The Second Law of Thermodynamics and Entropy
... Having established that temperature difference (or more strictly, temperature gradient) is a thennodynamic driving potential for work production, one can easily recognize that there are other thermodynamic potentials for work production, such as pressl,lre gradients (piston engines and turbines, for ...
... Having established that temperature difference (or more strictly, temperature gradient) is a thennodynamic driving potential for work production, one can easily recognize that there are other thermodynamic potentials for work production, such as pressl,lre gradients (piston engines and turbines, for ...
2013 8th Grade Physical Science End of Year Exam Study Guide
... Know how the terms solvent, solute, and concentration relate to each other. What is a mixture? How is a mixture and compound different? Describe the properties of acids and bases and know examples. ...
... Know how the terms solvent, solute, and concentration relate to each other. What is a mixture? How is a mixture and compound different? Describe the properties of acids and bases and know examples. ...
7. The Second Law and the Concept of Entropy
... No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature. Spontaneously, heat cannot flow from cold regions to hot regions without external work being performed on the system, which is evident from ordinary experience of refrig ...
... No process is possible whose sole result is the transfer of heat from a body of lower temperature to a body of higher temperature. Spontaneously, heat cannot flow from cold regions to hot regions without external work being performed on the system, which is evident from ordinary experience of refrig ...
Document
... temperature and chemical composition of stars • Identified the spectral sequence as a temperature sequence and correctly concluded that the large variations in absorption lines seen in stars is due to ionization and not abundances • Found abundances of silicon, carbon, etc on sun similar to earth • ...
... temperature and chemical composition of stars • Identified the spectral sequence as a temperature sequence and correctly concluded that the large variations in absorption lines seen in stars is due to ionization and not abundances • Found abundances of silicon, carbon, etc on sun similar to earth • ...
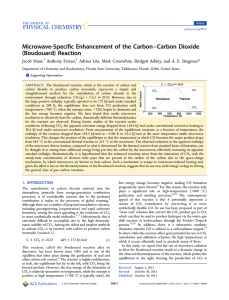
Microwave-Specific Enhancement of the Carbon−Carbon Dioxide
... Thermal. For flow reactions carried out under thermal conditions, a reaction tube of the same diameter and having the same mass of carbon, spread out over a quartz frit, is brought up to the desired temperature in a Lindberg tube furnace packed with quartz wool to insulate the tube. After the system ...
... Thermal. For flow reactions carried out under thermal conditions, a reaction tube of the same diameter and having the same mass of carbon, spread out over a quartz frit, is brought up to the desired temperature in a Lindberg tube furnace packed with quartz wool to insulate the tube. After the system ...
Chapter Two The Thermodynamic Laws
... an energy transfer by heat from a cooler to a hotter body. Another statement by Clausius is: "Heat cannot of itself pass from a colder to a hotter body." This statement implies an inequality of the heat transfer between a hot body and a cold body. Heat transfer from a hot body to a cold body can spo ...
... an energy transfer by heat from a cooler to a hotter body. Another statement by Clausius is: "Heat cannot of itself pass from a colder to a hotter body." This statement implies an inequality of the heat transfer between a hot body and a cold body. Heat transfer from a hot body to a cold body can spo ...
Material
... uniform temperature is attained throughout the combined system. The system is then in thermal equilibrium. Experience shows, all systems which are in thermal equilibrium with a given system are also in thermal equilibrium with each other. This principle defines the temperature of a thermodynamic sys ...
... uniform temperature is attained throughout the combined system. The system is then in thermal equilibrium. Experience shows, all systems which are in thermal equilibrium with a given system are also in thermal equilibrium with each other. This principle defines the temperature of a thermodynamic sys ...
q 2 - q 1
... obtained from quantity of that ( q′1- q1 ) by absorbing it from the low- temperature heat reservoir without leaving aching in any other thermodynamic system . Though this is in agreement with the first law of thermodynamics , it conflict with human experience fact of impossibility of certain of a pe ...
... obtained from quantity of that ( q′1- q1 ) by absorbing it from the low- temperature heat reservoir without leaving aching in any other thermodynamic system . Though this is in agreement with the first law of thermodynamics , it conflict with human experience fact of impossibility of certain of a pe ...
Energy and Exergy Analysis of Organic Rankine
... Heat is recovered by organic Rankine cycles (ORC).Low grade heat is useful for operation of Organic Rankine cycle the Low grade heat is available from various heat sources such as solar ,ocean , thermal , geothermal, biomass, and available source of heat from engine exhaust or industrial process was ...
