established and quantified [26]. However, cerium (IV) in perchloric acid medium does not indicate complex formation, although Ce4+, Ce(OH)3+, (Ce-O-Ce)6+ and (HOCe-O-CeOH)4+ species of cerium (IV) are we ...
... sulphuric acid medium and sulphato complexes, such as CeSO42+, Ce(SO4)2 and Ce(SO4)32- have been established and quantified [26]. However, cerium (IV) in perchloric acid medium does not indicate complex formation, although Ce4+, Ce(OH)3+, (Ce-O-Ce)6+ and (HOCe-O-CeOH)4+ species of cerium (IV) are we ...
analytical chemistry - Львівський національний медичний
... form various acids and acids force increases with increasing of element oxidation degree. For oxidising-reducing properties the anions divide on anions-oxidisers with high oxidation degree (nitrate-anion), anions-reducers with lower oxidation degree (chlorides, bromides, iodides) and neutral anions, ...
... form various acids and acids force increases with increasing of element oxidation degree. For oxidising-reducing properties the anions divide on anions-oxidisers with high oxidation degree (nitrate-anion), anions-reducers with lower oxidation degree (chlorides, bromides, iodides) and neutral anions, ...
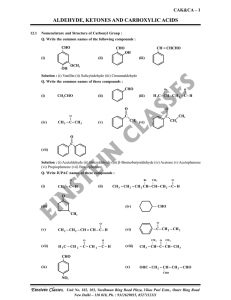
aldehyde, ketones and carboxylic acids
... intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehydes are more reactive than Ketones ? Solution : There are two reasons for this, they are as follows : 1. Steri ...
... intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehydes are more reactive than Ketones ? Solution : There are two reasons for this, they are as follows : 1. Steri ...
CHAPTER 23 THE TRANSITION ELEMENTS AND THEIR
... chromium in CrO3 is +6 and in CrO is +2, based on the -2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
... chromium in CrO3 is +6 and in CrO is +2, based on the -2 oxidation state of oxygen. The oxide of the higher oxidation state, CrO3, produces a more acidic solution. ...
Acid - Net Texts
... There are three common definitions for acids: the Arrhenius definition, the Brønsted-Lowry definition, and the Lewis definition. The Arrhenius definition states that acids are substances which increase the concentration of hydronium ions (H3O+) in solution. The Brønsted-Lowry definition is an expans ...
... There are three common definitions for acids: the Arrhenius definition, the Brønsted-Lowry definition, and the Lewis definition. The Arrhenius definition states that acids are substances which increase the concentration of hydronium ions (H3O+) in solution. The Brønsted-Lowry definition is an expans ...
Word Pro
... This is the contents of a Quiz 1 from a few years ago in the days of Chemistry 1000. (It has been reformatted to save paper) Answer ALL of the questions in the spaces provided. The mark that you obtain for this test will be used in calculating your final grade for the course. 1. Name the following c ...
... This is the contents of a Quiz 1 from a few years ago in the days of Chemistry 1000. (It has been reformatted to save paper) Answer ALL of the questions in the spaces provided. The mark that you obtain for this test will be used in calculating your final grade for the course. 1. Name the following c ...
Supplementary Exercise 1B Topic 5
... In the electrochemical series, the position of calcium is higher than that of sodium. The order is different from that in the reactivity series. This is because calcium atom loses electrons more readily in cell reactions than in reaction with air, water and dilute acids. ...
... In the electrochemical series, the position of calcium is higher than that of sodium. The order is different from that in the reactivity series. This is because calcium atom loses electrons more readily in cell reactions than in reaction with air, water and dilute acids. ...
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. A hydroxide attached to a strongly electropositive center may itself ionize, liberating a hydrogen cation (H+), making the parent compound an acid.The corresponding electrically neutral compound •HO is the hydroxyl radical. The corresponding covalently-bound group -OH of atoms is the hydroxyl group.Hydroxide ion and hydroxyl group are nucleophiles and can act as a catalyst in organic chemistry.Many inorganic substances which bear the word ""hydroxide"" in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxyl groups.