PPT
... • First, we’ll figure out where vapor, liquid and ice are in equilibrium, and the energy changes associated with the transitions – Can calculate changes such as heat released so we can treat the energy changes in our parcel – Can also figure out if the change is favored (e.g., if the transition is a ...
... • First, we’ll figure out where vapor, liquid and ice are in equilibrium, and the energy changes associated with the transitions – Can calculate changes such as heat released so we can treat the energy changes in our parcel – Can also figure out if the change is favored (e.g., if the transition is a ...
Science-M2-Basic-Che..
... boiling – This change is the rapid vaporization of a liquid, which typically occurs when a liquid is heated to its boiling point: the temperature at which the vapor pressure of the liquid is equal to the pressure exerted on the liquid by the surrounding environmental pressure. Sometimes a liquid may ...
... boiling – This change is the rapid vaporization of a liquid, which typically occurs when a liquid is heated to its boiling point: the temperature at which the vapor pressure of the liquid is equal to the pressure exerted on the liquid by the surrounding environmental pressure. Sometimes a liquid may ...
Advanced Placement Chemistry Test
... When the substances in the equation above are at equilibrium at pressure P and temperature T, the equilibrium can be shifted to favor the products by (A) increasing the pressure by means of a moving piston at constant T (B) increasing the pressure by adding an inert gas such as nitrogen (C) decreasi ...
... When the substances in the equation above are at equilibrium at pressure P and temperature T, the equilibrium can be shifted to favor the products by (A) increasing the pressure by means of a moving piston at constant T (B) increasing the pressure by adding an inert gas such as nitrogen (C) decreasi ...
File
... 68. The phase diagram for a pure substance is shown above. The solid and gaseous phases of the substances can exist in equilibrium at conditions corresponding to which of the following? A) Point I only B) Point III only C) Any point on the curve from I to II. D) Any point on the curve from II to II ...
... 68. The phase diagram for a pure substance is shown above. The solid and gaseous phases of the substances can exist in equilibrium at conditions corresponding to which of the following? A) Point I only B) Point III only C) Any point on the curve from I to II. D) Any point on the curve from II to II ...
CHAPTER 1-MATTER AND ITS PROPERTIES The
... used in ____the paper industry____ 18. In order to seperate iron powder and pellicle components from a mixture we use the method of ____Floating and Precipitation______ 19. If a heterogenous mixture comprises solid and liquid components with different densities, than a further precipitation process ...
... used in ____the paper industry____ 18. In order to seperate iron powder and pellicle components from a mixture we use the method of ____Floating and Precipitation______ 19. If a heterogenous mixture comprises solid and liquid components with different densities, than a further precipitation process ...
Mr Alasdair Ross at Southpointe Academy
... The vapour pressure of the solvent above a solution ( Psolv ) is the product of the vapour pressure of the pure solvent ( P 0 solv ) and the mole fraction of the solvent in solution ( x solv ). Psolv x solv P 0 solv ...
... The vapour pressure of the solvent above a solution ( Psolv ) is the product of the vapour pressure of the pure solvent ( P 0 solv ) and the mole fraction of the solvent in solution ( x solv ). Psolv x solv P 0 solv ...
Chem 430 - Fall 1994
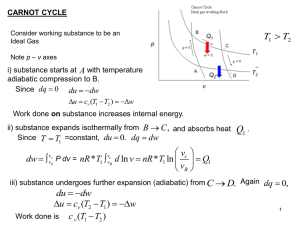
... Chem 430 - Fall 2001 - Test 1 - Page 5 ( 2) 10. What is the SSYS for one cycle of a Carnot Engine? ...
... Chem 430 - Fall 2001 - Test 1 - Page 5 ( 2) 10. What is the SSYS for one cycle of a Carnot Engine? ...
Phase-separation in ion-containing mixtures in electric fields
... tant because they bring about large field gradients will slide with a certain velocity vm with respect to even in a flat electrode geometry. Field gradients each other. In the presence of electric field, (demixed state), give rise to a dielectrophoretic force which acts to pull the liquid with high ...
... tant because they bring about large field gradients will slide with a certain velocity vm with respect to even in a flat electrode geometry. Field gradients each other. In the presence of electric field, (demixed state), give rise to a dielectrophoretic force which acts to pull the liquid with high ...
CYL100 2013–14 I semester Homework 2 Solutions 1. Consider a
... We see that Y = − G T , which suggests that (dY )T,P ≥ 0 would be the condition for spontaneity and equilibrium. 11. It is often claimed that one can skate on ice because the pressure of the skate causes the ice to melt, thus dramatically reducing the friction between skate and ice. While this makes ...