... Heat is recovered by organic Rankine cycles (ORC).Low grade heat is useful for operation of Organic Rankine cycle the Low grade heat is available from various heat sources such as solar ,ocean , thermal , geothermal, biomass, and available source of heat from engine exhaust or industrial process was ...
Chapter 2 Classical Thermodynamics: The Second Law 2.1 Heat
... This is a substantial chapter, containing many important results and many techniques. There are two common technical difficulties for many students at the beginning: proof of theorems and partial derivatives. We will emphasize the understanding and applications of the theorems and we will do many pr ...
... This is a substantial chapter, containing many important results and many techniques. There are two common technical difficulties for many students at the beginning: proof of theorems and partial derivatives. We will emphasize the understanding and applications of the theorems and we will do many pr ...
253 Chapter 12 Thermodynamics GOALS When you have mastered
... Much of your travel is made possible by a device that we call a heat engine. We can think of a heat engine as being any device which operates by heat energy input, does work, and has a heat energy output. The human body is a heat engine. The refrigerator in your home is a type of heat engine. In thi ...
... Much of your travel is made possible by a device that we call a heat engine. We can think of a heat engine as being any device which operates by heat energy input, does work, and has a heat energy output. The human body is a heat engine. The refrigerator in your home is a type of heat engine. In thi ...
THERMODYNAMICS LECTURE NOTES
... disregard the atomic nature of a substance and view it as a continuous, homogeneous matter with no holes, that is, a continuum. In Macroscopic approach of thermodynamics the substance is considered to be continuous whereas every matter actually comprises of myriads of molecules with intermolecular s ...
... disregard the atomic nature of a substance and view it as a continuous, homogeneous matter with no holes, that is, a continuum. In Macroscopic approach of thermodynamics the substance is considered to be continuous whereas every matter actually comprises of myriads of molecules with intermolecular s ...
Slide 1
... it contracts in such a way that it occupies a volume that is only the size of the helium atoms at –273.15°C. • At this temperature, all the thermal energy that can be removed has been removed from the gas. • It is impossible to reduce the temperature any further. • Therefore, there can be no tempera ...
... it contracts in such a way that it occupies a volume that is only the size of the helium atoms at –273.15°C. • At this temperature, all the thermal energy that can be removed has been removed from the gas. • It is impossible to reduce the temperature any further. • Therefore, there can be no tempera ...
6 The ocean lithosphere
... about 1o C/km in the solid mantle. If the system becomes partly molten, then the change in volume with pressure is larger and T changes more quickly. Since the temperature of the convective mantle is not constant but lies on an adiabat we characterize it by its potential temperature (Tm ), which is ...
... about 1o C/km in the solid mantle. If the system becomes partly molten, then the change in volume with pressure is larger and T changes more quickly. Since the temperature of the convective mantle is not constant but lies on an adiabat we characterize it by its potential temperature (Tm ), which is ...
Thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of charged particles in matter. An object with a temperature greater than absolute zero emits thermal radiation. When the temperature of the body is greater than absolute zero, interatomic collisions cause the kinetic energy of the atoms or molecules to change. This results in charge-acceleration and/or dipole oscillation which produces electromagnetic radiation, and the wide spectrum of radiation reflects the wide spectrum of energies and accelerations that occur even at a single temperature.Examples of thermal radiation include the visible light and infrared light emitted by an incandescent light bulb, the infrared radiation emitted by animals and detectable with an infrared camera, and the cosmic microwave background radiation. Thermal radiation is different from thermal convection and thermal conduction—a person near a raging bonfire feels radiant heating from the fire, even if the surrounding air is very cold.Sunlight is part of thermal radiation generated by the hot plasma of the Sun. The Earth also emits thermal radiation, but at a much lower intensity and different spectral distribution (infrared rather than visible) because it is cooler. The Earth's absorption of solar radiation, followed by its outgoing thermal radiation are the two most important processes that determine the temperature and climate of the Earth.If a radiation-emitting object meets the physical characteristics of a black body in thermodynamic equilibrium, the radiation is called blackbody radiation. Planck's law describes the spectrum of blackbody radiation, which depends only on the object's temperature. Wien's displacement law determines the most likely frequency of the emitted radiation, and the Stefan–Boltzmann law gives the radiant intensity.Thermal radiation is one of the fundamental mechanisms of heat transfer.