... We see that Y = − G T , which suggests that (dY )T,P ≥ 0 would be the condition for spontaneity and equilibrium. 11. It is often claimed that one can skate on ice because the pressure of the skate causes the ice to melt, thus dramatically reducing the friction between skate and ice. While this makes ...
ALB974_STABROM_MAXXIS TankEquip
... Density, 0 °C, g/cm3 (lbs/gal), approx..........................................………..1.85 (15.4) Boiling point, 760 mm Hg, °C (°F), approx.....................................…...........-67 (-88) Melting point, °C (°F), approx.........................................................………..-87 (-124) ...
... Density, 0 °C, g/cm3 (lbs/gal), approx..........................................………..1.85 (15.4) Boiling point, 760 mm Hg, °C (°F), approx.....................................…...........-67 (-88) Melting point, °C (°F), approx.........................................................………..-87 (-124) ...
Examples of Colligative properties are
... At the freezing point, solid and liquid are at equilibrium. The temperature where the equilibrium occurs at a pressure of 1.0 atm is called the normal freezing point. The same entropy effects which cause the boiling point to be elevated in a solution cause the freezing point to be depressed. See the ...
... At the freezing point, solid and liquid are at equilibrium. The temperature where the equilibrium occurs at a pressure of 1.0 atm is called the normal freezing point. The same entropy effects which cause the boiling point to be elevated in a solution cause the freezing point to be depressed. See the ...
AP Lab - MW of Volatile Liquid - North Allegheny School District
... the hot flask is the same as that in the laboratory as shown in the barometer. There is a basic flaw in the method. We obtain the weight of vapor by subtracting the mass of the empty vessel from the final mass, on the assumption that the air content of the flask is the same during each weighing, whi ...
... the hot flask is the same as that in the laboratory as shown in the barometer. There is a basic flaw in the method. We obtain the weight of vapor by subtracting the mass of the empty vessel from the final mass, on the assumption that the air content of the flask is the same during each weighing, whi ...
Chemistry Name: LeChâtlier`s Principle Date: Chemical Equilibrium
... Dynamic Equilibrium: A forward reaction and reverse reaction will happen at the same rate, reaching equilibrium and no net change LeChâtlier’s Principle: When a stress is applied to a system in equilibrium, the reaction will shift to relieve the stress. What is meant by stress? For example: The conc ...
... Dynamic Equilibrium: A forward reaction and reverse reaction will happen at the same rate, reaching equilibrium and no net change LeChâtlier’s Principle: When a stress is applied to a system in equilibrium, the reaction will shift to relieve the stress. What is meant by stress? For example: The conc ...
STORING SAMPLES IN A LIQUID NITROGEN DEWAR
... Cryogens are substances used to produce very low temperatures of below -153 degrees Celsius. Liquid nitrogen, which has a boiling point of -196 degrees Celsius (-320 Fahrenheit), is a liquid that is commonly used in laboratories. All cryogens can be extremely hazardous if not handled properly. Direc ...
... Cryogens are substances used to produce very low temperatures of below -153 degrees Celsius. Liquid nitrogen, which has a boiling point of -196 degrees Celsius (-320 Fahrenheit), is a liquid that is commonly used in laboratories. All cryogens can be extremely hazardous if not handled properly. Direc ...
Chapter 3 PowerPoint Notes
... directly proportional to its temperature in kelvins if the pressure and the number of particles remains constant. • V = Volume • T = Temperature • 1 = before the change • 2 = after the change ...
... directly proportional to its temperature in kelvins if the pressure and the number of particles remains constant. • V = Volume • T = Temperature • 1 = before the change • 2 = after the change ...
ChE 215, Physical Chemistry
... The objective of this course is to introduce the basic topics in Kinetics, Thermodynamics and Statistical Mechanics. In fact this course present collection of distinct topics particularly useful to chemical engineering students. The basic principles in the course are demonstrated by six experiments ...
... The objective of this course is to introduce the basic topics in Kinetics, Thermodynamics and Statistical Mechanics. In fact this course present collection of distinct topics particularly useful to chemical engineering students. The basic principles in the course are demonstrated by six experiments ...
Unit 1 – Matter and Change
... • Magnetic Properties – Two substances can be separated if: • One substance is magnetic and the other is not – Magnet will attract one substance but not the other ...
... • Magnetic Properties – Two substances can be separated if: • One substance is magnetic and the other is not – Magnet will attract one substance but not the other ...























